Our research
We found a new enzyme involved in progeria and discovered that inhibiting this enzyme might increase life quality and life span for these children.
Research on CAAX-proteins raised exciting new possibilities
We perform curiosity-driven basic and translational research into cancer, arthritis, atherosclerosis, heart disease, and ageing. Our research begins with a biochemical pathway by which hundreds of CAAX-proteins, as they are known, are enzymatically modified by a cholesterol-like molecule – which is believed to activate CAAX-proteins by stimulating their interaction with membranes. When these CAAX-proteins (e.g. RAS and prelamin A) are dysfunctional they can cause cancer, inflammation, and ageing-like diseases. Our goals are to define the biochemical importance of the CAAX-protein modifications and thereby identify new strategies to treat these diseases.
Our studies have led to exciting and surprising discoveries and below are two examples.
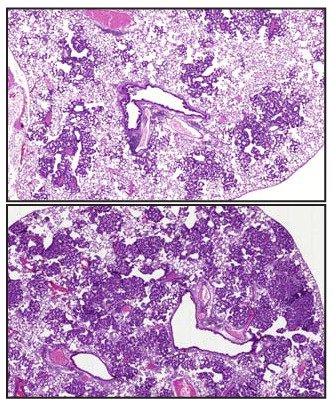
Antioxidants stimulate cancer progression
Healthy people and cancer patients alike use antioxidant supplements, including vitamins A, C, and E, as a daily cancer-fighting strategy despite lack of convincing scientific evidence. We discovered that dietary antioxidants actually accelerate lung cancer growth and metastasis. Antioxidants activate a protein called BACH1 which forces tumour cells to take up glucose and use it for aerobic glycolysis – i.e., the Warburg effect – which drives metastasis. Antioxidants produce similar effects in malignant melanoma and pancreatic cancer. The results suggest that cancer patients should avoid antioxidant supplements and that we may now design drugs that inhibit BACH1-induced glycolysis as a strategy to block metastasis.

A new treatment strategy for children with progeria
Progeria is a rare disease caused by a dysfunctional form of the CAAX-protein prelamin A. Dysfunctional prelamin A causes hair loss, slow growth, osteoporosis, muscle weakness, and death from heart attack or stroke in the teenage years. We discovered that inhibiting ICMT, an enzyme that modifies prelamin A, increases body weight and muscle strength, eliminates osteoporosis, and prevents death in mice with progeria; it also stimulates the growth of cells from children with progeria. We are now developing drugs that inhibit ICMT and preliminary data indicate that our strategy could be successful. But first we have to optimize the drug so it can be tested in children with progeria.
Looking for an internship or a MSc Project?
We are looking for excitable and dedicated master, project, and internship students who want to help us unravel the complex relationships between oxidative stress and metabolic adaptations in cancer cells and determine how these collaborate in tumor initiation, progression, and metastasis. We are also looking for students interested in the biology of accelerated aging who can help us in our efforts to develop and evaluate drugs to treat this deadly disease.
Contact Martin Bergö for more information.


