Our research aims to:
- Link clinical presentation of severe sepsis/septic shock to microbiological aetiology and associated pathogen-specific disease mechanisms.
- Identify central mediators in severe sepsis/septic shock and deduce their mechanistic action and potential value as prognostic risk markers.
- Identify pathogenic mechanisms contributing to severity of necrotizing soft tissue infections.
- Utilize identified disease mechanisms to develop tailored therapy and implement personalized medicine in severe infectious diseases
Of particular interest are the two Gram-positive bacteria Streptococcus pyogenes and Staphylococcus aureus, both of which may cause highly aggressive invasive infections such as toxic shock, necrotizing pneumonia and necrotizing fasciitis that are associated with substantial morbidity and mortality. The research strives to decipher bacterial properties contributing to disease outcome and events underlying tissue injury. A specific theme is to evaluate immunotherapeutic strategies in these diseases.
Keywords
Streptococcus, Staphylococcus, Sepsis, Macrophages, Neutrophils, Inflammation, Soft Tissue Infections
Open positions
If you are interested in doing research in our group, as a degree project, PhD student or postdoc, please feel free to contact the group leader Anna Norrby-Teglund. We are always interested in discussing with talented potential co-workers.
Projects
Disease signatures in group A streptococcal necrotizing soft tissue infections
In this project, we explore the role of macrophages and neutrophils in the necrotizing soft tissue infections caused by group A streptococcus. Specifically we propose to utilize transcriptomic analyses and advanced microscopy to identify and explore disease signatures in tissue biopsies of NSTI patients. Results will be further dissected and validated by use of human organotypic skin tissue models allowing for mechanistic studies of infection in a human tissue-like milieu. The translational nature of the project utilizing a study design based on patient samples, clinical strains, as well as optimized human tissue model systems promotes clinically relevant results. Together this puts us in a unique position to obtain new insight into pathogenic mechanisms of NSTI required to achieve improved diagnostic and therapeutic approaches for these patients.
Systems biology approach to understand necrotizing soft tissue infections; INFECT-project, 2013-2018

The INFECT-project included 14 partners from across Europe, Israel and the US, and was coordinated by Karolinska Institutet (Norrby-Teglund). The overall goal of the project was to advance our understanding of the pathophysiological mechanisms, prognosis, and diagnosis of the multifactorial highly lethal necrotizing soft tissue infections (NSTIs). NSTIs are rapidly spreading infections that may cause extensive soft tissue or limb loss, multiorgan failure and are associated with a considerable fatality rate. There is an urgent need for novel diagnostic and therapeutic strategies in order to improve outcome of NSTIs. To achieve this, a comprehensive and integrated knowledge of diagnostic features, causative microbial agent, treatment strategies, and pathogenic mechanisms (host and bacterial disease traits and their underlying interaction network) was sought. INFECT obtained such insights through an integrated systems biology approach in patients and different clinically relevant experimental models. A key achievement of INFECT was the enrollment of NSTI patients and the creation of the world’s largest patient cohort and associated biobank. Analyses of the comprehensive clinical registry generated an advanced understanding of these patients and underlying disease mechanisms.
Personalized medicine in infectious Diseases: PerAID and PerMIT projects
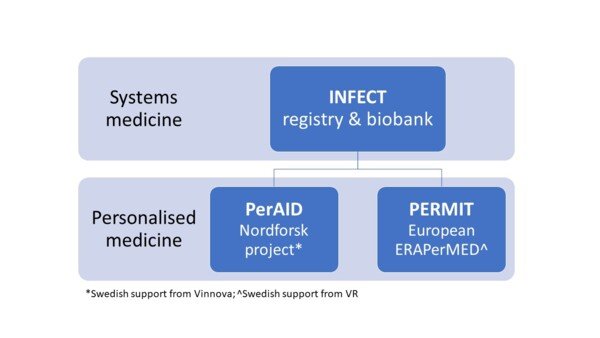
The INFECT clinical registry and biobank now offer a resource for recently started multinational projects, PerAID and PERMIT, building on the advances made through the systems medicine approach to achieve personalized medicine in infectious diseases. The two projects are ambitious covering both severe soft tissue infections and the large heterogeneous group of sepsis. Activities range from establishment of a Nordic platform for personalized medicine in infections, to translational research aimed to identify disease signatures and biomarkers that can be used for individualized therapy, to development of clinical decision support tools.
Key collaborators
- Steinar Skrede, University of Bergen, Norway
- Ole Hyldegaard, Rigshospitalet and University of Copenhagen, Denmark
- Per Arnell, Sahlgrenska Hospital, Sweden
- Michael Nekludov, Karolinska University Hospital, Sweden
- Edoardo Saccenti, Wageningen University and Research, the Netherlands
- Vitor Martin dos Santos, WUR and LifeGlimmer, Berlin, Germany
- Jan-Kristian Damås and Erik Solligård, St Olav’s Hospital and NTNU, Trondheim, Norway
- Annebeth de Vries, Red Cross, the Netherlands
- Malak Kotb and Suba Nookala, University of North Dakota, USA
- Annelies Zinkernagel, University of Zürich, Switzerland
- Nikolai Siemens. Greifswald University, Germany
- John McCormick, Western University, Canada
- Jim Musser, Hoston Medical Center, US
- Fredric Carlsson, Lund University, Sweden

