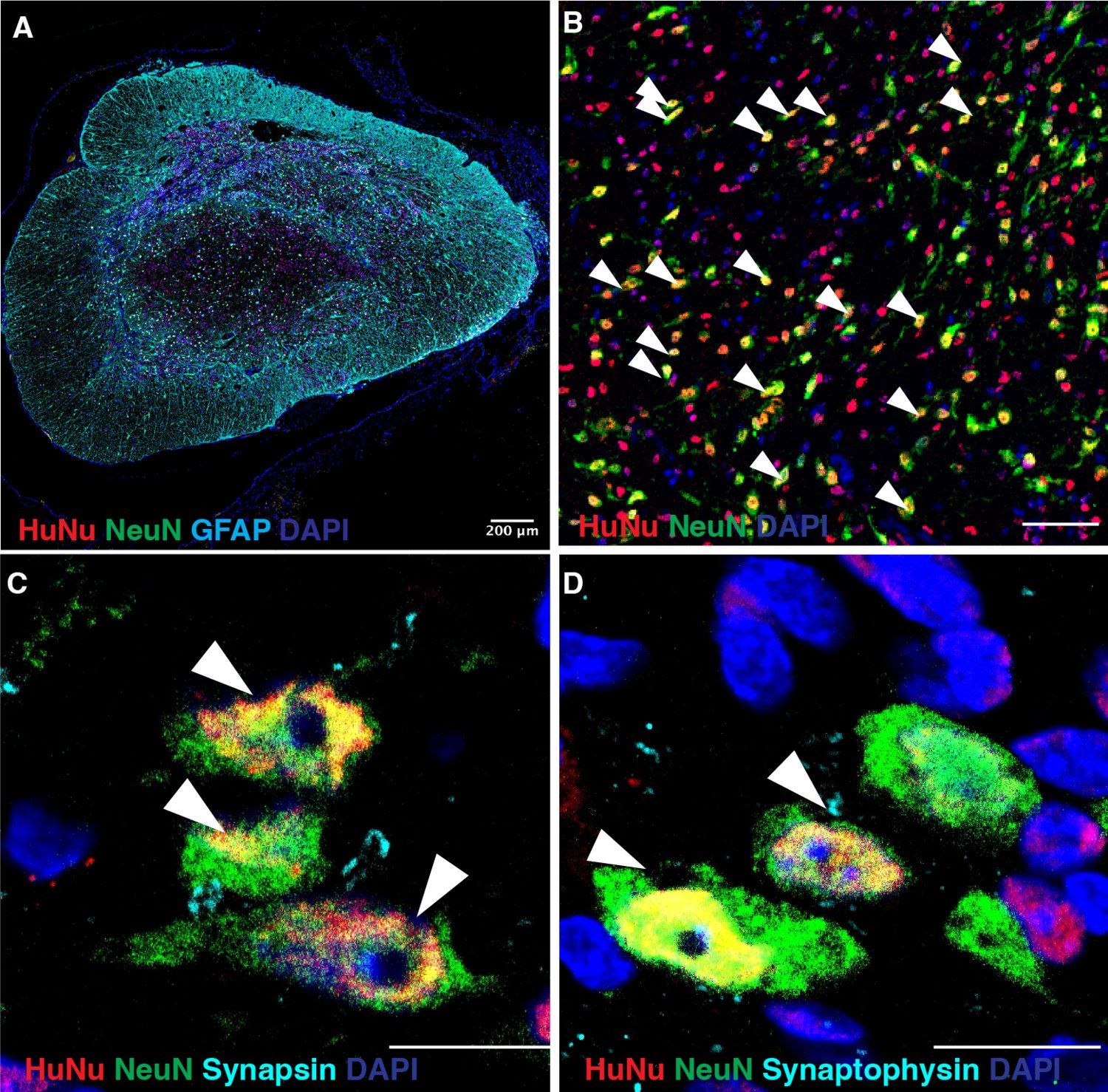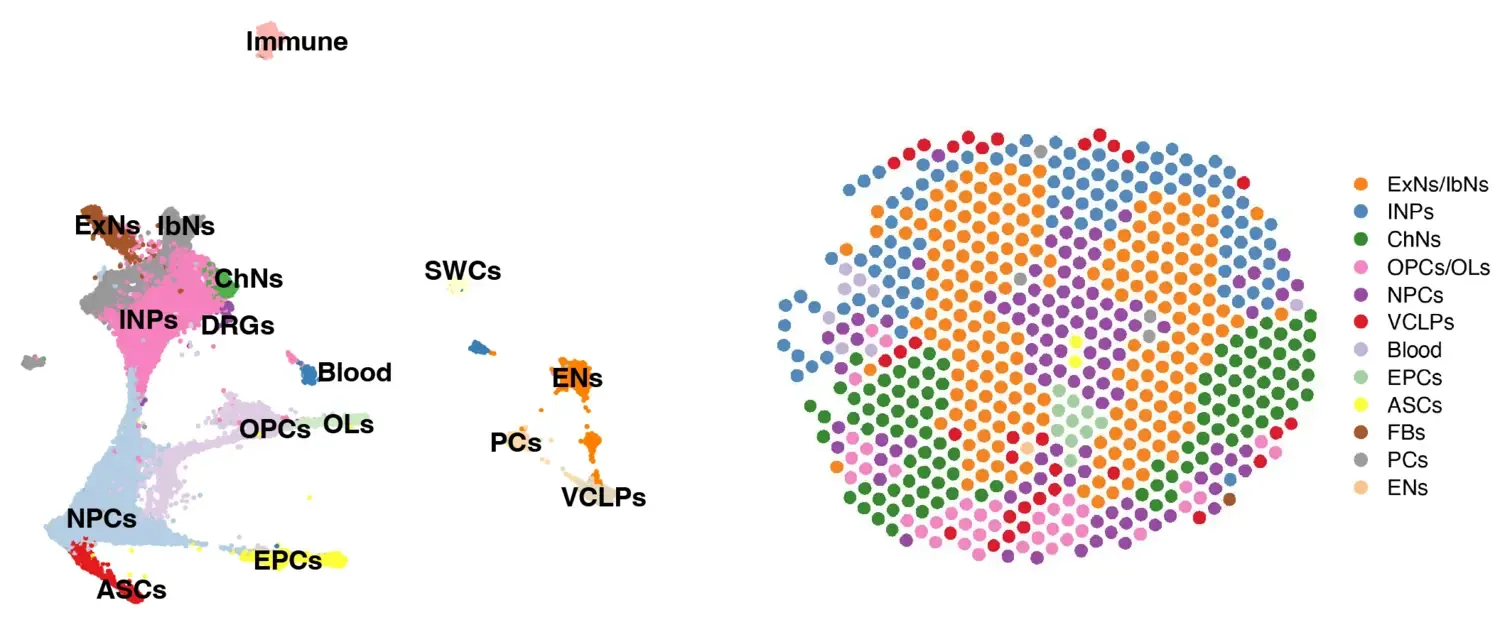
Research focus
Stem cells are the origin of the mammalian system. As stem cells possess the remarkable ability to self-renew and differentiate into diverse specialized cell types, they are the fundamental driving force behind both early embryonic development and the profound regenerative potential required to repair damaged tissues throughout our lives.
Our research explores the intricate mechanisms underlying neurodevelopment and regeneration, with a primary focus on the molecular regulation of neural stem and progenitor cells across the lifespan and following injury in the central nervous system (CNS). Our ultimate goal is to leverage the profound insights of developmental and stem cell biology for regenerative medicine.
To elucidate the molecular and cellular dynamics of stem cells, my lab utilizes cutting-edge technologies, including:
- Single-cell and spatial omics
- Advanced computational algorithms
- Human organoids
- CRISPR and gene editing
Our ongoing research comprises three main directions, all with a strong emphasis on human stem cell biology:
1. Stem Cells and Development
Goal: To investigate the stem cell subtypes that give rise to distinct neural subpopulations in the developing human CNS, and to leverage this knowledge to develop new stem cell therapies for precise cell fate control in CNS injuries.
Our previous work has revealed the vast heterogeneity of different neural cells, including neural stem cells in the developing human brain and spinal cord. However, a major ongoing challenge in the field is that single-cell and spatial omics cannot always fully distinguish between a true "subtype" and a "transient cell state" due to the highly dynamic molecular changes occurring during development.
To overcome this, we are currently utilizing high-resolution spatial transcriptomics on early developmental human CNS tissues and developing novel bioinformatic pipelines to precisely identify spatially organized stem cell subtypes. Moving forward, we aim to translate these findings into advanced treatments for CNS injuries, utilizing combinatorial approaches with genetic perturbation.
2. Stem Cells and Aging
Goal: To elucidate how human neural stem and progenitor cells lose their regenerative potential during maturation and aging, and to discover pathways to rejuvenate the aged CNS.
A fundamental barrier to regenerating a damaged CNS is the loss of robust stem cell properties as the human body matures and ages. It remains highly controversial whether human neural stem cells in adulthood can be safely reactivated, or whether functional neural stem cells even persist in the adult human CNS. Our lab is actively investigating whether these cells exist throughout the human lifespan and mapping the key genetic and epigenetic regulations that drive them into states of quiescence or senescence.
3. Stem Cells and Disease Origin
Goal: To reveal the developmental origins of severe CNS diseases and identify novel therapeutic targets.
Expanding beyond development and regeneration, I launched two new research lines in 2022 focusing on the origins of CNS diseases, specifically targeting pediatric brain tumors (ependymomas) and Alzheimer’s disease (AD).
Our prior research demonstrated that cancer stem cells in ependymomas actively recapitulate neurodevelopment. This revealed critical molecular signatures that can serve as targets for the tumor's origin without interfering with the normal developing brain. We are now applying single-cell and spatial omics to human ependymomas to correlate these molecular mechanisms with patient survival and tumor recurrence. Furthermore, we are investigating whether age-related neurodegenerative diseases like AD may also harbor a developmental origin, utilizing genetically modified animal models alongside human patient-derived organoids.