Our research
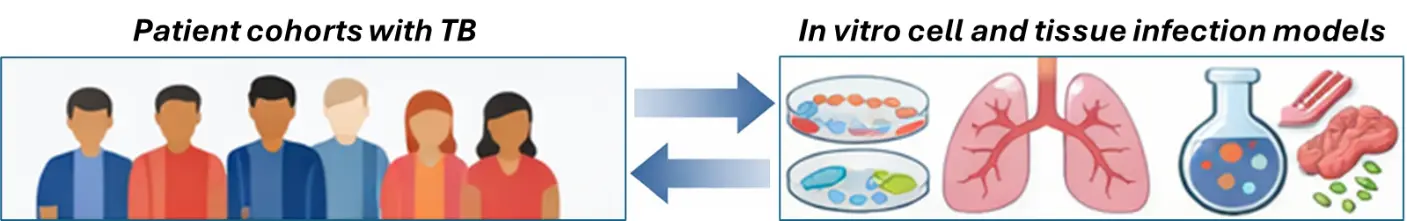
Our research focuses on defining the immune mechanisms that shape human susceptibility and response to Mycobacterium tuberculosis (Mtb). We investigate how cellular immunity and inflammation in tuberculosis (TB) are initiated and regulated by diverse immune cell subsets at the site of infection compared to the peripheral circulation. By integrating clinical samples from well‑characterized patient cohorts with advanced in vitro cell and tissue models, we aim to uncover the pathogenic processes that drive TB disease progression (see image below).
Key Aims
- Identify and correct immune dysfunction in TB by defining antimicrobial effector pathways in macrophages and T cells and developing immunomodulatory strategies to restore effective host immunity.
- Develop precision medicine tools for TB through immune endotyping of heterogenous patient groups to identify prognostic signatures that enable tailored host‑directed therapies that improve outcomes.
- Translate mechanistic discoveries into clinical impact by evaluating host‑directed therapeutic interventions in collaborations with TB‑endemic counties, accelerating the development of individualized treatment options.
Over the years, our work on the cellular and molecular interactions between host cells and Mtb has revealed multiple defects in immune responses among TB patients—defects that limit the ability of immune cells to control and kill bacteria. Building on these discoveries, we now explore strategies to correct Mtb‑induced immune dysregulation using a new class of immunomodulatory compounds designed to strengthen essential antimicrobial pathways.
A central component of our research is the immune endotyping of TB patients, allowing us to identify biologically distinct subgroups and define who may benefit from personalized treatment approaches. This precision‑medicine strategy aims to enhance therapeutic outcomes and reduce the emergence of antibiotic resistance.
Our research program is supported by strong clinical and academic collaborations in TB‑endemic regions such as Ethiopia and Bangladesh, as well as in Sweden. These partnerships enable us to translate fundamental findings into clinical studies and randomized trials evaluating host‑directed therapies (HDTs) in real‑world settings.
Our overarching goal is to accelerate the development of next‑generation, individualized therapies for patients with difficult‑to‑treat TB.
Tuberculosis
TB remains one of the leading infectious causes of death globally. Every second, one person becomes infected with Mtb, and every 25 seconds someone dies from the disease. Despite being curable, TB continues to pose a major public health challenge.
A key threat to global TB control is antimicrobial resistance (AMR). Drug‑resistant TB accounts for roughly one‑third of all AMR‑related infections worldwide. Current treatment relies on complex multidrug regimens taken daily for at least six months, which contributes to the rising incidence of multidrug‑resistant TB (MDR‑TB) and unsatisfactory treatment success rates.
While antibiotics effectively kill intracellular bacteria, they do not address persistent inflammation, tissue destruction, or the immune dysfunction that contributes to relapse and poor clinical outcomes. These gaps underscore the urgent need for host‑directed therapies, interventions that work alongside antibiotics to reduce harmful inflammation, restore antimicrobial immunity, and improve recovery.
Effective host‑directed therapies act on multiple immune pathways, aiming to shorten treatment duration, reduce disease severity, limit adverse effects, and combat drug resistance.
The central role of immunity in TB is evident from clinical observations: individuals with primary immunodeficiencies, HIV infection, or those receiving TNF‑α blockers or immune checkpoint inhibitors e.g. PD‑1 blockade, have a significantly increased risk of developing active TB. These insights highlight that balanced immune regulation is crucial for both protection against infection and effective disease control in TB.

Key collaborations
Karolinska Institutet and Karolinska University Hospital, Stockholm:
- Ass. prof. Magda Lourda, Dept of Laboratory Medicine
- Assoc. prof. Lina Davies Forsman, MD PhD, Dept of Medicine, Solna
- MD PhD Hilmir Asgeirsson, Dept of Medicine, Huddinge
- Prof. Peter Bergman, Dept of Laboratory Medicine
Linköping University, Department of Clinical and Experimental Medicine: Prof. Maria Lerm, PhD Sadaf Kalsum, and Prof. Thomas Schön

International
Addis Ababa University (AAU) and Black Lion University Hospital, Ethiopia: Dr. Amsalu Bekele, Dr. Wondwossen Amogne, Dr. Endale Kassa and Prof. Getachew Aseffa
Armauer Hansen Research Institute (AHRI), Ethiopia: Assoc. prof. Alemseged Abdissa (Scientific Director of AHRI)
AHRI/ALERT MDR-TB clinic, Ethiopia: Dr. Ekram Husein
International Centre for Diarrhoeal Disease Research (icddrb), Bangladesh: Dr. Rubhana Raqib
Jinan University, China: Assoc. prof Yang Zhou

