
- >
- >
- Research area Medicine at KI DS
- Movement, Activity and Health – Eva Broström's research group
- Multidimensional health in old age – Amaia Calderón-Larrañaga group
- Molecular epidemiology of aging – Sara Hägg's research group
- Long noncoding RNA in the adipocyte – Alastair Kerr Research Group
- Leukemia niche – Hong Qian group
- Disease mechanisms in severe acute bacterial infections – Anna Norrby-Teglund group
- Gastrointestinal pediatric surgery – Tomas Wester's research group
- Developmental biology and regenerative medicine – Igor Adameyko's research group
- Experimental audiology – Barbara Canlon's research group
- Translational Immunology in Neuroinflammation – Olivia Thomas' research group
- Sickness absence, health and living conditions – Emilie Friberg's research group
- Health Systems and Policy – Cecilia Stålsby Lundborg's research group
- Endocrinology and autoimmune diseases – Research group Sophie Bensing
- Health economics of Alzheimer disease and other dementias – Linus Jönsson Group
- Epidemiology: Diet, contaminants and health – Agneta Åkesson's research group
- Neuronal circuits of anxiety – Janos Fuzik group
- Paediatric rheumatology – Research group Helena Erlandsson Harris
- Cardiovascular Epidemiology – Karin Leander research group
- Molecular cardiology – Research group Francesco Cosentino
- Björn Högberg group
- Regeneration mechanisms – Andras Simon's Group
- Assessing and managing chemical risks – Linda Schenk's research group
- Autoimmunity and adaptive immunity in rheumatic disease – Research group Vivianne Malmström
- Rheumatic systemic diseases – Team Marie Holmqvist
- Heart failure with reduced and preserved ejection fraction. Clinical and translational aspects. – Research group Lars Lund
- Gene Regulation and Alternative Splicing in development and disease – Ziegenhain Group
- Psychiatric epidemiology – Paul Lichtenstein's research group
- Structural basis of RNA biogenesis – Martin Hällberg's research group
- Hematopoietic Stem Cell Biology Group – Sten Eirik W. Jacobsen Lab
- Psychiatric Genetic Epidemiology – Sarah Bergen's research group
- Renal Medicine at Novum (Baxter Novum) – Bengt Lindholm Group
- Research groups in biochemistry
- Psychological treatment: internet and exposure – Brjánn Ljótsson's research group
- Research groups in immunology
- Research groups in ophthalmology
- Research groups in urology and nephrology
- Research groups in sociology and migration
- Integrative physiology – Juleen Zierath and Anna Krook
- Hormonal signalling and cancer – Cecilia Williams research group
- Cell Biology of Cancer – Staffan Strömblad's research group
- Chronic inflammation with focus on rheumatology and cancer – Research group Per-Johan Jakobsson
- Health services and social work research – Lena Von Koch's research group
- Nitrate-nitrite-NO pathway in health and disease – Eddie Weitzberg's research group
- Medical sensors and diagnostics – Team Onur Parlak
- Uncovering the molecular and physical principles governing early embryonic division and nuclear organization – Jan Ellenberg Group
- Nanoscale lipid flux architecture – Veijo Salo’s Research Group
- Residual conditions after polio and neuromuscular diseases – Eva Melin's research group
- Amyotrophic Lateral Sclerosis - ALS – Caroline Ingre's research group
- Saving the brain in critically ill neonates – Ulrika Ådén's research group
- Regulatory Transcriptomics – Kutter group
- NeuroimAging – Grégoria Kalpouzos group
- Section of Pharmacogenetics – MIS-lab
- Biological and clinical aspects of the myelodysplastic syndrome – Magnus Tobiasson group
- Human innate lymphoid cell lab – Jenny Mjösberg group
- Viruses and their interactions with the immune system – Sara Gredmark Russ group
- Protein degradation pathways – Helin Norberg's research group
- Translational Auditory Neuroscience – Christopher Cederroth's research group
- Neuroradiology: Neurodegeneration and Neuroinflammation – Tobias Granberg's research group
- Tumor Immunology and Immunotherapy Group – Dhifaf Sarhan
- Causal inference in epidemiologic research – Arvid Sjölander's research group
- The role of autophagy in Aβ metabolism and neurodegeneration in Alzheimer’s disease – Per Nilsson group
- Cholinergic Pathobiology & PET-Imaging – Darreh Shori Group
- PROCOME – Hanna Öfverström's and Marta Roczniewska's group
- Molecular brain imaging of neurodegenerative disorders – Andrea Varrone's research group
- Alcohol and Drug Epidemiology – Mats Ramstedt's research group
- Anxiety related disoders and genetics – Christian Rück's research group
- Psychotherapy Research – Tobias Lundgren's research group
- Human olfaction – Mats J. Olsson's research group
- Tissue stem cells and aging – Pekka Katajisto's Group
- SOSVASC – David Lindström's research group
- Yenan Bryceson group
- Epidemiology of sarcoidosis and systemic lupus erythematosus – Team Elizabeth Arkema
- Viral genomics and metagenomics – Tobias Allander Group
- Defining the metabolic signatures of tissue resident human ILCs – Chris Tibbitt team
- Human papillomavirus which causes cervical cancer – Research Group Karin Sundström
- Research groups in bioinformatics and systems biology
- Cognitive epidemiology – Bo Melin's research group
- Gilad Silberberg group
- Research groups in oto-rhino-laryngology and logopaedics
- Immunoregulation of environmental-induced airway inflammation in severe asthma and bronchiectasis – Apostolos Bossios's research group
- Multimorbidity and frailty in older adults – Davide Liborio Vetrano group
- Psychiatric and pharmaco-epidemiology – Zheng Chang's research group
- Charting normal and malignant hematopoiesis – Team Joakim Dahlin
- Molecular mechanisms governing oxygen homeostasis in development, evolution, and disease – Kragesteen's research group
- Precision Medicine – Infrastructure team
- Research groups in medical epigenetics and epigenomics
- Spinal circuits for whole-body motor coordination – Laurence Picton group
- Clinical HIV epidemiology and comorbidity – Christina Carlander Research group
- Head and neck pathology – Anders Näsman's Team
- Carin Håkansta's research group – Digital work environment and health
- Proteomes of Metabolism – Florian Rosenberger's Research Group
- Pediatric and respiratory epidemiology – Catarina Almqvist Malmros' research group
- Sexual and Reproductive Health – Marie Klingberg-Allvin research group
- Angiogenesis, Cancer, Metabolic disease, Cardiovascular disease, Eye disease – Yihai Cao Group
- Cardiovascular and psychosocial health in resilience and healthy brain aging – Chengxuan Qiu Group
- Translational Psychiatry – Catharina Lavebratt's research group
- Rare metabolic liver diseases, coagulation and hepatocellular carcinoma – Stål and Wahlin research group
- Blood Engineering – Vanessa Lundin group
- Development, regulation and function of human innate lymphoid cells throughout life – Jakob Michaëlsson group
- Clinical Genetics – Richard Rosenquist Brandell's research group
- Oxysterols – Ingemar Björkhem and Ulf Diczfalusy research
- Molecular exercise physiology – Carl Johan Sundberg's research group
- Large-scale Network Connectivity in the Human Brain – Peter Fransson's research group
- Environmental physiology
- The Systems Virology Lab – Ujjwal Neogi
- Cancer Cell Invasion – Marco Gerling's research group
- Molecular brain imaging, neuropsychopharmacology, depression and anxiety disorders – Johan Lundberg's research group
- Organization and operation of postural neuronal networks in health and disease – Tatiana Deliagina group
- Paediatrics – Anna Lindholm Olinder's research group
- Petzold group
- Molecular and epidemiological studies of ascending aortic aneurysms – Research group Hanna Björck
- Prevention, Intervention and Mechanisms in Public Health (PRIME Health) – Cecilia Magnusson's research group
- The interplay between circadian 3D genome organization and metabolism in complex diseases – Anita Göndör's Group
- Reconstructive Plastic Surgery and Global Surgery – Jenny Löfgren's research group
- Real-world effectiveness of psychopharmacological treatment – Jari Tiihonen's Research Group
- Center for Heart Failure and Arrhythmia – Gianluigi Savarese
- Sports Medicine – Anders Stålman's research group
- Women's Mental Health Epidemiology – Donghao Lu Lab
- The SMC complexes, chromosome dynamics and stability – Camilla Björkegren's Group
- Extracellular vesicles in immunity – Research group Susanne Gabrielsson
- Precision Cancer Medicine in Lung Cancer - Preclinical, Translational & Clinical Research – Simon Ekman's research group
- RNA-guided DNA repair and cancer – Marianne Farnebo's research group
- Laboratory of translational fertility preservation – Kenny Rodriguez-Wallberg's group
- Research groups in cancer and oncology
- Common Mental Disorders and Behavioral Medicine in Primary Care – Erik Hedman's research group
- Neuronal membrane trafficking – Oleg Shupliakov group
- Research groups in paediatrics
- Biostatistics With Focus on Statistical Methods in Cancer Epidemiology and Screening – Keith Humphreys' research group
- Research groups in nanotechnology
- Gastroenterology – clinical epidemiology and clinical trials – Research group Ola Olén
- Precision engineering of T cells in vivo for therapies of cancer – William Nyberg team
- Protein Misfolding and Prevention by Molecular Chaperones in Cancer and Neurodegenerative Disease – Gefei Chen's research group
- Social Perception – Arvid Guterstam's research group
- Neuroepidemiology – Kyla McKay and Katarina Fink's research group
- Experimental growth research – Lars Sävendahl's research group
- Neuronal circuits of the Basal Ganglia – Maya Ketzef group
- Psychological Interventions - innovation, improvement and implementation – Viktor Kaldo's research group
- Susanna Ranta team
- Neurovascular diseases – Christina Sjöstrand research group
- Lifestyle, Prevention and Health – Ylva Trolle Lagerros research group
- Antibody-mediated neuroinflammation – Susanna Brauner's research group
- Selenoproteins and redox biology – Elias Arnér research group
- Gynecologic Cancer – Henrik Falconer's research group
- B cell biology, from immunodeficiency to cancer – Pan Hammarström Lab
- Developmental Cognitive Neuroscience – Torkel Klingberg group
- Rare Diseases – Ann Nordgren och Anna Lindstrand's research group
- Research on steatotic liver disease – Hagström Group
- Stem Cell Size – Jette Lengefeld Team
- Natural Killer Cell Biology and Cell Therapy – Kalle Malmberg group
- Mast Cell Biology – Research group Gunnar Nilsson
- Traumatic Brain Injuries and Neuromonitoring – Eric Thelin's research group
- Medical Statistics team
- Augmented Neurosurgery – Erik Edström and Adrian Elmi Terander's research group
- Molecular neurophysiology – Karima Chergui's research group
- Injuries Social Aetiology and Consequences (ISAC) – Lucie Laflamme's research group
- Unconventional T cells in human pathology – Andrea Ponzetta team
- Inflammation and immune regulation during infection – Team Christopher Sundling
- Orthopaedics – Anders Enocson's research group
- Endothelial dysfunction in atherosclerotic cardiovascular disease – Research Group John Pernow
- Clinical epidemiology of multiple sclerosis and inflammatory joint diseases – Team Thomas Frisell
- Cancer stem cells and clonal structure in acute lymphoblastic leukemia – Martin Enge's Group
- Mitochondrial metabolism in health and disease – Anna Wredenberg group
- Biomolecular Medicine and Advanced therapies- focus on delivery of RNA therapeutics – Research group Samir EL Andaloussi
- Antimicrobial resistance – Christian Giske research group
- Tissue immunology – Research group Helen Kaipe
- Health Economics and Economic Evaluation – Niklas Zethraeus' group
- Mental Health Neural Dynamics Lab – Kristoffer Månsson's research group
- Stem Cell Biology – Jonas Muhr's Group
- VIVAC research group: Vaccines and Immunotherapies against Viruses And Cancer – Matti Sällberg
- The passage of the mRNP particle through the nuclear pore – Bertil Daneholt's research
- Therapeutic Immunology and Transfusion Medicine – Michael Uhlin’s research group
- Musculoskeletal conditions and sports medicine research – Iben Axén's research group
- Research groups in cancer and oncology
- Nutritional neuroscience – Janina Seubert's Research Group
- Tobias Karlsson's group
- Research groups in pharmacology and toxicology
- Precision Psychiatry – Lu Yi's research group
- Research groups in medicinal chemistry
- Agneta Richter-Dahlfors group
- Cancer epidemiology with focus on breast cancer and applied biostatistics – Anna Johansson's research group
- Autism, ADHD and other neurodevelopmental conditions – Sven Bölte's research group
- Twinstudies on health conditions and sickness absence – Pia Svedberg's research group
- Psychiatric comorbidity and intergenerational transmission of mental health problems – Erik Pettersson's research group
- Genetic and pharmacological epidemiology – Zeberg laboratory
- REACH – digital treatment and prevention – David Moulaee Conradsson's research group
- Mucosal immunology lab – Charlotte Thålin research group
- Stroke – Annika Lundströms research group
- Loh
- Surgery and Urology at KI DS
- Single-cell and spatial omics to investigate mechanisms underlying multiple sclerosis and disease-associated oligodendroglia – Gonçalo Castelo-Branco Group
- Pediatric Neuroimmunology and Epilepsy – Ronny Wickström's research group
- Explores various cell signaling phenomena and their impact on critical biological and medical processes using advanced light microscopy – Per Uhlén Group
- Neural stem cells – Anna Falk group
- Diabetes and Associated Complications – Sergiu-Bogdan Catrina's research group
- Myelodysplastic syndromes – Eva Hellström Lindberg group
- Immune responses to human viral infections and cancer – Hans-Gustaf Ljunggren group
- Health services research in neurological conditions – Charlotte Ytterberg's research group
- Neurosurgery – Mikael A. Svensson's research group
- Westman neuroimaging group
- Stemcells and Inflammation – Lou Brundin's research group
- Pharmacological nitric oxide research – Jon Lundberg's research group
- Epidemiology of Psychiatric Conditions, Substance use and Social Environment (EPiCSS) – Emilie Agardh's & Renee Gardner's Research Group
- Autoimmune neurology – Jakob Theorell team
- Reducing antibiotic resistance – Research group Pontus Nauclér
- Orthopaedics – Karl Eriksson's research group
- Endocrine Surgery – Robert Bränström's research group
- Paediatrics – Inger Kull's research team
- Cancer proteogenomics - from methods to clinical applications – Janne Lehtiö's Group
- Primary Immunodeficiency, Innate Immunity and Antimicrobial peptides – Peter Bergman Research Group/The AMP-group
- Clinical Virology and Immunology – Annika Karlsson's research group
- Jussi Taipale Group
- Global Child Health and the Sustainable Development Goals – Tobias Alfvén's research group
- Topoisomerases, chromatin biology and cancer – Laura Baranello's group
- Stem cells and neural development – Johan Ericson's Group
- Vascular morphogenesis and function in health and disease – Lars Jakobsson group
- Epidemiology and Public Health Intervention Research (EPHIR) – Jette Möller's research group
- Epithelial stem cells in development and disease – Maria Genander's group
- Research Group Gustaf Edgren
- Drug discovery, pancreatic beta-cell regeneration – Olov Andersson's Group
- Unit of Biostatistics – Matteo Bottai
- Research groups in cardiology and cardiovascular diseases
- Sleep, cognition and health – John Axelsson's research group
- Research groups in infectious medicine
- Research groups in physiology and anatomy
- Cardiometabolic epidemiology and aging – Ida Karlsson's research group
- How the overview of research fields is made
- Precision Cancer Control Group – Martin Eklund's research group
- Liver and monocyte remodelling in non-alcoholic fatty liver disease and cardiovascular disease – Rongrong Fan's research group
- Early childhood development and neurodevelopmental conditions – Terje Falck-Ytter's research group
- Mental health and social integration (Mente) – Ellenor Mittendorfer-Rutz's research group
- Health outcomes in autistic people – Mark Taylor's research group
- Mechanisms of protein aggregation and inhibition – Axel Abelein group
- Accelerating drug discovery using molecular modeling and machine learning – Andreas Luttens' group
- Pediatric Healthcare Science – Cecilia Bartholdson research group
- Digital Strategies for AF Detection and Outcome Prevention – Emma Svennberg Team
- Speech-Language Pathology – Liza Bergström's research group
- Arrhythmia – Johan Engdahl's research group
- Research at Ersta Hospital – Anders Thorell's group
- Environment, nutrition and health – Anna Bergström's research group
- Genetic and epigenetic factors in asthma and allergy – Cilla Söderhäll's research group
- Cardiovascular epidemiology – Team Bruna Gigante
- Neurobiology of Stress and Treatment Response – Juan Pablo Lopez group
- Vascular Surgery – Ulf Hedin's research group
- Annika Bergquist group
- Acute myeloid leukemia – Sören Lehmann group
- Human tissue-resident NK cells – Niklas Björkström group
- Neuropsychoimmunology – Sophie Erhardt's research group
- Diabetes epidemiology – Sofia Carlsson's research group
- Stroke - Acute Intervention and Secondary Prevention – Niaz Ahmed's research group
- Molecular muscle physiology and pathophysiology – Lanner Lab
- Community nutrition and physical activity (CoNPA) – Liselotte Schäfer Elinder's research group
- Inflammatory bowel disease (IBD) – Research group Eduardo Villablanca
- CRISPR-based drug target discovery in cancer and autoimmunity – Research group Fredrik Wermeling
- Emergency care – Kristian Ängeby's research group
- Clinical Cancer Epidemiology – Research group Karin Ekström Smedby
- Cancer prevention and screening – Johannes Blom's research group
- Somatosensation – Patrik Ernfors group
- DNA replication & Cancer Genetics – Lemmens Group
- Laboratory testing for alcohol and drugs of abuse – Anders Helander research
- Anaesthesia and Intensive care – Rebecka Rubenson Wahlin/Anna Schandl's research group
- Jonas Fuxe Group
- SCF ubiquitin ligases, cell cycle, transcription and cancer development – Olle Sangfelt's Group
- Toxicological Mechanisms – Emma Wincent's research group
- Stem Cells in Tissue Homeostasis and Regenerative Medicine – Jonas Frisén's Group
- Urology – Olof Akre's research group
- The New World of Work – Theo Bodin's research group
- Midbrain dopaminergic neuron development
- Research group Michael Fored
- Neurobiology of pain & Therapeutics – Saida Hadjab Group
- Risk assessment – Mattias Öberg's research group
- Research groups in cell and molecular biology
- Developmental Psychology: Digital Media and ADHD – Lisa Thorell's research group
- Research groups in education and pedagogy
- Research groups in biostatistics and probability theory
- ESSI – Emotion regulation, Self-injury, Suicide, and Intervention – Johan Bjureberg's research group
- Medical ethics – Gert Helgesson's group
- Treatment of substance use disorders – Johan Franck's Research Group
- Sex hormones and sex differences in diseases of the brain – Ivan Nalvarte's research group
- Cilia in the brain – and their connections to human brain disorders – Peter Swoboda's research group
- Evidence-based methods in autism and ADHD – Tatja Hirvikoski's research group
- Long-Term Outcomes after Perioperative and Intensive Care – Max Bell research group
- Translational and clinical research in acute myeloid leukemia – Team Martin Jädersten
- CEREBRA – Susanne Palmcrantz's research group
- Ute Römling group
- Perioperative quality – Jan Jakobssons research group
- Medical Education and Implementation – Anna Kiessling's research group
- Translational Pharmacology – Kent Jardemark's team
- Global and Sexual Health (GloSH) – Anna Mia Ekström's research group
- Medical Inflammation Research – Rikard Holmdahl group
- Neuro-infections & Neuroinflammation – Federico Iovino Group
- Clinical Chemistry and Blood Coagulation – Jovan Antovic's research group
- Translational research on human microbial infections and its consequences – Anders Sönnerborg's group
- Human tissue-resident NK cells in homeostasis and disease – Nicole Marquardt team
- Etiology and pathogenesis of type 1 diabetes – Malin Flodström-Tullberg group
- Oxygen sensing, cancer and intratumor heterogeneity – Schlisio Lab
- Genetic Epidemiology of Neuroinflammatory Disorders – Ingrid Kockum's research group
- Retina – Anders Kvanta's research group
- Glaucoma – Pete Williams' research group
- Leadership in healthcare and academia – Mia von Knorring's group
- Luminal gastroenterology – Research group Charlotte Hedin
- Identifying molecular signals in the genital mucosa that determine susceptibility to sexually transmitted infections – Research group Kristina Broliden
- Coronary heart disease – Research group Per Svensson
- Educational development – Matti Nikkola's group
- Post-transcriptional regulation in mitochondria – Joanna Rorbach group
- Cellular diversity is generated during development, neuronal identity – Johan Holmberg's Group
- Lipid handling in health and cardiometabolic disease – Research group Carolina Hagberg
- Renal glomerulus biology and diseases – Jaakko Patrakka group
- Research group Caroline Palm Apergi
- Precision pathology and tumor heterogeneity – Johan Hartman's Group
- Lifelong Learning in Health Care Contexts – Terese Stenfors' group
- Translational Arthritis Research – Team Bence Réthi
- Stem cells, Developmental Biology, Heart Disease, Heart Development, Genetics, Biotechnology – Kenneth R. Chien's group
- Urological cancer – Lars Egevad's Group
- The ubiquitin/proteasome system in neurodegenerative diseases and cancer – Nico Dantuma's Group
- Experimental alcohol- and drug dependence research – Vladana Vukojević research group
- Chronic inflammatory disease epidemiology – Research group Johan Askling
- Statistical and bioinformatics analyses of high-throughput molecular data – Yudi Pawitan's research group
- Urban environment and children´s and youth health – Olena Gruzieva's research group
- Research groups in laboratory medicine
- Research groups in medical biotechnology
- Research groups in physiotherapy
- Breast cancer epidemiology – Kamila Czene's research group
- Research groups in human computer interaction
- Research group Liv Eidsmo
- Preventive Medicine – Susanna Larsson's research group
- Transition to adulthood for individuals with neurodevelopmental conditions – Ulf Jonsson's team
- Function and Health in Respiratory and Cardiovascular Conditions – Malin Nygren-Bonnier's research group
- Laboratory of lymphocyte biology – Team Taras Kreslavskiy
- Global Reproductive Outreach & Wellness (GROW) – Michael Wells
- Mats Marshall Heyman research group
- SLE/APS/Vasculitis group – Research group Aleksandra Antovic
- Itziar Martinez Gonzalez group
- CoNFIND Cognition and Fatigue – Marika Möllers research group
- Venous thromboembolism and emergency medicine – Eli Westerlund's research group
- Tissue immunosurveillance by cytotoxic lymphocytes – Takuya Sekine team
- Physical activity and Sports medicine with focus on prevention – Hagströmer research group
- Receptor biology and signaling – Schulte Lab
- Neurobiology of Obesity – Alessandro Furlan group
- Upper GI Surgery – Jesper Lagergren's research group
- Sepsis and COVID-19 – Kristoffer Strålin Group
- Harnessing Neutrophils for Precision Cell Therapy against solid tumors – Roland Fiskesund Team
- Human T cell immunity to evolving viruses and cancers – Marcus Buggert group
- Neuroradiology - MRI physics – Stefan Skare's research group
- Wilhelm lab
- Inflammatory responses in cancer and autoimmunity – Mikael Karlsson's Group
- Cultural Medicine – Solvig Ekblad's unit
- Working life, ergonomics, psychosocial factors, and health – Research group Daniel Falkstedt
- Aging and health – Welmer's research group
- NK Cell Recognition – Klas Kärre Group
- Computational Breast Imaging – Fredrik Strand's Group
- Drug treatment – Erik Eliasson research group
- Neural differentiation as a strategy for neuroblastoma treatment – Marie Arsenian Henriksson Group
- Cancer immunotherapy – Mattias Carlsten group
- Cell-based immune therapy for cancer – Andreas Lundqvist's Group
- Basic molecular structure of human skin – Lars Norlén's research
- Reproductive, Perinatal and Pediatric Epidemiology – Research group Olof Stephansson
- Research group Erik Melén
- Research group Joel Nordin
- MINT – Klas Karlgren's team
- Translational breast cancer research – Theodoros Foukakis' Group
- Genes function and molecular bases of diseases – Research group Magdalena Paolino
- Homeostasis and tissue repair mechanisms in the mammalian system, pericytes, stem cells – Christian Göritz Group
- The Perinatal Epidemiology Lifecourse Lab – Team Neda Razaz
- Experimental Cancer Medicine (ECM)
- Research group Therese Djärv
- Molecular and cellular neuroendocrinology – Tibor Harkany group
- Neuropsychiatric disorders – Håkan Karlsson group
- Research groups in dermatology and venereal diseases
- Evidence-based practice: prevention, intervention and implementation – Pia Enebrink's research group
- Research groups in radiology and medical imaging
- Research groups in psychiatry
- Maria Eriksdotter research group
- Research groups in precision medicine
- Breast cancer surgery – Jana de Boniface's research group
- ICare – Ann Langius-Eklöf's research group
- Immunology and Chronic Disease – Johan Frostegård's research group
- Health in Everyday Life (HELD) – Susanne Guidetti's research group
- Center for Resuscitation Science – Jacob Hollenberg's research group
- Applied Developmental Neurobiology – Carl Sellgren research group
- Reproductive Health/Reproductive Medicine – Kristina Gemzell Danielsson's research group
- Malin Holzmann's research group
- Radiation therapy/ Radiation Oncology – Pehr Lind Group
- Bacteriophages – Andrea Fossati's research group
- APP processing and Abeta localization at super-resolution and glycan biomarkers for Alzheimer disease – Sophia Schedin Weiss's research group
- Rehabilitation medicine at KI DS
- ALGOSH: Algorithmic Management at Work
- Development of autonomic control – Herlenius Research
- Maintenance and expression of mtDNA in disease and ageing – Nils-Göran Larsson Group
- Neuropharmacology - Movement Disorders – Per Svenningsson's research group
- Somatosensation & Gargalesis – Konstantina Kilteni group
- Signal Transduction – Ismael Valladolid Acebes' research group
- Microbiome and Infections – Piotr Nowak Research Group
- Immunological tolerance and transfusion immunology – Petter Höglund group
- Immunopathogenesis and new therapeutic strategies in tuberculosis – Susanna Brighenti group
- Pain and Brain Imaging – Karin Jensen's research group
- Susanne Nylén group
- Hand surgery – Maria Wilckes Research group
- Environmental impact on pulmonary host defence and chronic airflow obstruction – Anders Lindén's research group
- Rehabilitation, collaboration and aging – Elisabeth Rydwik's research group
- Tuberculosis Aerobiology – Antonio Rothfuchs group
- Development of new molecules of biomedical interest through cell-based phenotypic screens – Oscar Fernandez-Capetillo group
- Diversity within the T cell response, T cell memory – Research group Carmen Gerlach
- Clinical and translational melanoma research – Hanna Eriksson's Group
- Meiotic chromosome segregation – Christer Höög's research group
- B cells and autoantibodies in rheumatic disease – Team Caroline Grönwall
- Precision medicine in lymphoid malignancies including CLL; from new targets to real-world assessments – Anders Österborg's Group
- Molecular cancer research – Anthony Wright
- Genome integrity and nucleolar stress in cancer – Jiri Bartek group
- Translational Cardiology (@TransCardio) – Research group Magnus Bäck
- Gene regulation, genome-wide experimental and computational techniques – Rickard Sandberg's Group
- The Helleday Laboratory focuses on harnessing defects in the DNA damage response and metabolism to develop novel therapies
- Small RNAs in cancer development – Weng-Onn Lui's Team
- Geriatric pharmacoepidemiology – Kristina Johnell's research group
- Molecular neurodevelopment and neuro-oncology – Ola Hermanson group
- Research groups in anesthesiology and intensive care
- Gastrointestinal epidemiology – Jonas Ludvigsson's research group
- Health inequalities and minority stress – Richard Bränström's research group
- Research groups in microbiology
- Research groups in orthopaedics
- Hereditary hematological malignancies – Bianca Tesi team
- Mechanisms of establishment of blood vessels – Lars Holmgren's Group
- Substance use prevention – Johanna Gripenberg's research group
- Perioperative care – Ulrica Nilsson's research group
- Mechanisms behind HIV-1 latency and rebound – Peter Svensson's group
- ENGAGE- Enacting health and change through everyday activity – Patomella group
- Diagnosis and treatment of children with asthma and allergy: from molecular diagnosis to intervention – Jon Konradsen's research group
- Translational research in childhood cancer and histiocytic diseases – Nikolas Herold Research group
- Coronary artery disease – research at Danderyd hospital
- Mechanisms of malignant hematopoiesis – Pedro Moura team
- Regulation of B cell activation – Pia Dosenovic Group
- Complex interventions – Team Carina King
- Chronic pain and rehabilitation – Monika Löfgren's research group
- Reproductive endocrinology and metabolism – Elisabet Stener-Victorin's research group
- Tissue engineering – Magdalena Fossum team
- Inflammation and metabolism – Nicolas Pillon's team
- Neurobiology of Sensory Systems – François Lallemend group
- Experimental Traumatology Research Unit
- Genetic Modification of NK cells for Optimized Functions against Cancer – Arnika Wagner team
- NK cells in the development of adaptive immune responses – Benedict Chambers team
- Molecular pain research – Camilla Svensson's research group
- Brain Connectomics – Joana Pereira's research group
- Genomic Science and RNA Biology – Vicent Pelechano group
- Immunotherapy – Robert Harris' research group
- Infectious diseases "Venhälsan" – Jaran Eriksen's research group
- Hjerling-Leffler group
- Respiratory and invasive infections and microbial pathogenesis – Birgitta Henriques-Normark Laboratory
- Multimodal Brain Imaging – Daniel Lundqvist's research group
- Obsessive - Compulsive and Related Disorders Across the Lifespan – David Mataix-Cols research group
- With a focus on proteins for better cancer treatments – Pär Nordlund's Group
- Precision cancer medicine and precision radiotherapy in lung cancer - from molecular mechanisms/biomarkers to clinical trials – Kristina Viktorsson's team
- Cancer Bioinformatics – Nick Tobin's Team
- Lipoproteins and the Immune System – Research group Stephen Malin
- Equity and Health Policy (EHP) – Ann Liljas' research group
- Effects of hypoxia in physiological and pathological contexts, Hypoxia Inducible Factors (HIF) – Randall Johnson's Group
- Chemistry II – Jesper Z. Haeggström group
- Genomic analysis of parasites and viruses, metagenomic sequencing – Björn Andersson's Group
- Translational control of cancer – Ola Larsson's Group
- Gastrointestinal Surgery KI SÖS – Gabriel Sandblom's research group
- Understanding the mechanisms behind hantavirus-mediated pathogenesis – Jonas Klingström group
- The Gynaecological research team – Elisabeth Epstein
- Understanding how life develops – Emma Andersson's Group
- Statistical methods in epidemiology – Paul Dickman's research group
- Neurons and Neural Networks – Ole Kiehn group
- Research groups in developmental biology
- Behavioral, Resilience and Digital Health in Pain – Rikard Wicksell’s research group
- Research groups in neurology
- Research groups in epidemiology, global public health and social medicine
- Nordic Brain Network – Miia Kivipelto's research group
- Resilience and mental health – Serhiy Dekhtyar group
- Forensic behavioral and neuroscience – Katarina Howner's Research Group
- Immune Engineering – Team Leo Hanke
- Chemical and physical exposure in the work environment and health – Jenny Selander's research group
- Mechanobiology of cardiac regeneration – Elif Eroglu's Group
- Tumors of the female genital organs – Miriam Mints' research group
- Psychoneuroimmunology – Mats Lekander's research group
- Upper gastrointestinal surgical – research at Danderyd Hospital
- Research groups in odontology
- Neuroimmunovascular biology – Harald Lund's team
- Improved synthetic DNA structures by evolutionary selection – Erik Benson Group
- Lifestyle and Kidney Cancer – Stephanie Bonn Team
- The research area of Nephrology at KI DS
- Neuroplasticity and Regeneration – Konstantinos Ampatzis group
- Paediatric cell and molecular biology – Hjalmar Brismar's research group
- Quantitative Biology of the Nucleus – Magda Bienko Group
- Cardiometabolic factors, brain aging, and dementia care – Weili Xu group
- Cognitive Neuroscience of Body and Self – Henrik Ehrsson Group
- Surgical Care Science – Pernilla Lagergren's research group
- Clinical and translational studies on viral hepatitis – Soo Aleman group
- Cell and Gene Therapy – Evren Alici group
- Personalized Medicine and Drug Development – Lauschke-lab
- Electrophysiological neuropharmacology – Göran Engberg's research group
- Mechanisms of Pain and Treatment – Eva Kosek's research group
- Functional (Epi)genomics of Neuroinflammatory Diseases – Maja Jagodic's research group
- Molecular basis of gene regulation of diseases – Carsten Daub's research group
- Health risk assessment methodology – Anna Beronius' group
- Medical genetics – Catharina Larsson's Group
- Translational Genetics of Neurodegenerative disease – Caroline Graff's research group
- Development, transcription factors, neurons, dopamine – Thomas Perlmann's Group
- Clinical and translational research within lupus and autoimmunity – Team Ioannis Parodis
- Cell cycle, mitotic entry, and DNA-damage checkpoints – Arne Lindqvist's Group
- Breast Surgery – Irma Fredriksson's research group
- Protein dynamics and mutation – Laura Orellana's Group
- Research group Ali Mirazimi
- Early drug discovery research in chronic inflammatory diseases – Research group Michael Sundström
- Integrative Physiology – Juleen Zierath's research group
- Epigenetics, Synthetic and Systems Biology – Simon Elsässer group
- Clinical Neuroimmunology and Immunomodulation – Fredrik Piehl's research group
- Understanding mechanisms of vaccination – Research group Karin Loré
- Systems regenerative neurobiology – Enric Llorens Group
- Clinicial Cancer Genomics – Johan Lindberg's research group
- Jeanette Hellgren Kotaleski group
- Brain control of food intake and body weight – Björn Meister group
- Research groups in endocrinology and diabetes
- Research groups in gastroenterology and hepatology
- Research groups in neurosciences
- Research groups in respiratory medicine and allergy
- Caring in Community Care – Zarina Nahar Kabir's research group
- Molecular mapping of the nervous system in health and disease – Jan Mulder group
- Child and adolescent psychiatry – Jens Högström's research group
- Genetic mechanisms of ageing – Maria Eriksson's research group
- Digital Psychiatry – Philip Lindner's research group
- Synaesthesia, autism and perception – Janina Neufeld's team
- Vascular complications in cardiometabolic disease – Team Zhichao Zhou
- Urological Research at Danderyd Hospital – Tobias Nordström's research group
- Lars Karlsson team
- Sepsis and microcirculation – Sara Tehrani's research group
- Translational Clinical Orthopaedics – Michael Axenhus Research Group
- Antigen-specific T cell repertoires – Cassotta lab
- The research area of Neurology at Karolinska Institutet – Danderyd Hospital
- Translational Molecular Imaging – Nordberg Lab
- Neuroplasticity – Dagliyan group
- Developmental and Translational Neurobiology – Cristiana Cruceanu's research group
- Microbiota–gut–brain axis and neurodevelopment – Rochellys Diaz Heijtz group
- Thoracic Surgery – Anders Franco-Cereceda's research group
- Electrophysiology – Mats Jensen-Urstad group
- Role of T cells in human host defense – Johan Sandberg group
- Germ cell biology and developmental programming in epigenetic inheritance of diseases – Qiaolin Deng's research group
- Unit for Bioentrepreneurship – Hanna Jansson's group
- Health informatics – Sabine Koch's group
- Cancer, rapid ageing and nutrition – Martin Bergö's research group
- Clinical radiation therapy – Åsa Carlsson Tedgren's Group
- TRANslational Theranostics Group – Thuy Tran
- Pathogenic pathways in Alzheimer Disease – Lars Tjernberg's research group
- Hematological and solid tumors – Research group Xu Dawei
- Developmental Neurogenomics – Michael Ratz's Group
- Notch signaling, ES cells, myogenic and vascular progenit breast cancer – Urban Lendahl's Group
- Particle toxicology – Hanna Karlsson's group
- Mitochondrial dysfunction in Alzheimer Disease – Maria Ankarcrona's research group
- Molecular pathology of the lung and pleura – Katalin Dobra's Group
- Late effects and cancer survivorship after diagnosis and treatment of aggressive lymphoma – Team Sandra Eloranta
- Research Group Anna Nopp
- Coding and non-coding RNAs in cancer – Per Hydbring's Team
- Research group Mikael Björnstedt
- Sister chromatid cohesion in DNA damage responses, DSB repair, Genome Integrity, Gene regulation – Lena Ström's Group
- Inborn Errors of Endocrinology and Metabolism – Anna Wedell's research group
- Functional Precision Medicine – Brinton Seashore-Ludlow's Team
- Understand skin using modern biology – Maria Kasper's Group
- Integrative Epidemiology – Fang Fang's research group
- Molecular Biometry – Roman Zubarev group
- Idiopathic pulmonary fibrosis (IPF) – Research group Magnus Sköld
- Molecular Neuroscience – Carlos Ibáñez group
- Research groups in psychology
- Research groups in forensic science
- Research groups in medical genetics and genomics
- Research groups in nursing
- Research groups in rheumatology and autoimmunity
- Genetic epidemiology of prostate and testicular cancer – Fredrik Wiklund's research group
- Systems biology of aging – Juulia Jylhävä's research group
- Aging – basic mechanisms and interventions – Christian Riedel's research group
- Embryonal, foetal and brain development – Juha Kere's research group
- Research groups in occupational therapy
- Growth and Cartilage Biology – Ola Nilsson's research group
- Infections and immunity in children with cancer – Anna Nilsson's research group
- Oxygenation, early resuscitation in trauma and drug treatment in cardiac arrest – Team Malin Jonsson Fagerlund
- Prehospital Emergency Care – Veronica Vicente's Research Group
- The brain after surgery and trauma - neuroinflammation, cognitive impairment and dementia – Lars I Eriksson research group
- Personalized diet and medications for cognition – Garcia-Ptacek group
- Arthroplasty – Olof Sköldenberg’s research group
- Anna Undeman Asarnoj team
- Neuroinflammatory disorders – Anders Svenningsson's research group
- Visual Neuroscience – Rune Brautaset's research group
- Chemicals and female fertility – Pauliina Damdimopoulou Research Group
- Molecular and cellular exercise physiology – The Ruas Lab
- Neural circuits of cognition – Marie Carlén group
- Colorectal Surgery – Anna Martling and Caroline Nordenvall's research group
- Myeloma group – Evren Alici's research group
- Medicine and history of science – Eva Åhrén's group
- Cardiometabolic diseases – Ping Chen research group
- Anders Mutvei research group
- Ocular Oncology and Pathology – Gustav Stålhammar's research group
- Clinical Neurophysiology – Charith Cooray's research group
- Regulation of Gene Expression during Viral Infection – Gerald McInerney Group
- Social and affective learning and decision-making – Andreas Olsson's research group
- Translational Breast Cancer Research: Long-Term Risk and Endocrine Treatment Benefit – Linda Lindström's Group
- Notch3 and the cerebral small vessel disease CADASIL – from molecular mechanisms to treatment strategies – Helena Karlström's research group
- Dementia and Multimorbidity – Dorota Religa's research group
- Diagnostic Radiology – Lennart Nedar's research group
- Cognitive Neuropsychiatry – Predrag Petrovic's research group
- Chemical carcinogenesis – Ulla Stenius' group
- Metal toxicology – Maria Kippler's research group
- Cellular Stress in Health and Disease – Federico Pietrocola's group
- Autophagy and cardiovascular disease – Team Ewa Ehrenborg
- Research group Birgitta Sander & Birger Christensson
- Innate Immune Regulation – Quirin Hammer team
- Genetics of tumor predisposition and progression in breast cancer – Svetlana Bajalica Lagercrantz's Team
- Implementation and Quality (IMPAQT) – Claudia Hanson's research group
- Structural and Biophysical Immunology – Research group Adnane Achour
- Neural cell diversity in the Enteric Nervous System (ENS) – Ulrika Marklund group
- Sustainable work & occupational safety and health – Emma Brulin's research group
- Health Systems Leadership, Management, and Safety – Carl Savage's group
- Cell and Molecular Immunology - Research group Cecilia Söderberg Naucler
- Brain Circuits – Konstantinos Meletis group
- Petter Ljungman's research group- Health effects of the Ambient Environment
- Neuropsychology of music – Fredrik Ullén group
- Research groups in geriatrics and gerontology
- Research groups in nutrition and dietetics
- Research groups in sports and fitness sciences
- Prostate Cancer – Henrik Grönberg's research group
- Methods of health promotion in the workplace – Lydia Kwak's research group
- Autism and perinatal epidemiology – Sven Sandin research group
- Wengström's research group
- Modelling and simulation with applications to cancer epidemiology and health economics – Mark Clements' research group
- Health Technology Assessment – Monica Hultcrantz’s group
- Pediatric anesthesiology Stockholm – Per-Arne Lönnqvist's research group
- Biophysics – Erdinc Sezgin's Lab
- War, crisis, and security studies
- Systems Medicine – Johan Björkegren research group
- Trauma – Max Gordon's research group
- Obstetrics and Gynecology at KI DS
- Perpetration Prevention – Rahm and Joleby's research group
- SOLIID - Sustainable Organizational Learning, Innovation, Improvement and Development in Health and Social services – Monica Nyström's group
- Social Gerontology – Carin Lennartsson group
- Genetic and molecular basis of nervous system disorders – Andrea Carmine Belin group
- Chronobiology – Petrus lab
- Leukemic stem cells – Petter Woll group
- Nanomedicine and Spatial Biology – Teixeira Lab
- Global Disaster Medicine - Health Needs and Response – Johan von Schreeb's research group
- Translational cardiac and skeletal muscle physiology – Daniel Andersson's research group
- Neuroradiology – Staffan Holmin's research group
- Tumour genomics – Lauri Aaltonen's research group
- Neurobiology of Motor Actions – Abdel El Manira group
- Neuroinflammation in Alzheimer disease – Marianne Schultzberg research group
- Palliative Medicin – Linda Björkhem-Bergman's research group
- Nucleotide metabolism and molecular pharmacology – Sean Rudd's Group
- Cardiovascular Inflammation – Research group Peder Olofsson
- Mesenchymal stem cells – Katarina Le Blanc group
- Luigi De Petris' Team
- Autoimmunity and cancer – Research group Marie Wahren-Herlenius
- Atherothrombosis research – Team Nailin Li
- Precision cancer medicine – Olli Kallioniemi's group
- Research group Stephan Mielke
- Genetics of autoimmune diseases – Team Leonid Padyukov
- Musculoskeletal disorders; planetary health & biopsychosocial approaches – Nina Brodin's research group
- Vascular Functions in Metabolic and CNS Diseases – Ulf Eriksson group
- Sarcoma Genomics – Felix Haglund de Flon's Group
- Team Helene Alexanderson
- Injury and Repair in the Immature Brain – Blomgren group
- Statistical methods for cancer patient survival – Therese Andersson's research group
- Individual differences in cognition – Agneta Herlitz’ research group
- Research groups in health economy, healthcare management and policy
- Research groups in reproductive medicine and gynaecology
- Research groups in structural biology
- Research groups in artificial intelligence
- Work environment, health and productivity – Christina Björklund's research group
- Predictive medicine – Mattias Rantalainen's research group
- Epigenetic regulation of leukemia and normal blood development – Andreas Lennartsson's research group
- Epigenetic mechanisms underlying metabolic-inflammatory diseases – Eckardt Treuter's research group
- The IMPACT research group – Marie Löf
- Stockholm Center for Health Economics (StoCHE) – Emelie Heintz's team
- Towards personalized physiology in Intensive Care – Team Johan Mårtensson
- Ninib Baryawno research group
- Neurohormonal Basis of Physiological and Maladaptive Expressions of Survival Behaviors – Stefanos Stagkourakis group
- Breast cancer precision medicine – Marike Gabrielson's research group
- D-WHIN: Danderyd Wrist and Hand INitiative – Research group
- Gynecological research – at Danderyd Hospital
- Global infections – Research group Anna Färnert
- Translational Microbiome Research and Pandemic Preparedness – Lars Engstrand group
- EcoMind and biological pathways in cognitive aging – Debora Rizzuto group
- Circuits controlling Action and their Evolution – Sten Grillner group
- Mikael Rydén & Niklas Mejhert lab
- Understand Fundamental Immune Mechanisms to Develop New Treatments for Lung Diseases – Tim Willinger group
- Clinical Brain Imaging Methods – Anna Falk Delgado's research group
- Hyperbaric medicine – Peter Lindholm's research group
- Eye movements and vision – Tony Pansell's research group
- Lung toxicology – Lena Palmberg's research group
- The NeuroCardioMetabol Group – Thomas Nyström & Cesare Patrone
- Environmental and genetic factors influence on neurodevelopment – Sandra Ceccatelli group
- Skin wound healing – Research group Ning Xu Landén
- From development to disease: decoding stem cell programs for CNS repair – Erik Sundström's and Xiaofei Li's research group – Erik Sundström-Xiaofei Li group
- Neurodegenerative diseases and Aging – Daniel Ferreira Research Group
- Deep learning models of cancer mechanisms – Avlant Nilsson's Group
- Clinical Physiology – Marcus Carlsson's research group
- Biomaterials for Therapeutics and Regenerative Medicine – Molly Stevens group
- Molecular Mechanisms of Viral Oncogenesis – Maria G. Masucci's Group
- Björn Reinius group
- Quantitative principles in tumour biology – Jean Hausser's Group
- Bone biology – Sara Windahl Research group
- Research group Volkan Özenci
- Genetics and mechanisms of metals – Karin Broberg's research group
- Immunodeficiency Diseases – Lisa Westerberg Group
- Structural studies of fertilisation and zona pellucida module proteins – Luca Jovine's research group
- Tissue regeneration, focusing on fat cells (adipocytes) and obesity – Kirsty Spalding's Group
- Robert Månsson group
- Developmental and Regenerative Stem Cell Medicine – Lanner Lab
- Suicide and mental health lab – Vladimir Carli and Gergö Hadlaczky's group
- Medical, genetic and molecular decoding of neurodevelopmental disorders – Kristiina Tammimies' research group
- Working hours, Recovery and Safety in working life. – Anna Dahlgren's research group
- Research groups in hematology
- SOMIND: Somatic, Mental, and Neurodevelopmental Conditions – Agnieszka Butwicka's research group
- Research groups in drug abuse and addiction
- Research groups in biomaterials science
- Mikael Adner's research group
- Chemical and functional neuroanatomy of pain and stress systems – Tomas Hökfelt group
- Spider silk biology for biomedical applications – Anna Rising's research group
- Cancer Epidemiology and Causal Inference – Research group Maria Feychting
- Protein biochemistry for development of novel medical treatments – Janne Johansson's research group
- Healthcare financing and organization – Clas Rehnberg's group
- Space and environmental physiology – Rodrigo Fernandez-Gonzalo group
- Novel treatment approaches for neuroblastoma – Malin Wickström Näsman research group
- Cancer immune and gene therapy – Rolf Kiessling's Team
- Cell identity regulation in development and regeneration – Amanda Andersson Rolf's Group
- CAIR-LAB: Clinical AI Research Laboratory – group
- Research area orthopedics at KI DS
- Atopic dermatitis – Research group Emma Johansson
- Reproductive endocrinology and metabolism – Angelica Lindén Hirschberg's research group
- Genetic basis for B and T cell recognition and function – Gunilla Karlsson Hedestam Group
- Cognitive Aging and Mental Health – Erika Jonsson Laukka Group
- Molecular and circuit neuropharmacology – Gilberto Fisone group
- The role of immunity in shaping adipose tissue development and metabolism – Jurga Laurencikiene group
- Chronic myeloid malignancies – Johanna Ungerstedt group
- Studies of tissue microbiology and immunology – Mattias Svensson group
- Cellular composition of the human brain and brain cancer – Sten Linnarsson group
- Balance, gait, exercise and physical activity in neurological diseases – Franzén Group
- Global Health Pharmacology and Therapeutics (GH-Pharma) – Eleni Aklillu's research group
- Real-world-data in Multiple Sclerosis – Anna Glaser's research group
- Suicidology and transcultural psychiatry – Marie Dahlin's research group
- Experimental and Clinical Neuroendocrinology – Maria Petersson's research group
- Cutting edge translational research of common and rare skin disease – Research group Jakob Wikström
- Alzheimer’s Disease Risk Factors & Sex-Specific Mechanisms – Silvia Maioli's research group
- Frontal lobes and cognitive failure – Wahlund's research group
- Clinical Pharmacology – Research group Rickard Malmström
- Molecular Vascular Medicine – Team Lars Maegdefessel
- Molecular endocrine pathology – Christofer Juhlin's Group
- Presynaptic mechanisms – Lennart Brodin group
- Malignant melanoma – Hildur Helgadottir's Group
- Precision strategies to eradicate heterogeneous tumor cells in pediatric cancer – Kasper Karlsson's Group
- Respiratory and systemic immune responses in human pulmonary viral infection and inflammation – Research group Anna Smed Sörensen
- Genetic toxicology – Kristian Dreij's group
- Identifying new molecular targets for the treatment of motor neuron diseases – Eva Hedlund's research
- Epigenetics - basic mechanisms and disease – Karl Ekwall's research group
- Forensic medicine – Henrik Druid's Group
- Ageing and Health – Karin Modig's research group
- Perception Neuroscience – Johan Lundström's research group
- Computational medicine and bioinformatics – Trung Nghia Vu's research group
- Toxicology – Bertrand Joseph´s Research group
- Etiology, prevention and treatment of eating disorders – Ata Ghaderi's research group
- Research groups in medical ethics and history of science
- Research groups in occupational and environmental health
- Research groups in surgery
- Research groups in biophysics
- Research Group Johan Ärnlöv
- Narrative in health and social care – Research group
- Cellular and molecular mechanisms underlying intervertebral disk formation and Diversity and plasticity of adipose tissue cell niche – Meng Xie's research team
- Neuroimmunology of cancer – Sébastien Talbot
- Pediatric Systems Immunology – Petter Brodin's research group
- Microvascular oxidative stress in the triad of cardiovascular, metabolic and renal disease – Mattias Carlström group
- Cardio-renal epidemiology – Juan-Jesus Carrero's research group
- Human Translational Genetics – Stefano Romeo Group
- Clinical Pancreatology – Miroslav Vujasinovic research group
- Hemostasis and Thrombosis – Mika Skeppholms research group
- Molecular mechanisms of drug action – Shane Wright's team
Reproductive endocrinology and metabolism – Elisabet Stener-Victorin's research group
Our group's research activities aim to yield new key information on the pathophysiology of polystic ovary syndrome (PCOS), the most common female endocrine and metabolic disorder.

Latest News
Videos

Get inspired by a professor - Elisabet Stener-Victorin

Meet Elisabet Stener-Victorin, Professor at Karolinska Institutet

Vad är PCOS? Professor Elisabet Stener-Victorin svarar
 Photo: Creative Commons CC0
Photo: Creative Commons CC0Make a donation to our research
Your support means a lot to the success of research. This allows us to go further in our efforts to improve human health through research and education.
About us
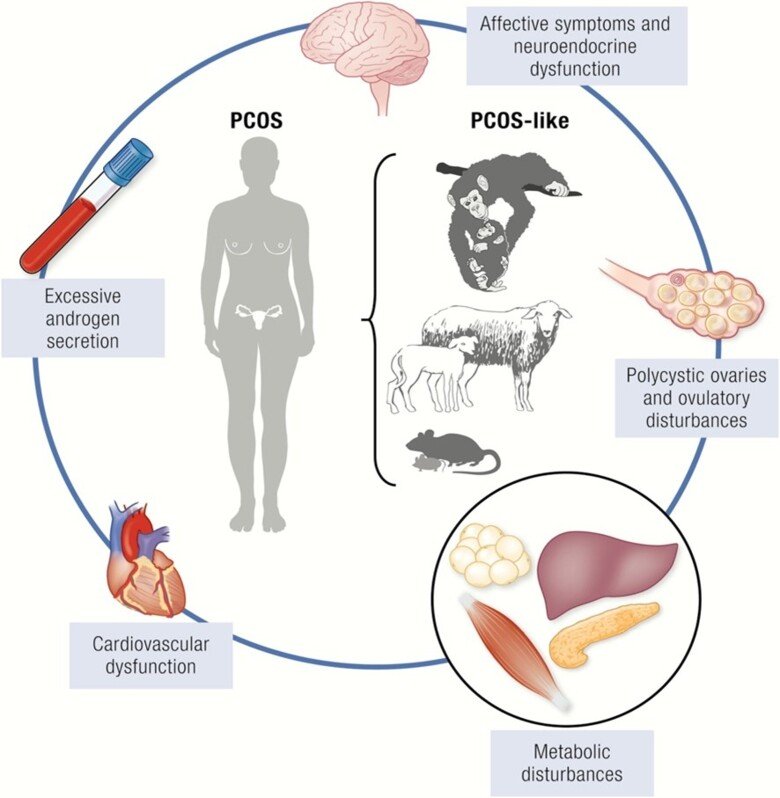
Our research aims to gain deeper understanding of the heritability of PCOS and associated comorbidities such as type 2 diabetes and mental-health disorders across generations by dissecting the molecular, mechanistic, and causal links, and to determine whether men can also transmit the disease. We use different PCOS-like mice models to delineate the main trigger of PCOS and define whether germ cells drive the transmission of the syndrome in the absence of PCOS-associated gene variants. Moreover, we translate our findings in mice to the clinical situation and perform detailed phenotyping of women with and without PCOS and collect serum/blood and endometrium, fat and skeletal muscle tissue biopsies and investigate the effect of different treatments.
Group members

Elisabet Stener-Victorin
Research group leaderI completed my PhD in the field of reproductive endocrinology at the University of Gothenburg (GU). This was followed by postdoctoral studies in Japan followed by research position at GU funded by Swedish Research Council (VR). 2015 I was recruited to the Department of Physiology and Pharmacology at KI and I moved with my research group. I am PI for the Reproductive Endocrinology and Metabolism (REM)-group and our research focus on dissecting genetic versus epigenetic heritability in the transmission of PCOS and associated comorbidities such as type 2 diabetes, endometrial dysfunctions, and mental-health disorders across generations by dissecting the molecular, mechanistic, and causal links, and to determine whether men can also transmit the disease. Recently we also start to investigate the role of the immune system in the pathology of PCOS.
Location: Biomedicum B0552

Sara Torstensson
PhD studentI have a Master of Science in Biomedicine and the overarching objective of my PhD project is to determine the role of the immune system in the pathology of polycystic ovary syndrome (PCOS). Using various in vivo models, I aim to characterize the immune profile and study the underlying mechanisms as well as the risk of transgenerational transmission.
Location: Biomedicum B0551

Haojiang Lu
PhD studentI have a Master of Science in Biomedicine and in my PhD I am studying the transgenerational inheritance of PCOS-like phenotypes in both male and female offspring; and trying to dissect possible mechanisms of such transmission. Mouse models are important part of my project, while in vitro experiments and sequencing techniques are also applied with the help of specialists.
Location: Biomedicum B0553

Gustaw Eriksson
PhD studentI have a Master of Science in Bioinformatics at Lund University, and I start my PhD studies in the Reproductive Endocrinology and Metabolism (REM) group during the fall 2021. My main objective is to generate and analyze single cell sequencing data from tissue biopsies from women with and without PCOS. Moreover, we have recently established endometrial organoids to unravel underlying mechanisms.
Location: Biomedicum B0553

Congru Li
PhD, PostdocI completed my PhD of Genetics at Chinese Academy of Sciences in Beijing. My research interest focusing on the epigenetic mechanisms of female reproductive diseases including embryo development and offspring health. I have experience and expertise in epigenetic and transcriptomic single cell and bulk sequencing and bioinformatic analysis. I am involved in single cell RNA sequencing and DNA methylation of tissue biopsies from women with and without PCOS.
Location: Biomedicum B0553

Eva Lindgren
MSc, Research EngineerI have a bachelor’s degree in biomedical laboratory science and started to work at KI 1980. Since 2016 I am working as Lab manager in the REM group. I have many administrative tasks in the group, and contributing to many of the research project, specifically the human cell culture projects.
Location: Biomedicum B0551

Anja Dekanski
PhD studentI am a PhD student at the Reproductive Endocrinology and Metabolism group lead by Elisabet Stener-Victorin. I come from Serbia, I graduated from Karolinska Institutet's Master in Biomedicine in 2023, and did my Bachelor degree at University College Maastricht in the Netherlands.
My current research focuses on understanding cellular-level functional changes in adipose and endometrium tissue from women with polycystic ovary syndrome. I work mostly with primary human adipose and endometrium cell cultures, including in vitro differentiation and organoids. However, my work also includes basic sequencing-based bioinformatics and clinical data analysis.

Tina Gorsek Sparovec
PhD, PostdocI completed my PhD in Obstetrics and Gynecology at the Medical University of Graz. The overarching objective of my post doctoral project is to dissect molecular, mechanistic and casual links that contribute to skeletal muscle dysfunction in PCOS. Using state-state-of-the-art approaches including single nuclei RNA sequencing, extracellular vesicles and in vitro cell culture models from our unique cohort, I aim to identify PCOS-specific fingerprint in skeletal muscle and underlying changes in tissue cross talk.
Camille Gauthier
PhD, PostdocI completed my PhD in human reproduction at the University of Tours (France) in 2023. I am currently a postdoctoral researcher working on reproductive endocrinology. My research focuses on developing an immunodeficient polycystic ovary syndrome (PCOS)-like mouse model to facilitate the evaluation of new therapeutics while avoiding immune-mediated drug clearance. I have expertise in in vitro experiments on mouse models, and histological analyses.
Location: Biomedicum B0551
Tanja Turunen
PhD, PostdocI completed my PhD related to human reproduction at the University of Eastern Finland in 2024. My current research focuses on cell type-specific endometrial dysfunctions across the menstrual cycle in polycystic ovary syndrome. My work is supported by the Sigrid Jusélius Fellowship (Finland) and the MSCA Postdoctoral Fellowship (European Commission). I have experience in epigenetic and transcriptomic analyses at bulk and single-cell levels as well as in cell culture experiments.
Location: Biomedicum B0553
Lena Schobloch
Research AssistantI am a Research Assistant in the Reproductive Endocrinology and Metabolism group and recently graduated from the Master of Science in Biomedicine at Karolinska Institutet (2025). My research focuses on investigating adipose tissue dysfunction in women with PCOS, using in vitro 2D and 3D adipose spheroid models derived from primary human adipocytes. In addition, I contribute to ongoing projects within the group by assisting with experiments or other laboratory tasks as needed.
Location: Biomedicum B0553
Publications
Selected publications
- Editorial: SCIENCE. 2024;384(6701):1174-1175Stener-Victorin E
- Review: NATURE REVIEWS DISEASE PRIMERS. 2024;10(1):27Stener-Victorin E; Teede H; Norman RJ; Legro R; Goodarzi MO; Dokras A; Laven J; Hoeger K; Piltonen TT
All publications from group members
- Journal article: INTERNATIONAL JOURNAL OF SPORT NUTRITION AND EXERCISE METABOLISM. 2026;36(2):65-75Podlogar T; Grmek B; Pungercar L; Cirnski S; Sparovec TG; Rowlands DS
- Preprint: BIORXIV. 2026Torstensson S; Lu H; Zhao A; Gauthier C; Eriksson G; Lindgren E; Deng Q; Johansson MH; Chambers BJ; Benrick A; Stener-Victorin E
- Journal article: ALLERGY. 2026Tossavainen T; Turunen T; Martikainen M-V; Kankaanpää M; Malinen M; Roponen M
- Journal article: REPRODUCTIVE MEDICINE AND BIOLOGY. 2026;25(1):e70013Li J; Wu Q; Cong J; Chang H; Ma H-L; Zhang D-J; Wang Y; Zhang R-Z; Kam RKT; Ho CS; Chan MHM; Ma RCW; Ng EHY; Mol BWJ; Stener-Victorin E; Legro RS; Wang CC; Wu X-K
- Article: ADVANCED SCIENCE. 2026;:e09691Jiang H; Derisoud E; Parreira D; Taebnia N; Jannig PR; Shafagh RZ; Zhao A; Li C; Ortiz M; Maliqueo MA; Stener-Victorin E; Lauschke VM; Deng Q
- Journal article: NATURE MEDICINE. 2025;31(12):4314Gao X; Zhao S; Du Y; Yang Z; Tian Y; Zhao J; Yuan X; Santos BR; Wei D; Cui L; Yan J; Qin Y; Shi Y; Tang R; Sun Y; Hu J; Ding L; Song X; Ha L; Li J; Zhou L; Bian Y; Wang Q; Zhao Y; Liang X; Liu J; Ma X; Li X; Gu Z; Li Y; Li J; Xia M; Wang S; Li Y; Qian Y; Ma J; He F; Gao S; Liu X; Li S; Jiang Y; Liu Y; Zhang Y; Zhang X; Tian Y; Zhang C; Zhang H; Zhao S; Zhao H; Zhang H; Spritzer PM; Yildiz BO; Stener-Victorin E; Yong E-L; Ou X-H; Legro RS; Zhao H; Chen Z-J
- Journal article: NATURE MEDICINE. 2025;31(12):4214-4224Gao X; Zhao S; Du Y; Yang Z; Tian Y; Zhao J; Yuan X; Santos BR; Wei D; Cui L; Yan J; Qin Y; Shi Y; Tang R; Sun Y; Hu J; Ding L; Song X; Ha L; Li J; Zhang H; Spritzer PM; Yildiz BO; Stener-Victorin E; Yong E-L; Ou X-H; Legro RS; Zhao H; Chen Z-J
- Article: CELL REPORTS MEDICINE. 2025;6(11):102454Zhao A; Wei Y; Kontidou E; Mahdi A; Jannig PR; Torstensson S; Jiang H; Larsson A; Collado A; Humoud R; Grunler J; Ersga D; Esen BO; Zheng X; Lu H; Lindgren E; Risal S; Zhao J; Stener-Victorin E; Sorensen HT; Pedersen L; Pernow J; Zhou Z; Carlsson S; Catrina S-B; Deng Q
- Journal article: IMETAOMICS. 2025Zeng Z; Wang X; Du H; Xing C
- Article: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 2025;110(11):e3810-e3817Kanina A; Stener-Victorin E; Butwicka A; Oberg AS; Rosenqvist MA; Cesta CE
- Journal article: GENOME MEDICINE. 2025;17(1):112de Sande AH; Turunen T; Bouvy-Liivrand M; Ord T; Palani S; Lahnalampi M; Tundidor-Centeno C; Liljenback H; Virta J; Niskanen H; Jayasingha B; Smalander O-P; Sinkkonen L; Mikkola L; Sauter T; Roivainen A; Lonnberg T; Kaikkonen MU; Heinaniemi M
- Preprint: BIORXIV. 2025Zeng Z; Wang X; Du H
- Journal article: HEREDITY. 2025;134(6):321-330Jokiniemi A; Turunen T; Kohonen M; Magris M; Ritari J; Kuusipalo L; Partanen J; Kekalainen J
- Article: NATURE MEDICINE. 2025;31(6):1925-1938Eriksson G; Li C; Sparovec TG; Dekanski A; Torstensson S; Risal S; Ohlsson C; Hirschberg AL; Petropoulos S; Deng Q; Stener-Victorin E
- Journal article: ENDOCRINE ABSTRACTS. 2025Cao T; Torstensson S; Hirschberg LA; Stener-Victorin E; Consiglio C
- Journal article: ENDOCRINE ABSTRACTS. 2025Dekanski A; Eriksson G; Turunen T; Vesterinen A; Li C; Zheng X; Gorsek-Sparovec T; Piltonen T; Linden HA; Taebnia N; Stener-Victorin E
- Journal article: ENDOCRINE ABSTRACTS. 2025Dekanski A; Li C; Schobloch L; Lindgren E; Wright S; Ferreira D; Lauschke V; Taebnia N; Stener-Victorin E
- Journal article: JOURNAL OF TRANSLATIONAL MEDICINE. 2025;23(1):515Jiang M; Bu W; Wang X; Ruan J; Shi W; Yu S; Huang L; Xue P; Tang J; Zhao X; Su L; Cheng D
- Journal article: INTERDISCIPLINARY MEDICINE. 2025;3(3)Kong M; Wu Z; Zheng Z; Zhang B; Zeng Y; Deng H; Feng D; Zhang W; Li C; Fu X; Wang L
- Review: TRENDS IN ENDOCRINOLOGY AND METABOLISM. 2025;36(5):472-481Stener-Victorin E; Deng Q
- Review: NATURE METABOLISM. 2025;7(5):875-894Loft A; Emont MP; Weinstock A; Divoux A; Ghosh A; Wagner A; Hertzel AV; Maniyadath B; Deplancke B; Liu B; Scheele C; Lumeng C; Ding C; Ma C; Wolfrum C; Strieder-Barboza C; Li C; Truong DD; Bernlohr DA; Stener-Victorin E; Kershaw EE; Yeger-Lotem E; Shamsi F; Hui HX; Camara H; Zhong J; Kalucka J; Ludwig JA; Semon JA; Jalkanen J; Whytock KL; Dumont KD; Sparks LM; Muir LA; Fang L; Massier L; Saraiva LR; Beyer MD; Jeschke MG; Mori MA; Boroni M; Walsh MJ; Patti M-E; Lynes MD; Blueher M; Ryden M; Hamda N; Solimini NL; Mejhert N; Gao P; Gupta RK; Murphy R; Pirouzpanah S; Corvera S; Tang S; Das SK; Schmidt SF; Zhang T; Nelson TM; O'Sullivan TE; Efthymiou V; Wang W; Tong Y; Tseng Y-H; Mandrup S; Rosen ED
- Article: FASEB JOURNAL. 2025;39(8):e70556Samad M; Ulfenborg B; Sani SS; Thorbruegge MB; Shrestha MM; Ohlsson C; Maliqueo M; Stener-Victorin E; Asterholm IW; Benrick A
- Journal article: TOXICS. 2025;13(2):118Wang X; Tian M; Shen Z; Tian K; Fei Y; Cheng Y; Ruan J; Mo S; Dai J; Xia W; Jiang M; Zhao X; Zhu J; Xiao J
- Journal article: PLACENTA. 2025;160:147Jiang H; Derisoud E; Parreira D; Jannig PR; Vanky E; Stener-Victorin E; Deng Q
- Journal article: ENVIRONMENTAL HEALTH AND PREVENTIVE MEDICINE. 2025;30:35Fei Y; Cheng Y; Wang X; Ruan J; Zheng D; Cao H; Wang X; Wang X; Zhao X; Yang J
- Journal article: TOXICS. 2024;12(12):918Cheng Y; Fei Y; Xu Z; Huang R; Jiang Y; Sun L; Wang X; Yu S; Luo Y; Mao X; Zhao X
- Journal article: TOXICS. 2024;12(12):908Bu W; Cui Y; Jin Y; Wang X; Jiang M; Huang R; Egbobe JO; Zhao X; Tang J
- Journal article: BIORESOURCE TECHNOLOGY. 2024;413:131539Li Q; Lu H; Tian T; Zhang H; Cheng F; Li X; Sun H; Wang X; Zhou J
- Journal article: ECLINICALMEDICINE. 2024;78:102927Teede HJ; Gibson M; Laven J; Dokras A; Moran LJ; Piltonin T; Costello M; Mousa A; Joham AE; Tay CT; International PCOS Network
- Article: MULTIPLE SCLEROSIS JOURNAL. 2024;30(14):1765-1774Jiang Y; Cesta CE; Liu Q; Kingwell E; Stridh P; Shchetynsky K; Olsson T; Kockum I; Stener-Victorin E; Jiang X; Manouchehrinia A
- Review: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 2024;109(12):3293-3305Cooney LG; Gyorfi K; Sanneh A; Bui LM; Mousa A; Tay CT; Teede H; Stener-Victorin E; Brennan L
- Journal article: SCIENTIFIC REPORTS. 2024;14(1):23014Schmidt-Christensen A; Eriksson G; Laprade WM; Pirzamanbein B; Hoernberg M; Linde K; Nilsson J; Skarsfeldt M; Leeming DJ; Mokso R; Verezhak M; Dahl A; Dahl V; Oennerhag K; Oghazi MR; Mayans S; Holmberg D
- Journal article: ECOTOXICOLOGY AND ENVIRONMENTAL SAFETY. 2024;283:116842Wang X; Wang X; Cheng Y; Luo C; Xia W; Gao Z; Bu W; Jiang Y; Fei Y; Shi W; Tang J; Liu L; Zhu J; Zhao X
- Journal article: PLACENTA. 2024;154:e5Derisoud E; Jiang H; Ortiz M; Li C; Zhao A; Maliqueo MA; Stener-Victorin E; Deng Q
- Conference publication: EUROPEAN JOURNAL OF IMMUNOLOGY. 2024;54:33Torstensson S; Lu H; Zhao A; Risal S; Lindgren E; Eriksson G; Chambers B; Johansson M; Benrick A; Stener-Victorin E
- Article: ADVANCED SCIENCE. 2024;11(36):e2309429Lu H; Jiang H; Li C; Derisoud E; Zhao A; Eriksson G; Lindgren E; Pui H-P; Risal S; Pei Y; Maxian T; Ohlsson C; Benrick A; Haider S; Stener-Victorin E; Deng Q
- Article: JOURNAL OF ENDOCRINOLOGICAL INVESTIGATION. 2024;47(9):2249-2260Subramanian N; Wiik A; Rullman E; Melin M; Lundberg TR; Flanagan J; Holmberg M; Dekanski A; Dhejne C; Arver S; Gustafsson T; Laurencikiene J; Andersson DP
- Journal article: BMC CANCER. 2024;24(1):1046Fan Z; Liu Y; Wang X; Xu Y; Huang R; Shi W; Qu Y; Ruan J; Zhou C; Zhao X; Liu L
- Journal article: EUROPEAN MEDICAL JOURNAL REPRODUCTIVE HEALTH. 2024Stener-Victorin E
- Journal article: MOLECULAR AND CELLULAR ENDOCRINOLOGY. 2024;589:112235Gauthier C; Raynaud P; Jean-Alphonse F; Vallet A; Vaugrente O; Jugnarain V; Boulo T; Gauthier C; Reiter E; Bruneau G; Crepieux P
- Review: ACTA OBSTETRICIA ET GYNECOLOGICA SCANDINAVICA. 2024;103(8):1615-1624Kataoka J; Stener-Victorin E; Schmidt J; Larsson I
- Journal article: JOURNAL OF TRANSLATIONAL MEDICINE. 2024;22(1):623Matilainen J; Berg V; Vaittinen M; Impola U; Mustonen A-M; Mannisto V; Malinen M; Luukkonen V; Rosso N; Turunen T; Kakela P; Palmisano S; Arasu UT; Sihvo SP; Aaltonen N; Harkonen K; Caddeo A; Kaminska D; Pajukanta P; Kaikkonen MU; Tiribelli C; Kakela R; Laitinen S; Pihlajamaki J; Nieminen P; Rilla K
- Article: ADVANCED SCIENCE. 2024;11(28):e2401772Torstensson S; Ascani A; Risal S; Lu H; Zhao A; Espinosa A; Lindgren E; Johansson MH; Eriksson G; Barakat M; Karlsson MCI; Svensson C; Benrick A; Stener-Victorin E
- Preprint: BIORXIV. 2024He W; Luo Q; Zhao J; Wang M; Feng L; Zhao A; Reda A; Lindgren E; Strukenborg J-B; Chen J; Deng Q
- Editorial: SCIENCE. 2024;384(6701):1174-1175Stener-Victorin E
- Article: SCIENTIFIC REPORTS. 2024;14(1):13495Kataoka J; Olsson M; Lindgren E; Larsson I; Schmidt J; Benrick A; Stener-Victorin E
- Article: JOURNAL OF ENDOCRINOLOGY. 2024;261(3):e230204Strauss L; Junnila A; Warri A; Manti M; Jiang Y; Loyttyniemi E; Stener-Victorin E; Lagerquist MK; Kukoricza K; Heinosalo T; Blom S; Poutanen M
- Review: NATURE REVIEWS DISEASE PRIMERS. 2024;10(1):27Stener-Victorin E; Teede H; Norman RJ; Legro R; Goodarzi MO; Dokras A; Laven J; Hoeger K; Piltonen TT
- Newspaper article: EUREKALERT. 2024
- Preprint: RESEARCH SQUARE. 2024Schmidt-Christensen A; Eriksson G; Laprade WM; Pirzamanbein B; Hörnberg M; Linde K; Nilsson J; Skarsfeldt M; Leeming DJ; Mokso R; Verezhak M; Dahl A; Dahl V; Önnerhag K; Oghazi MR; Mayans S; Holmberg D
- Article: BMC GENOMICS. 2024;25(1):208Burns K; Mullin BH; Moolhuijsen LME; Laisk T; Tyrmi JS; Cui J; Actkins KV; Louwers YV; Estonian Biobank Research Team; Davis LK; Dudbridge F; Azziz R; Goodarzi MO; Laivuori H; Mägi R; Visser JA; Laven JSE; Wilson SG; FinnGen; International PCOS Consortium; Day FR; Stuckey BGA
- Preprint: BIORXIV. 2024Torstensson S; Ascani A; Risal S; Lu H; Zhao A; Espinosa A; Lindgren E; Johansson M; Eriksson G; Barakat M; Karlsson MCI; Svensson C; Benrick A; Stener-Victorin E
- Journal article: FOOD AND CHEMICAL TOXICOLOGY. 2024;184:114378Liu L; Luo C; Zheng D; Wang X; Wang R; Ding W; Shen Z; Xue P; Yu S; Liu Y; Zhao X
- Doctoral thesis: 2024Lu H
- Article: ELIFE. 2024;12:rp87592Stener-Victorin E; Eriksson G; Shrestha MM; Paris VR; Lu H; Banks J; Samad M; Perian C; Jude B; Engman V; Boi R; Nilsson E; Ling C; Nystrom J; Wernstedt Asterholm I; Turner N; Lanner J; Benrick A
- Journal article: ELIFE. 2024;12Stener-Victorin E; Eriksson G; Mohan Shrestha M; Rodriguez Paris V; Lu H; Banks J; Samad M; Perian C; Jude B; Engman V; Boi R; Nilsson E; Ling C; Nyström J; Wernstedt Asterholm I; Turner N; Lanner J; Benrick A
- Article: SCIENTIFIC REPORTS. 2024;14(1):563Samad M; Ek J; Borchers S; Krieger J-P; Stener-Victorin E; Skibicka KP; Asterholm IW; Benrick A
- Journal article: JOURNAL OF AFFECTIVE DISORDERS. 2024;344:554-562Cheng Y; Zhang Z; Ma X; Wang X; Chen L; Luo Y; Cao X; Yu S; Wang X; Cao Y; Zhao X
- Journal article: FEBS LETTERS. 2024;598(2):220-232Raynaud P; Jugnarain V; Vaugrente O; Vallet A; Boulo T; Gauthier C; Inoue A; Sibille N; Gauthier C; Jean-Alphonse F; Reiter E; Crepieux P; Bruneau G
- Editorial: ACTA OBSTETRICIA ET GYNECOLOGICA SCANDINAVICA. 2024;103(1):7-12Forslund M; Melin J; Stener-Victorin E; Hirschberg AL; Teede H; Vanky E; Piltonen T
- Preprint: BIORXIV. 2023Lu H; Jiang H; Li C; Derisoud E; Zhao A; Eriksson G; Lindgren E; Pui H-P; Risal S; Pei Y; Maxian T; Ohlsson C; Benrick A; Haider S; Stener-Victorin E; Deng Q
- Preprint: ELIFE SCIENCES PUBLICATIONS, LTD. 2023Stener-Victorin E; Eriksson G; Mohan Shrestha M; Rodriguez Paris V; Lu H; Banks J; Samad M; Perian C; Jude B; Engman V; Boi R; Nilsson E; Ling C; Nyström J; Wernstedt Asterholm I; Turner N; Lanner JT; Benrick A
- Article: ADVANCED SCIENCE. 2023;10(34):e2304389Ivanova A; Badertscher L; O'Driscoll G; Bergman J; Gordon E; Gunnarsson A; Johansson C; Munson MJ; Spinelli C; Torstensson S; Vilén L; Voirel A; Wiseman J; Rak J; Dekker N; Lázaro-Ibáñez E
- Preprint: RESEARCH SQUARE. 2023Kataoka J; Olsson M; Lindgren E; Larsson I; Schmidt J; Benrick A; Stener-Victorin E
- Journal article: INTERNATIONAL JOURNAL OF MOLECULAR SCIENCES. 2023;24(21):15961Zehnaker A; Vallet A; Gourdon J; Sarti C; Jugnarain V; Hassan MH; Mathias L; Gauthier C; Raynaud P; Boulo T; Beauclair L; Bigot Y; Casarini L; Crepieux P; Poupon A; Piegu B; Jean-Alphonse F; Bruneau G; Reiter E
- Preprint: BIORXIV. 2023de Sande AH; Turunen T; Bouvy-Liivrand M; Örd T; Palani S; Tundidor-Centeno C; Liljenbäck H; Virta J; Smålander O-P; Sinkkonen L; Sauter T; Roivainen A; Lönnberg T; Kaikkonen MU; Heinäniemi M
- Conference publication: JOURNAL OF THE ENDOCRINE SOCIETY. 2023;7(Supplement_1):bvad114.1587Eriksson G; Li C; Risal S; Pui H-P; Torstensson S; Hirschberg AL; Petropoulos S; Deng Q; Stener-Victorin E
- Journal article: JOURNAL OF THE ENDOCRINE SOCIETY. 2023;7(Supplement_1):bvad114.1678Kempegowda P; Malhotra K; Khalil H; Davitadze M; Tay CT; Mousa A; Team PS; Teede HJ; Brennan L; Stener-Victorin E
- Conference publication: MULTIPLE SCLEROSIS JOURNAL. 2023;29:243Jiang Y; Cesta C; Liu Q; Oberg S; Kingwell E; Stridh P; Shchetynsky K; Olsson T; Kockum I; Stener-Victorin E; Jiang X; Manouchehrinia A
- Article: FERTILITY AND STERILITY. 2023;120(4):767-793Teede HJ; Tay CT; Laven J; Dokras A; Moran LJ; Piltonen TT; Costello MF; Boivin J; M Redman L; A Boyle J; Norman RJ; Mousa A; Joham AE; International PCOS Network
- Article: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 2023;108(10):2447-2469Teede HJ; Tay CT; Laven JJE; Dokras A; Moran LJ; Piltonen TT; Costello MF; Boivin J; Redman LM; Boyle JA; Norman RJ; Mousa A; Joham AE
- Article: HUMAN REPRODUCTION. 2023;38(9):1655-1679Teede HJ; Tay CT; Laven J; Dokras A; Moran LJ; Piltonen TT; Costello MF; Boivin J; Redman LM; Boyle JA; Norman RJ; Mousa A; Joham AE; International PCOS Network
- Preprint: BIORXIV. 2023Raynaud P; Jugnarain V; Vaugrente O; Vallet A; Boulo T; Gauthier C; Inoue A; Sibille N; Gauthier C; Jean-Alphonse F; Reiter E; Crépieux P; Bruneau G
- Article: EUROPEAN JOURNAL OF ENDOCRINOLOGY. 2023;189(2):g43-g64Teede HJ; Tay CT; Laven JJE; Dokras A; Moran LJ; Piltonen TT; Costello MF; Boivin J; Redman LM; Boyle JA; Norman RJ; Mousa A; Joham AE; International PCOS Network
- Review: EUROPEAN JOURNAL OF ENDOCRINOLOGY. 2023;189(2):R1-R9Davitadze M; Malhotra K; Khalil H; Hebbar M; Tay CT; Mousa A; PCOS SEva Team H; Teede H; Brennan L; Stener-Victorin E; Kempegowda P
- Article: ELIFE. 2023;12:e86454Ascani A; Torstensson S; Risal S; Lu H; Eriksson G; Li C; Teschl S; Menezes J; Sandor K; Ohlsson C; Svensson CI; Karlsson MCI; Stradner MH; Obermayer-Pietsch B; Stener-Victorin E
- Journal article: HUMAN REPRODUCTION. 2023;38(Supplement_1)Lu H; Jiang H; Li C; Pui HP; Lindgren E; Risal S; Torstensson S; Eriksson G; Ohlsson C; Benrick A; Stener-Victorin E; Deng Q
- Conference publication: HUMAN REPRODUCTION. 2023;38Lu H; Jiang H; Li C; Pui HP; Lindgren E; Risal S; Torstensson S; Eriksson G; Ohlsson C; Benrick A; Stener-Victorin E; Deng Q
- Preprint: ELIFE SCIENCES PUBLICATIONS, LTD. 2023Stener-Victorin E; Eriksson G; Shresta MM; Perian C; Jude B; Engman V; Boi R; Nilsson E; Ling C; Nyström J; Asterholm IW; Lanner JT; Benrick A
- Article: HUMAN MOLECULAR GENETICS. 2023;32(11):1875-1887Ronn T; Perfilyev A; Jonsson J; Eriksson K-F; Jorgensen SW; Brons C; Gillberg L; Vaag A; Stener-Victorin E; Ling C
- Article: CELL REPORTS MEDICINE. 2023;4(5):101035Risal S; Li C; Luo Q; Fornes R; Lu H; Eriksson G; Manti M; Ohlsson C; Lindgren E; Crisosto N; Maliqueo M; Echiburu B; Recabarren S; Petermann TS; Benrick A; Brusselaers N; Qiao J; Deng Q; Stener-Victorin E
- Journal article: ENDOCRINE ABSTRACTS. 2023Li C; Eriksson G; Lu H; Vannay A; Lindgren E; Hirschberg LA; Deng Q; Stener-Victorin E
- Journal article: ENDOCRINE ABSTRACTS. 2023Eriksson G; Li C; Risal S; Pui H-P; Torstensson S; Linden AH; Petropoulos S; Deng Q; Stener-Victorin E
- Article: JOURNAL OF HUMAN GENETICS. 2023;68(5):347-353Lyle SMM; Ahmed S; Elliott JEE; Stener-Victorin E; Nachtigal MWW; Drogemoller BII
- Newspaper article: HEJA LIVET. 2023
- Article: ENGINEERING. 2023;23:103-111Wu X; Wang CC; Cao Y; Li J; Li Z; Ma H; Gao J; Chang H; Zhang D; Cong J; Wang Y; Wu Q; Han X; Chung PWJ; Li Y; Zheng X; Chen L; Zeng L; Borchert A; Kuhn H; Chen Z-J; Ng EHY; Stener-Victorin E; Zhang H; Legro RS; Mol BWJ; Shi Y
- Preprint: MEDRXIV. 2023Stener-Victorin E; Eriksson G; Shrestha M; Paris R; Lu H; Banks J; Samad M; Perian C; Jude B; Engman V; Boi R; Nilsson E; Ling C; Nyström J; Asterholm W; Turner N; Lanner JT; Benrick A
- Journal article: FRONTIERS IN PHARMACOLOGY. 2023;14:1138452Meng S; Zhu T; Fan Z; Cheng Y; Dong Y; Wang F; Wang X; Dong D; Yuan S; Zhao X
- Preprint: BIORXIV. 2023Ascani A; Torstensson S; Risal S; Lu H; Eriksson G; Li C; Teshl S; Menezes J; Sandor K; Ohlsson C; Svensson C; Karlsson MCI; Stradner MH; Obermayer-Pietsch B; Stener-Victorin E
- Article: COMMUNICATIONS BIOLOGY. 2023;6(1):69Pei Y; Risal S; Jiang H; Lu H; Lindgren E; Stener-Victorin E; Deng Q
- Journal article: FRONTIERS IN PHARMACOLOGY. 2023;13:1098136Hong L; Wang X; Cui W; Wang F; Shi W; Yu S; Luo Y; Zhong L; Zhao X
- Journal article: FRONTIERS IN ONCOLOGY. 2022;12:1054564Gao L; Wang X; Wang X; Wang F; Tang J; Ji J
- Journal article: FRONTIERS IN ENDOCRINOLOGY. 2022;13:1048601Raynaud P; Gauthier C; Jugnarain V; Jean-Alphonse F; Reiter E; Bruneau G; Crepieux P
- Journal article: JOURNAL OF ONCOLOGY. 2022;2022:1-10Zhao H; Wang F; Wang X; Zhao X; Ji J
- Journal article: LANCET DIABETES & ENDOCRINOLOGY. 2022;10(11):E11Joham AE; Norman RJ; Stener-Victorin E
- Article: REPRODUCTIVE BIOLOGY AND ENDOCRINOLOGY. 2022;20(1):153Kataoka J; Larsson I; Lindgren E; Kindstrand LO; Schmidt J; Stener-Victorin E
- Review: ENDOCRINOLOGY. 2022;163(12):bqac164Stener-Victorin E
- Preprint: AUTHOREA. 2022Ma H; Wen X; Li J; Quan K; Lai M; Wen Q; Liu H; Hu Z; Li M; Liu J; Hu M; Li S; Yu C; Huang S; Zheng Y; Lin H; Mai Z; Liang X; Lu L; Pan H; Zhang C; Zhuang Z; Su Y; Zheng Y; Wu T; Ng HYE; Stener-Victorin E
- Journal article: FRONTIERS IN GENETICS. 2022;13:981603Wang F; Wang X; Liu L; Deng S; Ji W; Liu Y; Wang X; Wang R; Zhao X; Gao E
- Journal article: JCI INSIGHT. 2022;7(18):e161940Mao W; Cai Y; Chen D; Jiang G; Xu Y; Chen R; Wang F; Wang X; Zheng M; Zhao X; Mei J
- Journal article: BIOMED RESEARCH INTERNATIONAL. 2022;2022:8577821Gao E; Wang X; Wang F; Deng S; Xia W; Wang R; Wang X; Zhao X; Qian H
- Review: LANCET DIABETES & ENDOCRINOLOGY. 2022;10(9):668-680Joham AE; Norman RJ; Stener-Victorin E; Legro RS; Franks S; Moran LJ; Boyle J; Teede HJ
- Article: DIABETOLOGIA. 2022;65(9):1483-1494Liu Q; Tang B; Zhu Z; Kraft P; Deng Q; Stener-Victorin E; Jiang X
- Preprint: RESEARCH SQUARE. 2022Kataoka J; Larsson I; Lindgren E; Kindstrand LO; Schmidt J; Stener-Victorin E
- Journal article: FRONTIERS IN ONCOLOGY. 2022;12:995929Tang H; Shan J; Liu J; Wang X; Wang F; Han S; Zhao X; Wang J
- Preprint: RESEARCH SQUARE. 2022Deng Q; Pei Y; Risal S; Jiang H; Lu J; Lindgren E; Stener-Victorin E
- Journal article: FRONTIERS IN ONCOLOGY. 2022;12:955979Gu C; Lin C; Zhu Z; Hu L; Wang F; Wang X; Ruan J; Zhao X; Huang S
- Journal article: CELLS. 2022;11(14):2147Turunen T; Magris M; Malinen M; Kekalainen J
- Journal article: FRONTIERS IN GENETICS. 2022;13:925231Zeng L; Wang X; Wang F; Zhao X; Ding Y
- Journal article: ENDOCRINE ABSTRACTS. 2022Torstensson S; Ascani A; Lu H; Risal S; Eriksson G; Li C; Benrick A; Obermayer-Pietsch B; Stener-Victorin E
- Journal article: ENDOCRINE ABSTRACTS. 2022Ascani A; Torstensson S; Risal S; Lu H; Teschl S; Eriksson G; Li C; Menezes J; Sandor K; Svensson C; Stradner M; Obermayer-Pietsch B; Stener-Victorin E
- Journal article: ENDOCRINE ABSTRACTS. 2022Risal S; Li C; Luo Q; Fornes R; Manti M; Lu H; Eriksson G; Ohlsson C; Lindgren E; Crisosto N; Maliqueo M; Echiburu B; Recabarren S; Sir PT; Benrick A; Brusselaers N; Qiao J; Deng Q; Stener-Victorin E
- Journal article: ENDOCRINE ABSTRACTS. 2022Deng Q; Pei Y; Risal S; Jiang H; Stener-Victorin E
- Journal article: ENDOCRINE ABSTRACTS. 2022Eriksson G; Li C; Pui H-P; Risal S; Hirschberg ALN; Petropoulos S; Deng Q; Stener-Victorin E
- Journal article: ENDOCRINE ABSTRACTS. 2022Stener-Victorin E
- Journal article: ENDOCRINE ABSTRACTS. 2022Eriksson G; Nilsson E; Boi R; Nystrom J; Stener-Victorin E; Benrick A
- Journal article: ENDOCRINE ABSTRACTS. 2022Kataoka J; Olsson M; Larsson I; Schmidt J; Stener-Victorin E
- Journal article: BIOMEDICINES. 2022;10(5):1065Krstic J; Deutsch A; Fuchs J; Gauster M; Sparovec TG; Hiden U; Krappinger JC; Moser G; Pansy K; Szmyra M; Gold D; Feichtinger J; Huppertz B
- Letter: HUMAN REPRODUCTION. 2022;37(5):1098-1100Ma H; Hu M; Wen Q; Stener-Victorin E
- Article: INTERNATIONAL JOURNAL OF MOLECULAR SCIENCES. 2022;23(9):4939Mohan Shrestha M; Wermelin S; Stener-Victorin E; Wernstedt Asterholm I; Benrick A
- Conference publication: EUROPEAN JOURNAL OF HUMAN GENETICS. 2022;30(SUPPL 1):41-42Batkovskyte D; McKenzie F; Taylan F; Simsek-Kiper PO; Nikkel SM; Ohashi H; Miyahara H; Eriksson G; Ha T; Utine GE; Chiu T; Shimizu K; Hammarsjo A; Boduroglu K; Arts P; Babic M; Jackson MR; Papadogiannakis N; Lindstrand A; Nordgren A; Barnett CP; Scott HS; Chagin AS; Nishimura G; Grigelioniene G
- Journal article: STEM CELL RESEARCH. 2022;60:102671Sparovec TG; Markert UR; Reif P; Schoell W; Moser G; Feichtinger J; Mihalic ZN; Kargl J; Gargett CE; Gold D
- Journal article: INTERNATIONAL JOURNAL OF MOLECULAR SCIENCES. 2022;23(7):3428Lukasiewicz A; Huhta K; Ritari J; Perasaari J; Allinen P; Malinen M; Jokiniemi A; Turunen T; Partanen J; Kekalainen J
- Article: HUMAN REPRODUCTION. 2022;37(3):542-552Wen Q; Hu M; Lai M; Li J; Hu Z; Quan K; Liu J; Liu H; Meng Y; Wang S; Wen X; Yu C; Li S; Huang S; Zheng Y; Lin H; Liang X; Lu L; Mai Z; Zhang C; Wu T; Ng EHY; Stener-Victorin E; Ma H
- Article: BMC MEDICINE. 2022;20(1):66Liu Q; Zhu Z; Kraft P; Deng Q; Stener-Victorin E; Jiang X
- Preprint: SSRN ELECTRONIC JOURNAL. 2022Risal S; Li C; Luo Q; Fornes R; Lu H; Eriksson G; Manti M; Ohlsson C; Lindgren E; Crisosto N; Maliqueo M; Echiburú B; Recabarren S; Petermann TS; Benrick A; Brusselaers N; Qiao J; Deng Q; Stener-Victorin E
- Book chapter: 50 BIG DEBATES IN REPRODUCTIVE MEDICINE. 2021;p. 35-36Stener-Victorin E
- Review: NATURE REVIEWS ENDOCRINOLOGY. 2021;17(9):521-533Stener-Victorin E; Deng Q
- Editorial: TRENDS IN MOLECULAR MEDICINE. 2021;27(8):723-724Stener-Victorin E; Deng Q
- Article: HUMAN REPRODUCTION. 2021;36(8):2382-2391Jiang X; Deng Q; Stener-Victorin E
- Conference publication: REPRODUCTIVE SCIENCES. 2021;28(SUPPL 1):62ACoggin-Carr D; McElroy-Yaggy K; Taylor PD; David AL; Stener-Victorin E; Jetton T
- Journal article: SCIENTIFIC REPORTS. 2021;11(1):10815Jax E; Muller I; Borno S; Borlinghaus H; Eriksson G; Fricke E; Timmermann B; Pendl H; Fiedler W; Klein K; Schreiber F; Wikelski M; Magor KE; Kraus RHS
- Article: NATURE COMMUNICATIONS. 2021;12(1):2431Davegardh C; Sall J; Benrick A; Broholm C; Volkov P; Perfilyev A; Henriksen TI; Wu Y; Hjort L; Brons C; Hansson O; Pedersen M; Wurthner JU; Pfeffer K; Nilsson E; Vaag A; Stener-Victorin E; Pircs K; Scheele C; Ling C
- Article: FASEB JOURNAL. 2021;35(4):e21299Wu Y; Chanclon B; Micallef P; Stener-Victorin E; Wernstedt Asterholm I; Benrick A
- Journal article: NATURE COMMUNICATIONS. 2021;12(1):1064Bourgneuf C; Bailbe D; Lamaziere A; Dupont C; Moldes M; Farabos D; Roblot N; Gauthier C; D'Argent EM; Cohen-Tannoudji J; Monniaux D; Feve B; Movassat J; di Clemente N; Racine C
- Article: ENDOCRINE CONNECTIONS. 2021;10(2):154-163Mellembakken JR; Mahmoudan A; Morkrid L; Sundstrom-Poromaa I; Morin-Papunen L; Tapanainen JS; Piltonen TT; Hirschberg AL; Stener-Victorin E; Vanky E; Ravn P; Jensen RC; Andersen MS; Glintborg D
- Article: TRANSLATIONAL PSYCHIATRY. 2021;11(1):45Risal S; Manti M; Lu H; Fornes R; Larsson H; Benrick A; Deng Q; Cesta CE; Rosenqvist MA; Stener-Victorin E
- Journal article: COMPUTATIONAL AND STRUCTURAL BIOTECHNOLOGY JOURNAL. 2021;19:1944-1955Turunen T; de Sande AH; Polonen P; Heinaniemi M
- Article: FASEB JOURNAL. 2020;34(11):14440-14457Manti M; Pui H-P; Edstrom S; Risal S; Lu H; Lindgren E; Ohlsson C; Jerlhag E; Benrick A; Deng Q; Stener-Victorin E
- Review: FRONTIERS IN PHYSIOLOGY. 2020;11:573505Manti M; Stener-Victorin E; Benrick A
- Article: ACUPUNCTURE IN MEDICINE. 2020;38(5):310-318Li J; Wu W; Stener-Victorin E; Ng EHY; Li RHW; Li M; Liu H; Lai M; Meng Y; Zheng Y; Xia Y; Ma H
- Journal article: AGING-US. 2020;12(18):18251-18273Fan Y; Li C; Pei W; Tan T; Li R; Qiao J; Yu Y
- Review: ENDOCRINE REVIEWS. 2020;41(4):bnaa010Stener-Victorin E; Padmanabhan V; Walters KA; Campbell RE; Benrick A; Giacobini P; Dumesic DA; Abbott DH
- Journal article: CURRENT OPINION IN ENDOCRINE AND METABOLIC RESEARCH. 2020;12:72-77Stener-Victorin E
- Article: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 2020;105(6):dgaa165-2041Benrick A; Pillon NJ; Nilsson E; Lindgren E; Krook A; Ling C; Stener-Victorin E
- Article: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 2020;105(6):dgz326-1936Joo YY; Actkins K; Pacheco JA; Basile AO; Carroll R; Crosslin DR; Day F; Denny JC; Velez Edwards DR; Hakonarson H; Harley JB; Hebbring SJ; Ho K; Jarvik GP; Jones M; Karaderi T; Mentch FD; Meun C; Namjou B; Pendergrass S; Ritchie MD; Stanaway IB; Urbanek M; Walunas TL; Smith M; Chisholm RL; Kho AN; Davis L; Hayes MG; International PCOS Consortium
- Journal article: JOURNAL OF THE ENDOCRINE SOCIETY. 2020;4(Supplement_1):mon-035Lu H; Manti M; Risal S; Lindgren E; Benrick A; Deng Q; Stener-Victorin E
- Journal article: JOURNAL OF THE ENDOCRINE SOCIETY. 2020;4(Supplement_1):mon-032Manti M; Pui H-P; Edström S; Risal S; Lu H; Lindgren E; Jerlhag E; Benrick A; Deng Q; Stener-Victorin E
- Journal article: JOURNAL OF THE ENDOCRINE SOCIETY. 2020;4(Supplement_1):or20-or02Benrick A; Wu Y; Stener-Victorin E; Asterholm IW
- Journal article: JOURNAL OF THE ENDOCRINE SOCIETY. 2020;4(Supplement_1):or31-or03Wagner M; Yoshihara M; Douagi I; Damdimopoulos A; Panula S; Petropoulos S; Lu H; Pettersson K; Palm K; Katayama S; Hovatta O; Kere J; Lanner F; Damdimopoulou P
- Corrigendum: NATURE COMMUNICATIONS. 2020;11(1):2158Hayes MG; Urbanek M; Ehrmann DA; Armstrong LL; Lee JY; Sisk R; Karaderi T; Barber TM; McCarthy MI; Franks S; Lindgren CM; Welt CK; Diamanti-Kandarakis E; Panidis D; Goodarzi MO; Azziz R; Zhang Y; James RG; Olivier M; Kissebah AH; Reproductive Medicine Network; Stener-Victorin E; Legro RS; Dunaif A
- Study protocol: TRIALS. 2020;21(1):239Huang S; Hu M; Ng EHY; Stener-Victorin E; Zheng Y; Wen Q; Wang C; Lai M; Li J; Gao X; Wang X; Hu Z; Xia T; Hu R; Liu J; Wen X; Li S; Quan K; Liang X; Shang H; Ma H; Qiao J
- Article: NATURE COMMUNICATIONS. 2020;11(1):1147Wagner M; Yoshihara M; Douagi I; Damdimopoulos A; Panula S; Petropoulos S; Lu H; Pettersson K; Palm K; Katayama S; Hovatta O; Kere J; Lanner F; Damdimopoulou P
- Conference publication: REPRODUCTIVE SCIENCES. 2020;27(SUPPL 1):160ACarr DJ; McElroy-Yaggy K; Taylor PD; David AL; Stener-Victorin E; Jetton T
- Article: CARDIOVASCULAR RESEARCH. 2020;116(3):619-632Manti M; Fornes R; Pironti G; Haworth SM; Zhengbing Z; Benrick A; Carlstrom M; Andersson D; Stener-Victorin E
- Preprint: RESEARCH SQUARE. 2020Huang S; Hu M; Ng EHY; Stener-Victorin E; Zheng Y; Wen Q; Wang C; Lai M; Li J; Gao X; Wang X; Hu Z; Xia T; Hu R; Liu J; Wen X; Li S; Quan K; Liang X; Shang H; Ma H; Qiao J
- Preprint: RESEARCH SQUARE. 2020Huang S; Hu M; Ng EHY; Stener-Victorin E; Zheng Y; Wen Q; Wang C; Lai M; Li J; Gao X; Wang X; Hu Z; Xia T; Hu R; Liu J; Wen X; Li S; Quan K; Liang X; Shang H; Ma H; Qiao J
- Article: PLOS ONE. 2019;14(12):e0226074Piltonen TT; Ruokojarvi M; Karro H; Kujanpaa L; Morin-Papunen L; Tapanainen JS; Stener-Victorin E; Sundrstrom-Poromaa I; Hirschberg AL; Ravn P; Glintborg D; Mellembakken JR; Steingrimsdottir T; Gibson-Helm M; Vanky E; Andersen M; Arffman RK; Teede H; Falah-Hassani K
- Article: BMJ OPEN. 2019;9(12):e033780Ollila M-M; Kiviniemi A; Stener-Victorin E; Tulppo M; Puukka K; Tapanainen J; Franks S; Morin-Papunen L; Piltonen T
- Article: BMC PREGNANCY AND CHILDBIRTH. 2019;19(1):479Edvinsson A; Hellgren C; Kallak TK; Akerud H; Skalkidou A; Stener-Victorin E; Fornes R; Spigset O; Lager S; Olivier J; Sundstrom-Poromaa I
- Journal article: PLOS GENETICS. 2019;15(12):e1008517Day F; Karaderi T; Jones MR; Meun C; He C; Drong A; Kraft P; Lin N; Huang H; Broer L; Magi R; Saxena R; Laisk T; Urbanek M; Hayes MG; Thorleifsson G; Fernandez-Tajes J; Mahajan A; Mullin BH; Stuckey BGA; Spector TD; Wilson SG; Goodarzi MO; Davis L; Obermayer-Pietsch B; Uitterlinden AG; Anttila V; Neale BM; Jarvelin M-R; Fauser B; Kowalska I; Visser JA; Andersen M; Ong K; Stener-Victorin E; Ehrmann D; Legro RS; Salumets A; McCarthy MI; Morin-Papunen L; Thorsteinsdottir U; Stefansson K; Styrkarsdottir U; Perry JRB; Dunaif A; Laven J; Franks S; Lindgren CM; Welt CK
- Article: CLINICAL ENDOCRINOLOGY. 2019;91(6):750-758Kataoka J; Larsson I; Bjorkman S; Eliasson B; Schmidt J; Stener-Victorin E
- Article: NATURE MEDICINE. 2019;25(12):1894-1904Risal S; Pei Y; Lu H; Manti M; Fornes R; Pui H-P; Zhao Z; Massart J; Ohlsson C; Lindgren E; Crisosto N; Maliqueo M; Echiburu B; de Guevara AL; Sir-Petermann T; Larsson H; Rosenqvist MA; Cesta CE; Benrick A; Deng Q; Stener-Victorin E
- Article: INTERNATIONAL JOURNAL OF OBESITY. 2019;43(11):2176-2188Fornes R; Manti M; Qi X; Vorontsov E; Sihlbom C; Nystrom J; Jerlhag E; Maliqueo M; Hirschberg AL; Carlstrom M; Benrick A; Stener-Victorin E
- Article: REPRODUCTIVE BIOMEDICINE ONLINE. 2019;39(5):868-876Li J; Wu Q; Wang CC; Wang R; Ng EHY; Liu J-P; Mol BWJ; Wu X-K; Li W-T; Kuang H-Y; Ma H-L; Gao J-S; Xie L-Z; Hou L-H; Hu Z-X; Shao X-G; Ge J; Zhang J-F; Xue H-Y; Xu X-F; Liang R-N; Ma H-X; Yang H-W; Huang D-M; Sun Y; Hao C-F; Du S-M; Ding C-F; Gao Y-Q; Wu T-X; Stener-Victorin E; Zhang H; Legro RS
- Journal article: NATIONAL SCIENCE REVIEW. 2019;6(5):993-1003Xu X; Li G; Li C; Zhang J; Wang Q; Simmons DK; Chen X; Wijesena N; Zhu W; Wang Z; Wang Z; Ju B; Ci W; Lu X; Yu D; Wang Q-F; Aluru N; Oliveri P; Zhang YE; Martindale MQ; Liu J
- Preprint: RESEARCH SQUARE. 2019Huang S; Hu M; Ng EHY; Stener-Victorin E; Zheng Y; Wen Q; Li J; Yu C; Liu J; Wang C; Lai M; Wang X; Wen X; Li S; Quan K; Shang H; Ma H; Qiao J
- Review: MEDICAL SCIENCES. 2019;7(8):E86-86Stener-Victorin E; Manti M; Fornes R; Risal S; Lu H; Benrick A
- Article: CELLS. 2019;8(6):578Clarkson YL; Weatherall E; Waterfall M; McLaughlin M; Lu H; Skehel PA; Anderson RA; Telfer EE
- Article: ENDOCRINE CONNECTIONS. 2019;8(6):709-717Lingaiah S; Morin-Papunen L; Piltonen T; Sundstrom-Poromaa I; Stener-Victorin E; Tapanainen JS
- Article: REPRODUCTIVE BIOMEDICINE ONLINE. 2019;38(6):990-998Chang H; Xie L; Ge H; Wu Q; Wen Y; Zhang D; Zhang Y; Ma H; Gao J; Wang CC; Stener-Victorin E; Ng EHY; Wu X
- Journal article: JOURNAL OF THE ENDOCRINE SOCIETY. 2019;3(Supplement_1):sat-227Risal S; Pei Y; Lu H; Manti M; Fornes R; Zhao Z; Lindgren E; Benrick A; Deng Q; Stener-Victorin E
- Journal article: JOURNAL OF THE ENDOCRINE SOCIETY. 2019;3(Supplement_1):sat-201Lu H; Risal S; Manti M; Fornes R; Zhao Z; Lindgren E; Benrick A; Deng Q; Stener-Victorin E
- Journal article: JOURNAL OF THE ENDOCRINE SOCIETY. 2019;3(Supplement_1):mon-217Manti M; Fornes R; Pironti G; Haworth SM; Zhuge Z; Hirschberg AL; De Castro Barbosa T; Benrick A; Carlström M; Andersson D; Stener-Victorin E
- Journal article: JOURNAL OF THE ENDOCRINE SOCIETY. 2019;3(Supplement_1):sat-226Chanclón B; Fornes R; Stener-Victorin E; Asterholm IW; Benrick A
- Review: ENDOCRINE CONNECTIONS. 2019;8(3):R71-R75Rodgers RJ; Avery JC; Moore VM; Davies MJ; Azziz R; Stener-Victorin E; Moran LJ; Robertson SA; Stepto NK; Norman RJ; Teede HJ
- Study protocol: BMJ OPEN. 2019;9(1):e024733Stener-Victorin E; Zhang H; Li R; Friden C; Li D; Wang W; Wang H; Chang C; Li S; Huo Z; Zhang H; Ji X; Linden-Hirschberg A; Qiao J
- Article: PLOS GENETICS. 2018;14(12):e1007813Day F; Karaderi T; Jones MR; Meun C; He C; Drong A; Kraft P; Lin N; Huang H; Broer L; Magi R; Saxena R; Laisk T; Urbanek M; Hayes MG; Thorleifsson G; Fernandez-Tajes J; Mahajan A; Mullin BH; Stuckey BGA; Spector TD; Wilson SG; Goodarzi MO; Davis L; Obermayer-Pietsch B; Uitterlinden AG; Anttila V; Neale BM; Jarvelin M-R; Fauser B; Kowalska I; Visser JA; Andersen M; Ong K; Stener-Victorin E; Ehrmann D; Legro RS; Salumets A; McCarthy MI; Morin-Papunen L; Thorsteinsdottir U; Stefansson K; Styrkarsdottir U; Perry JRB; Dunaif A; Laven J; Franks S; Lindgren CM; Welt CK
- Article: JOURNAL OF THE ENDOCRINE SOCIETY. 2018;2(12):1363-1380Lindheim L; Manti M; Fornes R; Bashir M; Czarnewski P; Diaz OE; Seifert M; Engstrand L; Villablanca EJ; Obermayer-Pietsch B; Stener-Victorin E
- Article: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 2018;103(12):4465-4477Nilsson E; Benrick A; Kokosar M; Krook A; Lindgren E; Kallman T; Martis MM; Hojlund K; Ling C; Stener-Victorin E
- Article: JOURNAL OF EXPERIMENTAL MEDICINE. 2018;215(10):2536-2553Loyher P-L; Hamon P; Laviron M; Meghraoui-Kheddar A; Goncalves E; Deng Z; Torstensson S; Bercovici N; de Chanville CB; Combadiere B; Geissmann F; Savina A; Combadiere C; Boissonnas A
- Article: CLINICAL ENDOCRINOLOGY. 2018;89(3):251-268Teede HJ; Misso ML; Costello MF; Dokras A; Laven J; Moran L; Piltonen T; Norman RJ
- Article: HUMAN REPRODUCTION. 2018;33(9):1602-1618Teede HJ; Misso ML; Costello MF; Dokras A; Laven J; Moran L; Piltonen T; Norman RJ
- Journal article: CELL DISCOVERY. 2018;4:41Li C; Fan Y; Li G; Xu X; Duan J; Li R; Kang X; Ma X; Chen X; Ke Y; Yan J; Lian Y; Liu P; Zhao Y; Zhao H; Chen Y; Yu Y; Liu J
- Journal article: JAMS JOURNAL OF ACUPUNCTURE AND MERIDIAN STUDIES. 2018;11(4):201Carr D; Jetton T; Stener-Victorin E; Taylor P; David A
- Article: FERTILITY AND STERILITY. 2018;110(3):364-379Teede HJ; Misso ML; Costello MF; Dokras A; Laven J; Moran L; Piltonen T; Norman RJ
- Article: FASEB JOURNAL. 2018;32(8):4158-4171Manti M; Fornes R; Qi X; Folmerz E; Hirschberg AL; Barbosa TDC; Maliqueo M; Benrick A; Stener-Victorin E
- Article: MEDICINES. 2018;5(2):E51-51Oh B; Yeung A; Klein P; Larkey L; Ee C; Zaslawski C; Knobf T; Payne P; Stener-Victorin E; Lee R; Choi W; Chun M; Bonucci M; Lang H-D; Pavlakis N; Boyle F; Clarke S; Back M; Yang P; Wei Y; Guo X; Weng C-HD; Irwin MR; Elfiky AA; Rosenthal D
- Journal article: ENDOCRINE ABSTRACTS. 2018Women with PCOS display reduced cardiac vagal activity which is dependent on metabolic abnormalitiesOllila M-M; Kiviniemi AM; Stener-Victorin E; Puukka K; Ruokonen A; Tapanainen JS; Franks S; Morin-Papunen L; Piltonen T
- Letter: CHINESE JOURNAL OF INTEGRATIVE MEDICINE. 2018;24(5):399-400Kuang H-Y; Li Y; Zhang H-P; Legro RS; Stener-Victorin E
- Article: FERTILITY AND STERILITY. 2018;109(5):888-899Dokras A; Stener-Victorin E; Yildiz BO; Li R; Ottey S; Shah D; Epperson N; Teede H
- Article: HUMAN REPRODUCTION. 2018;33(4):617-625Li J; Wu Q; Wu X-K; Zhou Z-M; Fu P; Chen X-H; Yan Y; Wang X; Yang Z-W; Li W-L; Stener-Victorin E; Legro RS; Ng EH-Y; Zhang H; Mol BWJ; Wang CC
- Preprint: BIORXIV. 2018Day F; Karaderi T; Jones MR; Meun C; He C; Drong A; Kraft P; Lin N; Huang H; Broer L; Magi R; Saxena R; Laisk-Podar T; Urbanek M; Hayes MG; Thorleifsson G; Fernandez-Tajes J; Mahajan A; Mullin BH; Stuckey BGA; Spector TD; Wilson SG; Goodarzi MO; Davis L; Obermeyer-Pietsch B; Uitterlinden AG; Anttila V; Neale BM; Jarvelin M-R; Fauser B; Kowalska I; Visser JA; Anderson M; Ong K; Stener-Victorin E; Ehrmann D; Legro RS; Salumets A; McCarthy MI; Morin-Papunen L; Thorsteinsdottir U; Stefansson K; Team AR; Styrkarsdottir U; Perry J; Dunaif A; Laven J; Franks S; Lindgren CM; Welt CK
- Article: SCIENTIFIC REPORTS. 2018;8(1):1878Kokosar M; Benrick A; Perfilyev A; Nilsson E; Kallman T; Ohlsson C; Ling C; Stener-Victorin E
- Book chapter: INFERTILITY IN WOMEN WITH POLYCYSTIC OVARY SYNDROME. 2018;p. 227-245Stener-Victorin E; Benrick A; Fornes R; Maliqueo M
- Article: CLINICAL ENDOCRINOLOGY. 2017;87(6):673-679Lingaiah S; Morin-Papunen L; Piltonen T; Puurunen J; Sundstrom-Poromaa I; Stener-Victorin E; Bloigu R; Risteli J; Tapanainen JS
- Journal article: JOURNAL OF GENETICS AND GENOMICS. 2017;44(10):475-481Li G; Yu Y; Fan Y; Li C; Xu X; Duan J; Li R; Kang X; Ma X; Chen X; Ke Y; Yan J; Lian Y; Liu P; Zhao Y; Zhao H; Chen Y; Sun X; Liu J; Qiao J; Liu J
- Letter: JAMA-JOURNAL OF THE AMERICAN MEDICAL ASSOCIATION. 2017;318(15):1502Wu X-K; Stener-Victorin E; Zhang H
- Review: NUTRIENTS. 2017;9(9):E996-996Kataoka J; Tassone EC; Misso M; Joham AE; Stener-Victorin E; Teede H; Moran LJ
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2017;114(34):E7187-E7196Benrick A; Chanclon B; Micallef P; Wu Y; Hadi L; Shelton JM; Stener-Victorin E; Asterholm IW
- Article: SCIENTIFIC REPORTS. 2017;7(1):8066Fornes R; Maliqueo M; Hu M; Hadi L; Jimenez-Andrade JM; Ebefors K; Nystrom J; Labrie F; Jansson T; Benrick A; Stener-Victorin E
- Article: FASEB JOURNAL. 2017;31(8):3288-3297Benrick A; Kokosar M; Hu M; Larsson M; Maliqueo M; Marcondes RR; Soligo M; Protto V; Jerlhag E; Sazonova A; Behre CJ; Hojlund K; Thoren P; Stener-Victorin E
- Conference publication: HUMAN REPRODUCTION. 2017;32:454-455Deng Y; Xie L; Wu XK; Ng EH; Victorin ES; Zhang H; Legro RS; Mol BW
- Review: SEMINARS IN REPRODUCTIVE MEDICINE. 2017;35(4):353-358Stener-Victorin E
- Article: JAMA-JOURNAL OF THE AMERICAN MEDICAL ASSOCIATION. 2017;317(24):2502-2514Wu X-K; Stener-Victorin E; Kuang H-Y; Ma H-L; Gao J-S; Xie L-Z; Hou L-H; Hu Z-X; Shao X-G; Ge J; Zhang J-F; Xue H-Y; Xu X-F; Liang R-N; Ma H-X; Yang H-W; Li W-L; Huang D-M; Sun Y; Hao C-F; Du S-M; Yang Z-W; Wang X; Yan Y; Chen X-H; Fu P; Ding C-F; Gao Y-Q; Zhou Z-M; Wang CC; Wu T-X; Liu J-P; Ng EHY; Legro RS; Zhang H
- Journal article: BMJ OPEN. 2017;7(6):e010955corr1Li J; Ng EHY; Stener-Victorin E
- Article: MOLECULAR AND CELLULAR ENDOCRINOLOGY. 2017;448(C):66-76Marcondes RR; Maliqueo M; Fornes R; Benrick A; Hu M; Ivarsson N; Carlstrom M; Cushman SW; Stenkula KG; Maciel GAR; Stener-Victorin E
- Corrigendum: HUMAN REPRODUCTION. 2017;32(4):966Poromaa IS; Mellembakken JR; Morin-Papunen L; Piltonen T; Puurenen J; Tapanainen J; Stener-Victorin E; Hirschberg AL; Vanky E; Ravn P; Glintborg D; Andersen M
- Article: TRIALS. 2017;18(1):115Li J; Ng EHY; Stener-Victorin E; Hu Z; Shao X; Wang H; Li M; Lai M; Xie C; Su N; Yu C; Liu J; Wu T; Ma H
- Article: FERTILITY AND STERILITY. 2017;107(3):788-795.e2Pinola P; Puukka K; Piltonen TT; Puurunen J; Vanky E; Sundstrom-Poromaa I; Stener-Victorin E; Hirschberg AL; Ravn P; Andersen MS; Glintborg D; Mellembakken JR; Ruokonen A; Tapanainen JS; Morin-Papunen LC
- Article: EXPERIMENTAL PHYSIOLOGY. 2017;102(1):113-127Maliqueo M; Benrick A; Marcondes RR; Johansson J; Sun M; Stener-Victorin E
- Article: OBESITY SCIENCE & PRACTICE. 2016;2(4):426-435Stener-Victorin E; Maliqueo M; Soligo M; Protto V; Manni L; Jerlhag E; Kokosar M; Sazonova A; Behre CJ; Lind M; Ohlsson C; Hojlund K; Benrick A
- Article: HUMAN REPRODUCTION. 2016;31(12):2791-2795Poromaa IS; Mellembakken JR; Papunen LM; Piltonen T; Puurunen J; Tapanainen JS; Stener-Victorin E; Hirschberg AL; Vanky E; Ravn P; Glintborg D; Andersen M
- Article: HUMAN REPRODUCTION UPDATE. 2016;22(6):687-708Balen AH; Morley LC; Misso M; Franks S; Legro RS; Wijeyaratne CN; Stener-Victorin E; Fauser BCJM; Norman RJ; Teede H
- Article: BMJ OPEN. 2016;6(10):e010955Li J; Ng EHY; Stener-Victorin E; Hu Z; Wu W; Lai M; Wu T; Ma H
- Article: MOLECULAR AND CELLULAR ENDOCRINOLOGY. 2016;433(C):1-11Fornes R; Hu M; Maliqueo M; Kokosar M; Benrick A; Carr D; Billig H; Jansson T; Manni L; Stener-Victorin E
- Conference publication: FERTILITY AND STERILITY. 2016;106(3):E364-E365Chan JL; Kar S; Vanky E; Stener-Victorin E; Morin-Papunen L; Maciel G; Poromaa IS; Mellembakken J; Dokras A
- Conference publication: FERTILITY AND STERILITY. 2016;106(3):E375Wu X; Stener-Victorin E; Liu J; Wu T; Ng E; Legro RS; Zhang H
- Article: FERTILITY AND STERILITY. 2016;106(3):757-765.e1Wu X-K; Wang Y-Y; Liu J-P; Liang R-N; Xue H-Y; Ma H-X; Shao X-G; Ng EHY
- Article: PSYCHONEUROENDOCRINOLOGY. 2016;69:142-149Hellgren C; Edvinsson A; Olivier JD; Fornes R; Stener-Victorin E; Ubhayasekera SJKA; Skalkidou A; Bergquist J; Sundstrom-Poromaa I
- Conference publication: DIABETES. 2016;65:A444Garcia BC; Benrick A; Wu Y; Hadi L; Franks S; Stener-Victorin E; Asterholm IW
- Journal article: SCIENTIFIC REPORTS. 2016;6:25321Kokosar M; Benrick A; Perfilyev A; Fornes R; Nilsson E; Maliqueo M; Behre CJ; Sazonova A; Ohlsson C; Ling C; Stener-Victorin E
- Article: SCIENTIFIC REPORTS. 2016;6:22883Kokosar M; Benrick A; Perfilyev A; Fornes R; Nilsson E; Maliqueo M; Behre CJ; Sazonova A; Ohlsson C; Ling C; Stener-Victorin E
- Journal article: NATURE COMMUNICATIONS. 2016;7:10762Hayes MG; Urbanek M; Ehrmann DA; Armstrong LL; Lee JY; Sisk R; Karaderi T; Barber TM; McCarthy MI; Franks S; Lindgren CM; Welt CK; Diamanti-Kandarakis E; Panidis D; Goodarzi MO; Azziz R; Zhang Y; James RG; Olivier M; Kissebah AH; Stener-Victorin E; Legro RS; Dunaif A
- Article: CLINICAL NUTRITION. 2016;35(1):213-218Larsson I; Hulthen L; Landen M; Palsson E; Janson P; Stener-Victorin E
- Article: JOURNAL OF ALTERNATIVE AND COMPLEMENTARY MEDICINE. 2016;22(2):101-107MacPherson H; Hammerschlag R; Coeytaux RR; Davis RT; Harris RE; Kong J-T; Langevin HM; Lao L; Milley RJ; Napadow V; Schnyer RN; Stener-Victorin E; Witt CM; Wayne PM
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2015;112(46):14348-14353Hu M; Richard JE; Maliqueo M; Kokosar M; Fornes R; Benrick A; Jansson T; Ohlsson C; Wu X; Skibicka KP; Stener-Victorin E
- Review: ENDOCRINE REVIEWS. 2015;36(5):487-525Dumesic DA; Oberfield SE; Stener-Victorin E; Marshall JC; Laven JS; Legro RS
- Conference publication: LANCET. 2015;386:70Wu X-K; Wang Y-Y; Liu J-P; Hou L-H; Gao Y-Q; Du S-M; Yan Y; Zhang J-F; Xue H-Y; Li W-L; Liang R-N; Tan Z-Y; Fu J-Y; Ma H-X; Shao X-G; He F-J; An M-E; Li P-L; Ding C-F; Li X-B; Shen X-J; Yu G-Z; Tian F; Xie Y-M; Silver RM; Stener-Victorin E; Ernest HN
- Article: MOLECULAR AND CELLULAR ENDOCRINOLOGY. 2015;412(C):159-169Maliqueo M; Benrick A; Alvi A; Johansson J; Sun M; Labrie F; Ohlsson C; Stener-Victorin E
- Article: BIOLOGY OF REPRODUCTION. 2015;93(3):69-12Kauffman AS; Thackray VG; Ryan GE; Tolson KP; Glidewell-Kenney CA; Semaan SJ; Poling MC; Iwata N; Breen KM; Duleba AJ; Stener-Victorin E; Shimasaki S; Webster NJ; Mellon PL
- Article: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 2015;100(9):3400-3407Pinola P; Piltonen TT; Puurunen J; Vanky E; Sundstrom-Poromaa I; Stener-Victorin E; Ruokonen A; Puukka K; Tapanainen JS; Morin-Papunen LC
- Article: NATURE COMMUNICATIONS. 2015;6:7502Hayes MG; Urbanek M; Ehrmann DA; Armstrong LL; Lee JY; Sisk R; Karaderi T; Barber TM; McCarthy MI; Franks S; Lindgren CM; Welt CK; Diamanti-Kandarakis E; Panidis D; Goodarzi MO; Azziz R; Zhang Y; James RG; Olivier M; Kissebah AH; Stener-Victorin E; Legro RS; Dunaif A
- Journal article: EUROPEAN JOURNAL OF INTEGRATIVE MEDICINE. 2015;7(4):432-433Stener-Victorin E
- Article: HUMAN MOLECULAR GENETICS. 2015;24(13):3792-3813Ronn T; Volkov P; Gillberg L; Kokosar M; Perfilyev A; Jacobsen AL; Jorgensen SW; Brons C; Jansson P-A; Eriksson K-F; Pedersen O; Hansen T; Groop L; Stener-Victorin E; Vaag A; Nilsson E; Ling C
- Article: ENDOCRINOLOGY. 2015;156(7):2492-2502Nilsson ME; Vandenput L; Tivesten A; Norlen A-K; Lagerquist MK; Windahl SH; Borjesson AE; Farman HH; Poutanen M; Benrick A; Maliqueo M; Stener-Victorin E; Ryberg H; Ohlsson C
- Article: BMJ OPEN. 2015;5(4):e007757Zheng Y; Stener-Victorin E; Ng EHY; Li J; Wu X; Ma H
- Article: JOURNAL OF ALTERNATIVE AND COMPLEMENTARY MEDICINE. 2015;21(3):113-128Langevin HM; Schnyer R; MacPherson H; Davis R; Harris RE; Napadow V; Wayne PM; Milley RJ; Lao L; Stener-Victorin E; Kong J-T; Hammerschlag R
- Article: HUMAN REPRODUCTION. 2015;30(3):692-700Maliqueo M; Poromaa IS; Vanky E; Fornes R; Benrick A; Akerud H; Stridsklev S; Labrie F; Jansson T; Stener-Victorin E
- Article: ACTA OBSTETRICIA ET GYNECOLOGICA SCANDINAVICA. 2015;94(3):279-287Leonhardt H; Hellstrom M; Gull B; Lind A-K; Nilsson L; Janson PO; Stener-Victorin E
- Corrigendum: ACTA DIABETOLOGICA. 2015;52(1):219-220Benrick A; Maliqueo M; Johansson J; Sun M; Wu X; Manneras-Holm L; Stener-Victorin E
- Article: ACTA DIABETOLOGICA. 2014;51(6):963-972Benrick A; Maliqueo M; Johansson J; Sun M; Wu X; Manneras-Holm L; Stener-Victorin E
- Conference publication: FERTILITY AND STERILITY. 2014;102(3):E29Stener-Victorin E; Benrick A; Kokosar M; Maliqueo M; Behre C; Hojlund K; Sazonova A
- Review: CHINESE MEDICAL JOURNAL. 2014;127(18):3309-3315Ma H; Liang H; Gao J; Ma H; Liu J; Ng HE; Billhult A; Stener-Victorin E; Mu X; Wu X
- Article: ADIPOCYTE. 2014;3(3):190-196Manneras-Holm L; Benrick A; Stener-Victorin E
- Article: BMC COMPLEMENTARY AND ALTERNATIVE MEDICINE. 2014;14:187Vixner L; Schytt E; Stener-Victorin E; Waldenstrom U; Pettersson H; Martensson LB
- Conference publication: ENDOCRINE REVIEWS. 2014;35(3)Maliqueo M; Poromaa IS; Vanky E; Akerud H; Stridsklev S; Labrie F; Jansson T; Stener-Victorin E
- Article: FERTILITY AND STERILITY. 2014;101(6):1747-56.e1-3-U334Leonhardt H; Hellstrom M; Gull B; Lind A-K; Nilsson L; Janson PO; Stener-Victorin E
- Article: SEMINARS IN REPRODUCTIVE MEDICINE. 2014;32(3):183-193Maliqueo M; Benrick A; Stener-Victorin E
- Journal article: ENDOCRINE ABSTRACTS. 2014Stener-Victorin E
- Article: ACTA RADIOLOGICA. 2014;55(2):248-256Leonhardt H; Gull B; Stener-Victorin E; Hellstrom M
- Editorial: EVIDENCE-BASED COMPLEMENTARY AND ALTERNATIVE MEDICINE. 2014;2014:698921Wu X; Ng EHY; Stener-Victorin E; Legro RS
- Article: BMJ OPEN. 2013;3(11):e003934Li Y; Kuang H; Shen W; Ma H; Zhang Y; Stener-Victorin E; Ng EHY; Liu J; Kuang H; Hou L; Wu X
- Article: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 2013;98(8):3115-3120Riedl S; Springer A; Haeusler G; Price G; Richter-Unruh A; Stener-Victorin E; Wudy SA
- Article: MOLECULAR AND CELLULAR ENDOCRINOLOGY. 2013;373(1-2):83-90Stener-Victorin E
- Article: BMC COMPLEMENTARY AND ALTERNATIVE MEDICINE. 2013;13:131Stener-Victorin E; Holm G; Janson PO; Gustafson D; Waern M
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2013;304(9):E934-E943Johansson J; Redman L; Veldhuis PP; Sazonova A; Labrie F; Holm G; Johannsson G; Stener-Victorin E
- Article: FERTILITY AND STERILITY. 2013;99(3):889-896Ortega I; Sokalska A; Villanueva JA; Cress AB; Wong DH; Stener-Victorin E; Stanley SD; Duleba AJ
- Article: PLOS ONE. 2013;8(1):e54357Johansson J; Manneras-Holm L; Shao R; Olsson A; Lonn M; Billig H; Stener-Victorin E
- Article: ADVANCES IN PHARMACOLOGICAL SCIENCES. 2013;2013:808914-7Mansor F; Gu HF; Ostenson C-G; Manneras-Holm L; Stener-Victorin E; Mohamud WNW
- Review: EVIDENCE-BASED COMPLEMENTARY AND ALTERNATIVE MEDICINE. 2013;2013:762615-16Johansson J; Stener-Victorin E
- Article: EVIDENCE-BASED COMPLEMENTARY AND ALTERNATIVE MEDICINE. 2013;2013:527303-11Kuang H; Li Y; Wu X; Hou L; Wu T; Liu J; Ng EHY; Stener-Victorin E; Legro RS; Zhang H
- Article: EVIDENCE-BASED COMPLEMENTARY AND ALTERNATIVE MEDICINE. 2013;2013:964070-13Benrick A; Maliqueo M; Miao S; Villanueva JA; Feng Y; Ohlsson C; Duleba AJ; Stener-Victorin E
- Book chapter: CURRENT RESEARCH IN ACUPUNCTURE. 2013;p. 487-515Feng Y; Stener-Victorin E; Chen B
- Article: ENDOCRINOLOGY. 2013;154(1):434-445Maliqueo M; Sun M; Johansson J; Benrick A; Labrie F; Svensson H; Lonn M; Duleba AJ; Stener-Victorin E
- Journal article: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2012;303(12):E1506Raja-Khan N; Stener-Victorin E; Wu X; Legro RS
- Article: ACTA RADIOLOGICA. 2012;53(10):1195-1201Leonhardt H; Gull B; Kishimoto K; Kataoka M; Nilsson L; Janson PO; Stener-Victorin E; Hellstrom M
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2012;303(11):E1373-E1385Sun M; Maliqueo M; Benrick A; Johansson J; Shao R; Hou L; Jansson T; Wu X; Stener-Victorin E
- Article: FERTILITY AND STERILITY. 2012;98(5):1175-1185Shao R; Wang X; Wang W; Stener-Victorin E; Mallard C; Brannstrom M; Billig H
- Article: BLOOD PRESSURE. 2012;21(5):306-310Silverdal J; Mourtzinis G; Stener-Victorin E; Mannheimer C; Manhem K
- Journal article: BIOLOGY OF REPRODUCTION. 2012;87(Suppl_1):87Stener-Victorin E
- Journal article: FOCUS ON ALTERNATIVE AND COMPLEMENTARY THERAPIES. 2012;17(2):124-125Stener‐Victorin E
- Article: JOURNAL OF NEUROINFLAMMATION. 2012;9:111Halleskog C; Dijksterhuis JP; Kilander MBC; Becerril-Ortega J; Villaescusa JC; Lindgren E; Arenas E; Schulte G
- Letter: FERTILITY AND STERILITY. 2012;97(5):E21Stener-Victorin E
- Article: EXPERIMENTAL PHYSIOLOGY. 2012;97(5):651-662Feng Y; Johansson J; Shao R; Manneras-Holm L; Billig H; Stener-Victorin E
- Article: BMC COMPLEMENTARY AND ALTERNATIVE MEDICINE. 2012;12:42Bjersa K; Victorin ES; Olsen MF
- Article: BMC COMPLEMENTARY AND ALTERNATIVE MEDICINE. 2012;12:32Billhult A; Stener-Victorin E
- Review: BIOLOGY OF REPRODUCTION. 2012;86(4):131-7Shao R; Zou S; Wang X; Feng Y; Brannstrom M; Stener-Victorin E; Billig H
- Journal article: FOCUS ON ALTERNATIVE AND COMPLEMENTARY THERAPIES. 2012;17(1):43-44Stener‐Victorin E
- Article: FERTILITY AND STERILITY. 2012;97(2):501-508Stener-Victorin E; Baghaei F; Holm G; Janson PO; Olivecrona G; Lonn M; Manneras-Holm L
- Article: EVIDENCE-BASED COMPLEMENTARY AND ALTERNATIVE MEDICINE. 2012;2012:943198-9Vixner L; Martensson LB; Stener-Victorin E; Schytt E
- Article: PSYCHONEUROENDOCRINOLOGY. 2011;36(10):1470-1479Jedel E; Gustafson D; Waern M; Sverrisdottir YB; Landen M; Janson PO; Labrie F; Ohlsson C; Stener-Victorin E
- Conference publication: FERTILITY AND STERILITY. 2011;96(3):S114Ortega I; Stener-Victorin E; Villanueva JA; Sokalska A; Stanley SD; Duleba AJ
- Journal article: OBSTETRIC ANESTHESIA DIGEST. 2011;31(3):190-191Hjelmstedt A; Shenoy ST; Stener-Victorin E; Lekander M; Bhat M; Balakumaran L; Waldenström U
- Journal article: FOCUS ON ALTERNATIVE AND COMPLEMENTARY THERAPIES. 2011;16(3):214-215Stener‐Victorin E
- Article: HORMONES AND BEHAVIOR. 2011;60(4):427-438Feng Y; Shao R; Weijdegard B; Wang T; Johansson J; Sun S; Wang W; Egecioglu E; Billig H; Stener-Victorin E
- Article: FERTILITY AND STERILITY. 2011;96(2):452-458Li Y; Li Y; Ng EHY; Stener-Victorin E; Hou L; Wu T; Han F; Wu X
- Conference publication: HUMAN REPRODUCTION. 2011;26:I332Leonhardt H; Gull B; Kishimoto K; Kataoka M; Stener-Victorin E; Hellstrom M
- Review: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2011;301(1):E1-E10Raja-Khan N; Stener-Victorin E; Wu X; Legro RS
- Journal article: JOURNAL OF HYPERTENSION. 2011;29:e49Silverdal J; Mourtzinis G; StenerVictorin E; Mannheimer C; Manhem K
- Article: CLINICAL JOURNAL OF PAIN. 2011;27(5):448-456Soderberg EI; Carlsson JY; Stener-Victorin E; Dahlof C
- Article: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 2011;96(5):E869-E877Shao R; Norstrom A; Weijdegard B; Egecioglu E; Fernandez-Rodriguez J; Feng Y; Stener-Victorin E; Brannstrom M; Billig H
- Review: CLINICAL ENDOCRINOLOGY. 2011;74(4):424-433Pasquali R; Stener-Victorin E; Yildiz BO; Duleba AJ; Hoeger K; Mason H; Homburg R; Hickey T; Franks S; Tapanainen JS; Balen A; Abbott DH; Diamanti-Kandarakis E; Legro RS
- Article: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 2011;96(4):1068-1076Manneras-Holm L; Baghaei F; Holm G; Janson PO; Ohlsson C; Lonn M; Stener-Victorin E
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-REGULATORY, INTEGRATIVE AND COMPARATIVE PHYSIOLOGY. 2011;300(4):R869-R875Yang X; Zhang Y; Wu X; Bae CS; Hou L; Kuang H; Wang Y; Stener-Victorin E
- Editorial: EXPLORE-THE JOURNAL OF SCIENCE AND HEALING. 2011;7(2):120-123Stener-Victorin E; Manheimer E
- Journal article: FOCUS ON ALTERNATIVE AND COMPLEMENTARY THERAPIES. 2011;16(1):36-37Stener‐Victorin E; Choi I
- Article: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 2011;96(2):E304-E311Manneras-Holm L; Leonhardt H; Kullberg J; Jennische E; Oden A; Holm G; Hellstrom M; Lonn L; Olivecrona G; Stener-Victorin E; Lonn M
- Article: MIDWIFERY. 2011;27(1):e134-e139Bergh IHE; Stener-Victorin E; Wallin G; Martensson L
- Journal article: ENDOCRINOLOGY. 2011;152(1):332Mannerås-Holm L; Leonhardt H; Kullberg J; Jennische E; Odén A; Holm G; Hellström M; Lönn L; Olivecrona G; Stener-Victorin E; Lönn M
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2011;300(1):E37-E45Jedel E; Labrie F; Oden A; Holm G; Nilsson L; Janson PO; Lind A-K; Ohlsson C; Stener-Victorin E
- Journal article: ACTA OBSTETRICIA ET GYNECOLOGICA SCANDINAVICA. 2010;89(12):1622Jedel E; Kowalski J; Stener-Victorin E
- Journal article: FOCUS ON ALTERNATIVE AND COMPLEMENTARY THERAPIES. 2010;15(4):291-292Stener‐Victorin E
- Article: ACTA OBSTETRICIA ET GYNECOLOGICA SCANDINAVICA. 2010;89(11):1453-1459Hjelmstedt A; Shenoy ST; Stener-Victorin E; Lekander M; Bhat M; Balakumaran L; Waldenstrom U
- Article: AUTONOMIC NEUROSCIENCE-BASIC & CLINICAL. 2010;157(1-2):46-51Stener-Victorin E; Wu X
- Conference publication: JOURNAL OF GASTROENTEROLOGY AND HEPATOLOGY. 2010;25:A79Walsh MD; Rosty C; Buchanan DD; Walters RJ; Roberts A; Clendenning M; McKeone DM; Walker NI; Parry S; Young JP
- Book chapter: CURRENT MANAGEMENT OF POLYCYSTIC OVARY SYNDROME. 2010;p. 195-212Stener-Victorin E
- Article: JOURNAL OF STEROID BIOCHEMISTRY AND MOLECULAR BIOLOGY. 2010;122(1-3):82-90Alexanderson C; Stener-Victorin E; Kullberg J; Nilsson S; Levin M; Cajander S; Lonn L; Lonn M; Holmang A
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2010;299(4):E551-E559Johansson J; Feng Y; Shao R; Lonn M; Billig H; Stener-Victorin E
- Journal article: FOCUS ON ALTERNATIVE AND COMPLEMENTARY THERAPIES. 2010;15(3):226-228Stener‐Victorin E; Komesaroff P; O'Brien K; Varigos E
- Article: AMERICAN JOURNAL OF OBSTETRICS AND GYNECOLOGY. 2010;203(1):65.e1-65.e10Nutu M; Feng Y; Egecioglu E; Weijdegard B; Stener-Victorin E; Shao R
- Article: MOLECULAR AND CELLULAR ENDOCRINOLOGY. 2010;321(2):161-174Feng Y; Weijdegard B; Wang T; Egecioglu E; Fernandez-Rodriguez J; Huhtaniemi I; Stener-Victorin E; Billig H; Shao R
- Journal article: PLOS MEDICINE. 2010;7(6):e1000261MacPherson H; Altman DG; Hammerschlag R; Li Y; Wu T; White A; Moher D
- Editorial: OBSTETRICAL & GYNECOLOGICAL SURVEY. 2010;65(6):383-385Stener-Victorin E; Holm G; Labrie F; Nilsson L; Janson PO; Ohlsson C
- Conference publication: ENDOCRINE REVIEWS. 2010;31(3)Jedel E; Holm G; Labrie F; Oden A; Nilsson L; Janson PO; Ohlsson C; Stener-Victorin E
- Article: EUROPEAN JOURNAL OF OBSTETRICS & GYNECOLOGY AND REPRODUCTIVE BIOLOGY. 2010;150(1):66-71Gejervall A-L; Lundin K; Stener-Victorin E; Bergh C
- Article: EUROPEAN JOURNAL OF PAEDIATRIC NEUROLOGY. 2010;14(3):239-246Svedberg LE; Englund E; Malker H; Stener-Victorin E
- Conference publication: ENDOCRINE JOURNAL. 2010;57:S431Johansson JA; Feng Y; Shao R; Billig H; Lonn M; Stener-Victorin E
- Conference publication: ENDOCRINE JOURNAL. 2010;57:S345Feng Y; Shao R; Weijdegard B; Wang T; Johansson J; Egecioglu E; Sun S; Billig H; Stener-Victorin E
- Article: JOURNAL OF ETHNOPHARMACOLOGY. 2010;127(2):346-351Manneras L; Fazliana M; Nazaimoon WMW; Lonn M; Gu HF; Ostenson CG; Stener-Victorin E
- Journal article: ENDOCRINE REVIEWS. 2010;31(1):134Stener-Victorin E; Holm G; Labrie F; Nilsson L; Janson PO; Ohlsson C
- Article: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 2010;95(2):810-819Stener-Victorin E; Holm G; Labrie F; Nilsson L; Janson PO; Ohlsson C
- Article: HUMAN REPRODUCTION. 2010;25(2):450-456Jedel E; Waern M; Gustafson D; Landen M; Eriksson E; Holm G; Nilsson L; Lind A-K; Janson PO; Stener-Victorin E
- Journal article: ENDOCRINOLOGY. 2010;151(1):442Stener-Victorin E; Holm G; Labrie F; Nilsson L; Janson PO; Ohlsson C
- Article: AUTONOMIC NEUROSCIENCE-BASIC & CLINICAL. 2009;150(1-2):111-115Billhult A; Lindholm C; Gunnarsson R; Stener-Victorin E
- Conference publication: EUROPEAN JOURNAL OF NEUROLOGY. 2009;16:53Sverrisdottir Y; Jedel E; Janson PO; Stener-Victorin E
- Conference publication: DIABETOLOGIA. 2009;52:S203Fazliana M; Manneras L; Nazaimoon WW; Lonn M; Gu HF; Ostenson C-G; Stener-Victorin E
- Article: PLOS ONE. 2009;4(8):e6638Feng Y; Johansson J; Shao R; Manneras L; Fernandez-Rodriguez J; Billig H; Stener-Victorin E
- Journal article: AUTONOMIC NEUROSCIENCE-BASIC & CLINICAL. 2009;149(1-2):62Stener-Victorin E; Jedel E; Janson PO; Sverrisdottir YB
- Journal article: AUTONOMIC NEUROSCIENCE-BASIC & CLINICAL. 2009;149(1-2):62P1.7 Is polycystic ovary syndrome associated with high sympathetic nerve activity and size at birth?Sverrisdottir YB; Mogren T; Kataoka J; Janson PO; Stener-Victorin E
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-REGULATORY, INTEGRATIVE AND COMPARATIVE PHYSIOLOGY. 2009;297(2):R387-R395Stener-Victorin E; Jedel E; Janson PO; Sverrisdottir YB
- Article: ACTA PHYSIOLOGICA. 2009;196(3):303-314Lindqvist H; Sandberg A-S; Undeland I; Stener-Victorin E; Larsson BM; Sannaveerappa T; Lonn M; Holmang A
- Conference publication: JOURNAL OF HYPERTENSION. 2009;27:S439Sverrisdottir YB; Jedel E; Janson PO; Stener-Victorin E
- Article: JOURNAL OF ENDOCRINOLOGY. 2009;201(1):49-58Alexanderson C; Eriksson E; Stener-Victorin E; Lonn M; Holmang A
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-REGULATORY, INTEGRATIVE AND COMPARATIVE PHYSIOLOGY. 2009;296(4):R1124-R1131Manneras L; Cajander S; Loenn M; Stener-Victorin E
- Journal article: FOCUS ON ALTERNATIVE AND COMPLEMENTARY THERAPIES. 2009;14(1):8-11Stener‐Victorin E
- Article: BIOLOGY OF REPRODUCTION. 2009;80(2):264-271Dorfman M; Ramirez VD; Stener-Victorin E; Lara HE
- Article: JOURNAL OF APPLIED PHYSIOLOGY. 2009;106(2):631-639Yang J-N; Bjorklund O; Lindstrom-Tornqvist K; Lindgren E; Eriksson TM; Kahlstrom J; Chen J-F; Schwarzschild MA; Tobler I; Fredholm BB
- Article: BJOG-AN INTERNATIONAL JOURNAL OF OBSTETRICS AND GYNAECOLOGY. 2008;115(13):1655-1668Elden H; Fagevik-Olsen M; Ostgaard H-C; Stener-Victorin E; Hagberg H
- Article: EUROPEAN JOURNAL OF PHARMACOLOGY. 2008;597(1-3):92-101JohanssonA SM; Lindgren E; Yang J-N; Herling AW; Fredholm BB
- Journal article: FERTILITY AND STERILITY. 2008;90:s133Mannerås L; Lönn M; Stener-Victorin E
- Journal article: OBSTETRIC ANESTHESIA DIGEST. 2008;28(3):175Mårtensson L; Stener-Victorin E; Wallin G
- Published conference paper: JOURNAL OF ALTERNATIVE AND COMPLEMENTARY MEDICINE. 2008;14(7):861-869Napadow V; Ahn A; Longhurst J; Lao L; Stener-Victorin E; Harris R; Langevin HM
- Editorial: FORSCHENDE KOMPLEMENTARMEDIZIN. 2008;15(4):228-229Stener-Victorin E
- Conference publication: EUROPEAN JOURNAL OF NEUROLOGY. 2008;15:238Sverrisdottir YB; Mogren T; Kataoka J; Janson PO; Stener-Victorin E
- Article: ENDOCRINOLOGY. 2008;149(7):3559-3568Manneras L; Jonsdottir IH; Holmang A; Lonn M; Stener-Victorin E
- Review: ACUPUNCTURE IN MEDICINE. 2008;26(2):111-120White A; Cummings M; Barlas P; Cardini F; Filshie J; Foster NE; Lundeberg T; Stener-Victorin E; Witt C
- Conference publication: JOURNAL OF HYPERTENSION. 2008;26:S16-S17Sverrisdottir YB; Mogren T; Kataoka J; Janson PO; Stener-Victorin E
- Article: AUTONOMIC NEUROSCIENCE-BASIC & CLINICAL. 2008;140(1-2):88-95Billhult A; Lindholm C; Gunnarsson R; Stener-Victorin E
- Journal article: GYNAKOLOGISCHE ENDOKRINOLOGIE. 2008;6(2):67-71Stener-Victorin E; von Hagens C
- Review: JOURNAL OF NEUROENDOCRINOLOGY. 2008;20(3):290-298Stener-Victorin E; Jedel E; Manneras L
- Article: JOURNAL OF REHABILITATION MEDICINE. 2008;40(3):171-177Kaell LB; Kowalski J; Stener-Victorin E
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2008;294(3):E576-E581Sverrisdottir YB; Mogren T; Kataoka J; Janson PO; Stener-Victorin E
- Article: EUROPEAN JOURNAL OF PAEDIATRIC NEUROLOGY. 2008;12(2):89-96Suedberg LE; Englund E; Malker H; Stener-Victorin E
- Article: ACTA OBSTETRICIA ET GYNECOLOGICA SCANDINAVICA. 2008;87(12):1329-1335Jedel E; Kowalski J; Stener-Victorin E
- Article: ACTA OBSTETRICIA ET GYNECOLOGICA SCANDINAVICA. 2008;87(2):171-177Martensson L; Stener-Victorin E; Wallin G
- Article: ENDOCRINOLOGY. 2007;148(11):5369-5376Alexanderson C; Eriksson E; Stener-Victorin E; Lystig T; Gabrielsson B; Loenn M; Holmaeng A
- Article: ENDOCRINOLOGY. 2007;148(8):3781-3791Manneras L; Cajander S; Holmang A; Seleskovic Z; Lystig T; Lohn M; Stener-Victorin E
- Article: EUROPEAN JOURNAL OF PAIN. 2007;11(5):557-563Jedel E; Carlsson J; Stener-Victorin E
- Article: CLINICAL NURSING RESEARCH. 2007;16(2):85-99Billhult A; Stener-Victorin E; Bergbom I
- Published conference paper: ACTA PHYSIOLOGICA. 2007;190(1):87-96Johansson SM; Yang J-N; Lindgren E; Fredholm BB
- Article: REPRODUCTIVE BIOMEDICINE ONLINE. 2007;14(2):184-190Gejervall A-L; Stener-Victorin E; Cerne A; Borg K; Bergh C
- Article: ACTA OBSTETRICIA ET GYNECOLOGICA SCANDINAVICA. 2007;86(3):310-314Olsen MF; Elden H; Janson ED; Lilja H; Stener-Victorin E
- Article: JOURNAL OF ALTERNATIVE AND COMPLEMENTARY MEDICINE. 2007;13(1):53-57Billhult A; Bergbom I; Stener-Victorin E
- Review: ACUPUNCTURE IN MEDICINE. 2006;24(4):157-163Stener-Victorin E; Humaidan P
- Article: HUMAN REPRODUCTION. 2006;21(11):2916-2921Cerne A; Bergh C; Borg K; Ek I; Gejervall A-L; Hillensjo T; Olofsson JI; Stener-Victorin E; Wood M; Westlander G
- Article: CEPHALALGIA. 2006;26(11):1320-1329Soderberg E; Carlsson J; Stener-Victorin E
- Article: JOURNAL OF APPLIED PHYSIOLOGY. 2006;101(1):84-91Stener-Victorin E; Fujisawa S; Kurosawa M
- Article: REPRODUCTIVE BIOMEDICINE ONLINE. 2006;13(1):120-125Humaidan P
- Article: DISABILITY AND REHABILITATION. 2006;28(5):271-279Bunketorp L; Lindh M; Carlsson J; Stener-Victorin E
- Article: CLINICAL REHABILITATION. 2006;20(3):201-217Bunketorp L; Lindh M; Carlsson J; Stener-Victorin E
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-REGULATORY, INTEGRATIVE AND COMPARATIVE PHYSIOLOGY. 2006;290(3):R826-R835Manni L; Holmäng A; Cajander S; Lundeberg T; Aloe L; Stener-Victorin E
- Article: JOURNAL OF NEUROENDOCRINOLOGY. 2005;17(12):846-858Manni L; Cajander S; Lundeberg T; Naylor AS; Aloe L; Holmäng A; Jonsdottir IH; Stener-Victorin E
- Article: REPRODUCTIVE BIOLOGY AND ENDOCRINOLOGY. 2005;3:44Stener-Victorin E; Ploj K; Larsson BM; Holmäng A
- Letter: PAIN. 2005;117(1-2):237-238Stener-Victorin E; Cummings M; Lundeberg T
- Article: JOURNAL OF REHABILITATION MEDICINE. 2005;37(5):330-334Bunketorp L; Carlsson J; Kowalski J; Stener-Victorin E
- Article: REPRODUCTIVE BIOLOGY AND ENDOCRINOLOGY. 2005;3:21Manni L; Lundeberg T; Holmäng A; Aloe L; Stener-Victorin E
- Article: AUTONOMIC NEUROSCIENCE-BASIC & CLINICAL. 2005;118(1-2):79-87Manni L; Holmäng A; Lundeberg T; Aloe L; Stener-Victorin E
- Article: HUMAN REPRODUCTION. 2005;20(3):728-735Gejervall AL; Stener-Victorin E; Möller A; Janson PO; Werner C; Bergh C
- Review: HUMAN REPRODUCTION. 2005;20(2):339-349Stener-Victorin E
- Article: EUROPEAN SPINE JOURNAL. 2005;14(1):84-89Bunketorp L; Stener-Victorin E; Carlsson J
- Article: EUROPEAN JOURNAL OF PAEDIATRIC NEUROLOGY. 2005;9(5):347-354Svedberg LE; Stener-Victorin E; Nordahl G; Lundeberg T
- Letter: HUMAN REPRODUCTION. 2004;19(12):2966-2967Stener-Victorin E; Humaidan P
- Journal article: HUMAN REPRODUCTION. 2004;19(12):2965-2966Renckens C
- Journal article: EVIDENCE-BASED OBSTETRICS AND GYNECOLOGY. 2004;6(3):131-132
- Article: HUMAN REPRODUCTION. 2004;19(6):1367-1372Humaidan P; Stener-Victorin E
- Article: CLINICAL JOURNAL OF PAIN. 2004;20(3):179-185Stener-Victorin E; Kruse-Smidje C; Jung K
- Article: REPRODUCTIVE BIOLOGY AND ENDOCRINOLOGY. 2004;2:16Stener-Victorin E; Kobayashi R; Watanabe O; Lundeberg T; Kurosawa M
- Article: BIOLOGY OF REPRODUCTION. 2004;70(2):329-333Stener-Victorin E; Lindholm C
- Article: JOURNAL OF INVESTIGATIVE DERMATOLOGY. 2003;121(6):1301-1305Stener-Victorin E; Lundeberg T; Kowalski J; Opdal L; Sjöström J; Lundeberg L
- Article: AUTONOMIC NEUROSCIENCE-BASIC & CLINICAL. 2003;108(1-2):50-56Stener-Victorin E; Kobayashi R; Kurosawa M
- Journal article: EVIDENCE-BASED OBSTETRICS AND GYNECOLOGY. 2003;5(3):109-110
- Article: HUMAN REPRODUCTION. 2003;18(7):1454-1460Stener-Victorin E; Waldenström U; Wikland M; Nilsson L; Hägglund L; Lundeberg T
- Article: REPRODUCTIVE BIOLOGY AND ENDOCRINOLOGY. 2003;1:33Stener-Victorin E; Lundeberg T; Cajander S; Aloe L; Manni L; Waldenström U; Janson PO
- Journal article: INTERNATIONAL CONGRESS SERIES. 2002;1238:3-10Lundeberg T; Stener-Victorin E
- Journal article: INTERNATIONAL CONGRESS SERIES. 2002;1238:149-156Stener-Victorin E
- Journal article: ACUPUNCTURE IN MEDICINE. 2002;20(2-3):111-127Stener-Victorin E
- Article: HUMAN REPRODUCTION. 2002;17(8):1942-1946Stener-Victorin E; Wikland M; Waldenström U; Lundeberg T
- Article: ANESTHESIA AND ANALGESIA. 2002;95(1):151-157Stener-Victorin E; Kowalski J; Lundeberg T
- Article: NEUROPEPTIDES. 2001;35(5-6):227-231Stener-Victorin E; Lundeberg T; Waldenström U; Bileviciute-Ljungar I; Janson PO
- Conference publication: HUMAN REPRODUCTION. 2001;16:207-208Stener-Victorin E; Lundeberg T; Waldenstroem U; Manni L; Aloe L; Bileviciute-Ljungar I; Gunnarsson S; Janson PO
- Article: BIOLOGY OF REPRODUCTION. 2000;63(5):1497-1503Stener-Victorin E; Lundeberg T; Waldenström U; Manni L; Aloe L; Gunnarsson S; Janson PO
- Article: ACTA OBSTETRICIA ET GYNECOLOGICA SCANDINAVICA. 2000;79(3):180-188Stener-Victorin E; Waldenström U; Tägnfors U; Lundeberg T; Lindstedt G; Janson PO
- Article: HUMAN REPRODUCTION. 1999;14(10):2480-2484Stener-Victorin E; Waldenström U; Nilsson L; Wikland M; Janson PO
- Journal article: ACUPUNCTURE IN MEDICINE. 1998;16(2):80-82Stener-Victorin E
- Conference publication: HUMAN REPRODUCTION. 1998;13:258Stener-Victorin E; Waldenström U; Tägnfors U; Janson PO
- Conference publication: HUMAN REPRODUCTION. 1998;13:181Stener-Victorin E; Waldenström E; Josefsson B; Nilsson L; Wikland M; Hammar M; Nilsson E; Törnqvist R
- Article: HUMAN REPRODUCTION. 1996;11(6):1314-1317StenerVictorin E; Waldenstrom U; Andersson SA; Wikland M
- Journal article: NAUNYN-SCHMIEDEBERGS ARCHIVES OF PHARMACOLOGY. 1988;337(5):477-483FREDHOLM BB; LINDGREN E
- Book chapter: NEUROTRANSMITTERS AND CORTICAL FUNCTION. 1988;p. 437-451Fredholm BB; Dunér-Engström M; Fastbom J; Jonzon B; Lindgren E; Nordstedt C
- Journal article: ACTA PHYSIOLOGICA. 1987;131(4):543-551FREDHOLM BB; LINDGREN E; LINDSTROM K; NORDSTEDT C
- Journal article: BASIC & CLINICAL PHARMACOLOGY & TOXICOLOGY. 1987;61(2):121-125HU PS; LINDGREN E; JACOBSON KA; FREDHOLM BB
- Journal article: ACTA PHYSIOLOGICA. 1987;130(1):95-105FREDHOLM BB; LINDGREN E
- Book chapter: TOPICS AND PERSPECTIVES IN ADENOSINE RESEARCH. 1987;p. 509-520Fredholm BB; Dunér-Engstrom M; Fastbom J; Jonzen B; Lindgren E; Nordstedt C; Pedata F; van der Ploeg I
- Book chapter: RECEPTOR-RECEPTOR INTERACTIONS: WENNER-GREN CENTER INTERNATIONAL SYMPOSIUM SERIES. 1987;p. 308-317Fredholm BB; Nordstedt C; van der Ploeg I; Lindgren E; Ng J; Jondal M
- Book chapter: RECEPTOR-RECEPTOR INTERACTIONS: WENNER-GREN SYMPOSIUM. 1987;p. 308-317Fredholm BB; Nordstedt C; van der Ploeg I; Lindgren E; Ng J; Jondal M
- Journal article: ACTA PHYSIOLOGICA. 1986;128(4):659-660FREDHOLM BB; HU PS; LINDGREN E
- Journal article: ACTA PHYSIOLOGICA. 1986;127(3):381-386FREDHOLM BB; FASTBOM J; LINDGREN E
- Journal article: ACTA PHYSIOLOGICA. 1986;126(2):307-309FREDHOLM BB; LINDGREN E
- Journal article: BRITISH JOURNAL OF PHARMACOLOGY. 1985;86(2):509-513FREDHOLM BB; LINDGREN E; LINDSTROM K
- Journal article: ACTA PHARMACOLOGICA ET TOXICOLOGICA. 1984;55(4):297-302ELFMAN L; LINDGREN E; WALUM E; FREDHOLM BB
- Journal article: ACTA PHYSIOLOGICA. 1984;122(1):55-59FREDHOLM BB; JONZON B; LINDGREN E
- Journal article: ACTA PHARMACOLOGICA ET TOXICOLOGICA. 1984;54(1):64-71FREDHOLM BB; LINDGREN E
- Journal article: BIOCHEMICAL PHARMACOLOGY. 1983;32(18):2832-2834FREDHOLM BB; LINDGREN E
- Journal article: ACTA PHARMACOLOGICA ET TOXICOLOGICA. 1983;53(3):236-244FREDHOLM BB; LINDGREN E; LINDSTROM K; VERNET L
- Journal article: ACTA PHYSIOLOGICA. 1983;:7-10FREDHOLM BB; JONZON B; LINDGREN E
- Journal article: JOURNAL OF NEUROCHEMISTRY. 1982;39(1):165-175FREDHOLM BB; JONZON B; LINDGREN E; LINDSTROM K
- Conference publication: ACTA PHYSIOLOGICA. 1982;:31FREDHOLM BB; LINDGREN E
- Conference publication: ACTA PHARMACOLOGICA ET TOXICOLOGICA. 1981;49:29LINDGREN E; FREDHOLM BB
- Show more
Full list of publications
Funding
Grants
- Swedish Research Council1 January 2023 - 31 December 2026Polycystic ovary syndrome (PCOS) is the leading cause of female infertility and linked to type-2 diabetes and endometrial cancer. Progress in managing the disorder is hindered by a lack of insight into the underlying mechanisms. We know that male hormones play a key role and that PCOS runs in families. Recently we made the discovery that prenatal androgen-induced PCOS-like traits in female and male mice are passed down across multiple generations. Thus, growing evidence suggests that genetic and epigenetic mechanisms act in concert in complex traits as PCOS.Yet fundamental questions remain:What are the cell-type-specific epigenetic and transcriptomic disease signatures and signaling pathways in PCOS endometrium and adipose tissue? Can such perturbations be reversed?What are the key triggers and molecular pathways that drive transmission of PCOS in the absence of PCOS-associated gene variants?Can germ cells drive transmission in the absence of direct intrauterine effects? If so, can PCOS be transmitted to future generations also by sons?We will integrate human and mouse studies with state-of-the-art molecular techniques to answer these questions. The fundamental novel insights that the proposed projects will yield into PCOS etiology and pathophysiology will have several important translational implications that could shape how PCOS, and associated comorbidities is managed. The long-term goal is to give targeted treatment and to predict response to treatment.
- Dissecting the molecular, mechanistic and causal pathways of Polycystic Ovary Syndrome and how these are passed on across generationsNovo Nordisk Foundation1 June 2022 - 31 May 2027Polycystic ovary syndrome (PCOS) is the leading cause of female infertility and linked to type-2 diabetes and cancer. Progress in managing the disorder is hindered by lack of insight into the underlying mechanisms. We know that male hormones plays a key role and that PCOS runs in families. Recently we made the discovery that PCOS-like symptoms, induced by exposing pregnant mice to male hormone, are passed down from mothers to great-granddaughters. Moreover, we have indications that sons can transmit the disease as well. _x000D_ _x000D_ I will take a multidisciplinary approach to get better insights into how PCOS is passed on in families. We will use human and mouse studies as well as state-of-the-art molecular techniques to dissect the key mechanisms that influence how the syndrome is passed on across generations both by women and men. My vision is to open new horizons for prevention strategies rather than managing symptoms, thereby markedly reducing the burden of the disease in both women and men._x000D_
- Transgenerational inheritance of disease – the role of germline transmission and impact of exerciseNovo Nordisk Foundation1 October 2019 - 30 September 2022
- Swedish Research Council1 January 2019 - 31 December 2022
- Transgenerational epigenetic germline inheritance – the impact of exerciseNovo Nordisk Foundation1 October 2018 - 30 September 2021
Staff and contact
Group leader
All members of the group
- Hanna DaryapeymaAffiliated to Research
- Anja DekanskiPhd Student
- Gustaw ErikssonPhd Student
I have a background in bioinformatics and am currently a PhD candidate in the Reproductive Endocrinology and Metabolism (REM) group, which I joined in the fall of 2021. My main focus is on uncovering
- Camille GauthierPostdoctoral Researcher
- Tina Gorsek SparovecPostdoctoral Researcher
- Mikaela Seoyeon HuhAffiliated to Research
- Congru LiResearch Specialist
- Eva LindgrenResearch Engineer
- Haojiang LuPostdoctoral Researcher
- Lena SchoblochResearch Assistant
- Luzi Sophie SchuchmannAffiliated to Research
- Dorothea TheurerAffiliated to Research
- Sara TorstenssonPhd Student
- Tanja TurunenPostdoctoral Researcher
- Tauska WangAffiliated to Research
Former group members
Angelo Ascani - PhD
Elizabet Jedel – PhD
Julia Johannson – PhD
Lisa Lindheim – PhD Candidate, Medical University of Graz, Austria
Louise Mannerås Holm – PhD
Luigi Manni – PhD
Min Hu – Postdoc
Milana Kokosar – PhD student
Maria Manti – PhD
Rodrigo Marcondes – PhD student
Romina Fornes – Postdoc
Sanjiv Risal - PhD
Yang Qian – PhD
Zhiyi Zhang, PhD, visiting researcher from Shandong Provincial Qianfoshan Hospital, Jinan, China
Group members outside Karolinska Institutet
Anna Benrick – MSc, PhD, Researcher, Gothenburg University, Sweden
Josefin Katakoa – MD, PhD, Gothenburg University, Sweden
Manuel Maliqueo – PhD, University of Chile, Santiago
Collaborations
National collaborators
- Associate Professor Qiaolin Deng, Karolinska Institutet
- Professor Angelica Lindén Hirschberg, Karolinska Institutet
- Professor Camilla Svensson, Karolinska Institutet
- Assistant Professor Xia Jiang, Karolinska Institutet
- Assistant Professor Carolina Hagberg, Karolinska Institutet
- Associate Professor Carolyn Cesta, Karolinska Institutet
- Associate Professor Mina Rosenqvist, Karolinska Institutet
- Professor Charlotte Ling, Lund University
- Professor Inger Sundström-Pooroma, Uppsala University
- Professor Claes Ohlsson, University of Gothenburg
International collaborators
- Professor Richard Legro, Pennsylvania State University, US
- Professor Andrea Dunaif, Icahn School of Medicine and Mount Sinai Health System, US
- Professor Thomas Jansson, University of Colorado Denver, US
- Professor Helena Teede, Monash University, Australia
- Professor Adam Balen, Leeds University Hospital, UK
- Professor Jie Qiao, Peking University Third Hospital, China
- Professor Matti Poutanen, Turku University, Finland
- Nordic PCOS Network: Dorte Glintborg and Marianne Andersen, Odense University, DK; Eszter Vanky, NTNU Trondheim, Norway; Terhi Piltonen and Laure Morin Papunen, University of Oulu, Finland; Juha Tapanainen, University of Helsinki
Research projects
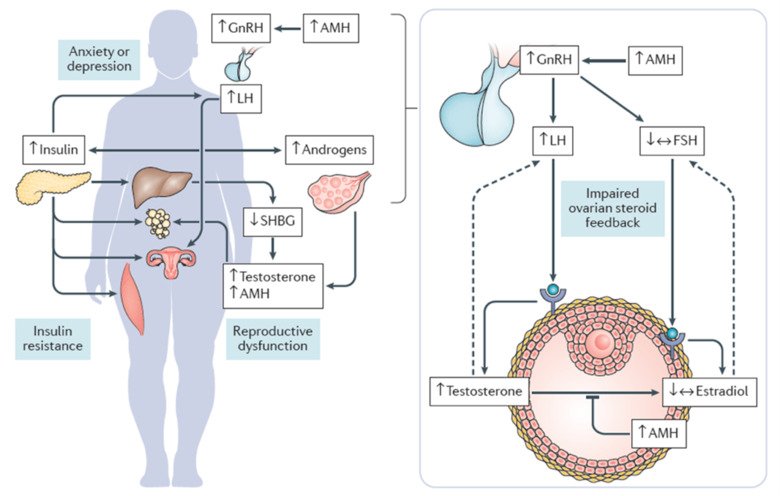
Polycystic ovary syndrome (PCOS) is linked to reproductive and metabolic disturbances as well as to mental health conditions such as anxiety and depression. Our research aims to gain deeper understanding of the etiology and heritability of PCOS and associated comorbidities such as type 2 diabetes, endometrial dysfunction, and mental-health disorders across generations by dissecting the molecular, mechanistic, and causal links, and to determine whether men can also transmit the disease.
Delineate the roles of different androgen triggers and molecular pathways that drive transgenerational transmission of PCOS-like traits
We know that hyperandrogenemia plays a key pathogenic role and that PCOS runs in families, with an estimated heritability of 70%. In a register-based study of nearly 30,000 daughters of women with or without PCOS, we found that daughters of women with PCOS have a five-fold increased risk of being diagnosed with the syndrome (Risal et al. Nature Medicine, 2019). But how PCOS is inherited remains unclear.
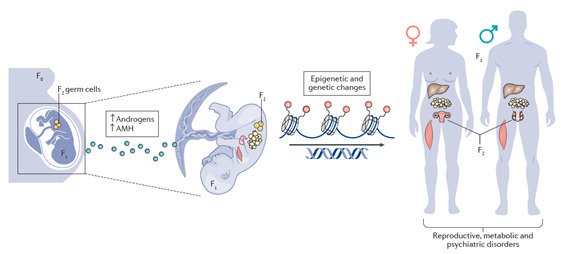
Accumulating evidence suggests that an epigenetic process triggered by an adverse maternal-fetal environment could yield the same phenotypic heritability as conventional genetics. Recently we made the discovery that PCOS-like traits induced by maternal androgen-exposure (Risal et al. Nature Medicine, 2019) can be passed on in mice from mothers (F0) to daughters (F1), granddaughters (F2), and even to great-granddaughters (F3), and that transcriptional and mitochondrial perturbations of oocytes accompany the transgenerational transmission. These intriguing findings raise the possibility that epigenetic modifications carried by germ cells and/or somatic cells can transmit PCOS across multiple generations. But they do not account for how males are affected and for the genetic contribution of the inheritance. Neither do these experiments disentangle the contribution of germ cells from that of the in utero environment in driving transgenerational transmission of PCOS.
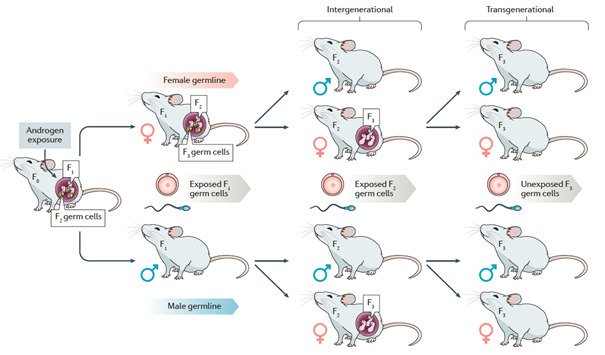
Define cell-type-specific disease signatures in endometrium, adipose and skeletal muscle tissues
Despite increasing evidence that multiple aspects of adipose tissue, skeletal muscle, and endometrial dysfunctions in women with PCOS are regulated by both genetic and epigenetic mechanisms, the contribution of cellular heterogeneity to these tissue dysfunctions is unclear. To gain insight into the cellular complexity and to tease apart genetic and epigenetic influence, we will characterize gene expression and DNA methylation at the single-cell level in endometrial, skeletal muscle and adipose tissues of women with and without the syndrome.
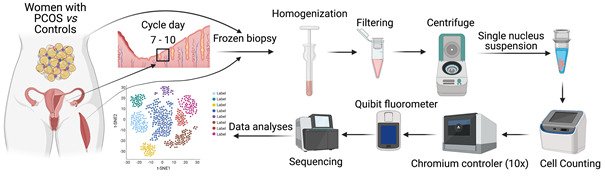
Establishment of endometrial organoids and adipocyte spheroids
Endometrial organoids (EOs) and adipocyte spheroids (ASs) circumvent constraints associated with fresh tissue and are unique powerful tools for pathophysiological research, functional testing of cell-type-specific disease markers, and drug screening of novel therapeutic targets. We establish EOs of endometrial tissue and ASs of adipose tissue (in collaboration with Carolina Hagberg, KI) of women with PCOS and controls. These will be used to test how e.g. sex steroids and/or insulin affect altered signaling pathways identified by cell-type specific large-scale sequencing.
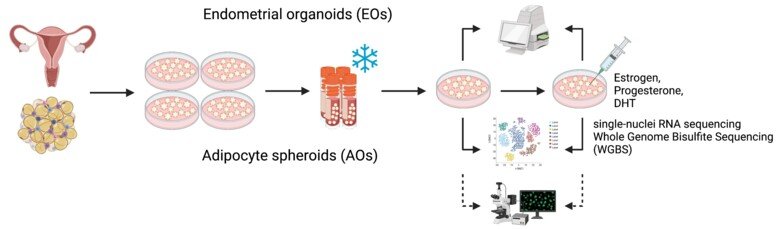
To elucidate the effect and mechanisms of metformin and acupuncture on insulin resistance and key signaling pathways in target tissues in women with PCOS
Prevention of PCOS is limited and management inadequate due to lack of mechanistic insights of PCOS. Changes in diet and lifestyle is the first choice for improving insulin sensitivity and preventing type 2 diabetes. Metformin is used in those with impaired glucose tolerance and type 2 diabetes who do not respond to lifestyle changes and works mainly by reducing hepatic gluconeogenesis and to a lesser extent by increasing glucose uptake in skeletal muscle and adipose tissue. An alternative approach to metformin and exercise is electroacupuncture treatment. We have in an uncontrolled study showed that repeated low-frequency electroacupuncture improves insulin sensitivity, decreases excess androgen levels, and restores altered epigenetic and transcriptional changes in adipose tissue and that the effect is mediated by neuronal circuits in the sympathetic and endocrine system. Moreover, our recent findings provide evidence that a single bout of electroacupuncture normalizes gene expression in skeletal muscle in a manner like acute exercise. Electroacupuncture might therefore be a useful way of assisting those who have difficulties performing exercise. In an on-going randomized control (RCT) (NCT01457209) we investigate the effectiveness of metformin, low-frequency electroacupuncture, and lifestyle management for improving insulin sensitivity in women with PCOS. Moreover, we explore whether these treatments can remodel transcriptional changes and restore molecular dysfunctions and in endometrium, adipose tissue and skeletal muscle tissue biopsies at baseline, after 16 weeks of treatment and at follow up to advance our understanding of PCOS-specific insulin resistance.
Determine the role of the immune system in the pathology of PCOS
Women with PCOS suffer from chronic low-grade inflammation and some even develop autoimmune diseases, which are further aggravated by metabolic disturbances that characterize the syndrome. Accumulating evidence suggests a link between PCOS and immune responses, but whether the observed immune hyperactivation is a cause or consequence of hyperandrogenism, and whether this immunophenotype is passed on to subsequent generations is unknown. To investigate what role the immune system play in the pathology of PCOS we currently investigate whether:
- Transfer of self-reactive IgG from women with PCOS to mice induce a PCOS-like phenotype?
- Characterize immunophenotype in androgen-induced PCOS mice.
- Determine whether transferring B cells from androgen-induced PCOS mice to reconstituted B-null mice induces development of a PCOS-like immunophenotype.
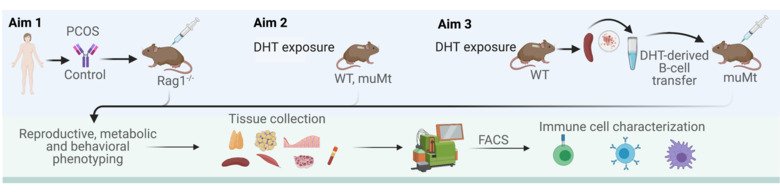
Dissertations
Main supervisor: group leader Elisabet Stener-Victorin
2020 – Maria Manti, MSc, Nutrition Science
The Title of the thesis: Developmental origins of polycystic ovary syndrome: Role of early adverse life events on adult health. Department of Physiology and Pharmacology, Karolinska Institutet
2018 – Milana Kokosar, Msc Pharmacy. PhD.
Title of PhD project: Polycystic ovary syndrome – Androgen Excess and Insulin resistance in women: Identification of molecular targets to improve glucose homeostasis. Institute of Neuroscience and Physiology, Endocrinology, University of Gothenburg.
2017 – Romina Fornes, Msc Reprod Biology and Midwife. PhD.
Title of PhD project: Polycystic ovary syndrome – Role of androgens and obesity on placenta function and fetal development. Department of Physiology and Pharmacology, Karolinska Institutet
2013 – Julia Johansson, Msc Medical Engineering. PhD.
Title of thesis: Polycystic ovary syndrome - Effect of acupuncture on insulin resistance and neuroendocrine function. Institute of Neuroscience and Physiology, Endocrinology, University of Gothenburg.
2010 – Elizabeth Jedel, PhD.
Title of thesis: Polycystic Ovary Syndrome: Studies of affective symptoms in association with sex steroids and evaluation of electroacupuncture and physical exercise. Institute of Clinical Neuroscience, OCIM, Karolinska Institute, Stockholm.
2010 – Louise Mannerås Holm, PhD.
Title of thesis: Polycystic ovary syndrome: Studies of metabolic and ovarian disturbances and effects of physical exercise and electro-acupuncture. Institute of Neuroscience and Physiology, University of Gothenburg.
2009 – Lena Svedberg, PhD.
Title of thesis: Cold feet in children with neurological disorders. Institute of Neuroscience and Physiology, University of Gothenburg.
2007 – Annika Billhult, PhD.
Title of thesis: The effect of massage for women with breast cancer. Institute of Neuroscience and Physiology, University of Gothenburg.
2005 – Luigi Manni, PhD.
Title of thesis: Adrenoceptors and Nerve Growth Factor: Effects of Electro-Acupuncture and Physical Exercise in Rats with Steroid-Induced Polycystic Ovaries. Cardiovascular Institute, University of Gothenburg