
- >
- >
- Neuroplasticity and Regeneration – Konstantinos Ampatzis group
- Paediatric cell and molecular biology – Hjalmar Brismar's research group
- Quantitative Biology of the Nucleus – Magda Bienko Group
- Cardiometabolic factors, brain aging, and dementia care – Weili Xu group
- Cognitive Neuroscience of Body and Self – Henrik Ehrsson Group
- Surgical Care Science – Pernilla Lagergren's research group
- Clinical and translational studies on viral hepatitis – Soo Aleman group
- Cell and Gene Therapy – Evren Alici group
- Personalized Medicine and Drug Development – Lauschke-lab
- Electrophysiological neuropharmacology – Göran Engberg's research group
- Mechanisms of Pain and Treatment – Eva Kosek's research group
- Functional (Epi)genomics of Neuroinflammatory Diseases – Maja Jagodic's research group
- Molecular basis of gene regulation of diseases – Carsten Daub's research group
- Health risk assessment methodology – Anna Beronius' group
- Medical genetics – Catharina Larsson's Group
- Translational Genetics of Neurodegenerative disease – Caroline Graff's research group
- Development, transcription factors, neurons, dopamine – Thomas Perlmann's Group
- Clinical and translational research within lupus and autoimmunity – Team Ioannis Parodis
- Cell cycle, mitotic entry, and DNA-damage checkpoints – Arne Lindqvist's Group
- Breast Surgery – Irma Fredriksson's research group
- Protein dynamics and mutation – Laura Orellana's Group
- Research group Ali Mirazimi
- Early drug discovery research in chronic inflammatory diseases – Research group Michael Sundström
- Integrative Physiology – Juleen Zierath's research group
- Epigenetics, Synthetic and Systems Biology – Simon Elsässer group
- Clinical Neuroimmunology and Immunomodulation – Fredrik Piehl's research group
- Understanding mechanisms of vaccination – Research group Karin Loré
- Systems regenerative neurobiology – Enric Llorens Group
- Clinicial Cancer Genomics – Johan Lindberg's research group
- Jeanette Hellgren Kotaleski group
- Brain control of food intake and body weight – Björn Meister group
- Research groups in endocrinology and diabetes
- Research groups in gastroenterology and hepatology
- Research groups in neurosciences
- Research groups in respiratory medicine and allergy
- Caring in Community Care – Zarina Nahar Kabir's research group
- Molecular mapping of the nervous system in health and disease – Jan Mulder group
- Child and adolescent psychiatry – Jens Högström's research group
- Genetic mechanisms of ageing – Maria Eriksson's research group
- Digital Psychiatry – Philip Lindner's research group
- Synaesthesia, autism and perception – Janina Neufeld's team
- Vascular complications in cardiometabolic disease – Research group Zhichao Zhou
- Urological Research at Danderyd Hospital – Tobias Nordström's research group
- Lars Karlsson team
- Sepsis and microcirculation – Sara Tehrani's research group
- Regulation of B cell activation – Pia Dosenovic Group
- Complex interventions – Team Carina King
- Chronic pain and rehabilitation – Monika Löfgren's research group
- Translational Molecular Imaging – Nordberg and Chiotis Lab
- Dagliyan Lab
- Developmental and Translational Neurobiology – Cristiana Cruceanu's research group
- Microbiota–gut–brain axis and neurodevelopment – Rochellys Diaz Heijtz group
- Thoracic Surgery – Anders Franco-Cereceda's research group
- Electrophysiology – Mats Jensen-Urstad group
- Role of T cells in human host defense – Johan Sandberg group
- Germ cell biology and developmental programming in epigenetic inheritance of diseases – Qiaolin Deng's research group
- Unit for Bioentrepreneurship – Hanna Jansson's group
- Health informatics – Sabine Koch's group
- Cancer, rapid ageing and nutrition – Martin Bergö's research group
- Clinical radiation therapy – Åsa Carlsson Tedgren's Group
- TRANslational Theranostics Group – Thuy Tran
- Pathogenic pathways in Alzheimer Disease – Lars Tjernberg's research group
- Hematological and solid tumors – Research group Xu Dawei
- Developmental Neurogenomics – Michael Ratz's Group
- Notch signaling, ES cells, myogenic and vascular progenit breast cancer – Urban Lendahl's Group
- Particle toxicology – Hanna Karlsson's group
- Mitochondrial dysfunction in Alzheimer Disease – Maria Ankarcrona's research group
- Molecular pathology of the lung and pleura – Katalin Dobra's Group
- Late effects and cancer survivorship after diagnosis and treatment of aggressive lymphoma – Team Sandra Eloranta
- Research Group Anna Nopp
- Coding and non-coding RNAs in cancer – Per Hydbring's Team
- Research group Mikael Björnstedt
- Sister chromatid cohesion in DNA damage responses, DSB repair, Genome Integrity, Gene regulation – Lena Ström's Group
- Inborn Errors of Endocrinology and Metabolism – Anna Wedell's research group
- Functional Precision Medicine – Brinton Seashore-Ludlow's Team
- Understand skin using modern biology – Maria Kasper's Group
- Integrative Epidemiology – Fang Fang's research group
- Molecular Biometry – Roman Zubarev group
- Idiopathic pulmonary fibrosis (IPF) – Research group Magnus Sköld
- Molecular Neuroscience – Carlos Ibáñez group
- Research groups in psychology
- Research groups in forensic science
- Research groups in medical genetics and genomics
- Research groups in nursing
- Research groups in rheumatology and autoimmunity
- Genetic epidemiology of prostate and testicular cancer – Fredrik Wiklund's research group
- Systems biology of aging – Juulia Jylhävä's research group
- Aging – basic mechanisms and interventions – Christian Riedel's research group
- Embryonal, foetal and brain development – Juha Kere's research group
- Research groups in occupational therapy
- Growth and Cartilage Biology – Ola Nilsson's research group
- Infections and immunity in children with cancer – Anna Nilsson's research group
- Oxygenation, early resuscitation in trauma and drug treatment in cardiac arrest – Team Malin Jonsson Fagerlund
- Prehospital Emergency Care – Veronica Vicente's Research Group
- The brain after surgery and trauma - neuroinflammation, cognitive impairment and dementia – Lars I Eriksson research group
- Personalized diet and medications for cognition – Garcia-Ptacek group
- Improved synthetic DNA structures by evolutionary selection – Erik Benson Group
- Lifestyle and Kidney Cancer – Stephanie Bonn Team
- The research area of Nephrology at KI DS
- Visual Neuroscience – Rune Brautaset's research group
- Chemicals and female fertility – Pauliina Damdimopoulou Research Group
- Molecular and cellular exercise physiology – The Ruas Lab
- Neural circuits of cognition – Marie Carlén group
- Colorectal Surgery – Anna Martling and Caroline Nordenvall's research group
- Myeloma group – Evren Alici's research group
- Medicine and history of science – Eva Åhrén's group
- Cardiometabolic diseases – Ping Chen research group
- Anders Mutvei research group
- Ocular Oncology and Pathology – Gustav Stålhammar's research group
- Clinical Neurophysiology – Charith Cooray's research group
- Regulation of Gene Expression during Viral Infection – Gerald McInerney Group
- Social and affective learning and decision-making – Andreas Olsson's research group
- Translational Breast Cancer Research: Long-Term Risk and Endocrine Treatment Benefit – Linda Lindström's Group
- Notch3 and the cerebral small vessel disease CADASIL – from molecular mechanisms to treatment strategies – Helena Karlström's research group
- Dementia and Multimorbidity – Dorota Religa's research group
- Diagnostic Radiology – Lennart Nedar's research group
- Cognitive Neuropsychiatry – Predrag Petrovic's research group
- Chemical carcinogenesis – Ulla Stenius' group
- Metal toxicology – Maria Kippler's research group
- Cellular Stress in Health and Disease – Federico Pietrocola's group
- Autophagy and cardiovascular disease – Team Ewa Ehrenborg
- Research group Birgitta Sander & Birger Christensson
- Innate Immune Regulation – Quirin Hammer team
- Genetics of tumor predisposition and progression in breast cancer – Svetlana Bajalica Lagercrantz's Team
- Implementation and Quality (IMPAQT) – Claudia Hanson's research group
- Structural and Biophysical Immunology – Research group Adnane Achour
- Neural cell diversity in the Enteric Nervous System (ENS) – Ulrika Marklund group
- Sustainable work & occupational safety and health – Emma Brulin's research group
- Health Systems Leadership, Management, and Safety – Carl Savage's group
- Cell and Molecular Immunology - Research group Cecilia Söderberg Naucler
- Brain Circuits – Konstantinos Meletis group
- Petter Ljungman's research group- Health effects of the Ambient Environment
- Neuropsychology of music – Fredrik Ullén group
- Research groups in geriatrics and gerontology
- Research groups in nutrition and dietetics
- Research groups in sports and fitness sciences
- Prostate Cancer – Henrik Grönberg's research group
- Methods of health promotion in the workplace – Lydia Kwak's research group
- Autism and perinatal epidemiology – Sven Sandin research group
- Wengström's research group
- Modelling and simulation with applications to cancer epidemiology and health economics – Mark Clements' research group
- Health Technology Assessment – Monica Hultcrantz’s group
- Pediatric anesthesiology Stockholm – Per-Arne Lönnqvist's research group
- Biophysics – Erdinc Sezgin's Lab
- War, crisis, and security studies
- Systems Medicine – Johan Björkegren research group
- Translational Clinical Orthopaedics – Michael Axenhus Research Group
- Antigen-specific T cell repertoires – Cassotta lab
- The research area of Neurology at Karolinska Institutet – Danderyd Hospital
- Perpetration Prevention – Rahm and Joleby's research group
- SOLIID - Sustainable Organizational Learning, Innovation, Improvement and Development in Health and Social services – Monica Nyström's group
- Social Gerontology – Carin Lennartsson group
- Genetic and molecular basis of nervous system disorders – Andrea Carmine Belin group
- Chronobiology – Petrus lab
- Leukemic stem cells – Petter Woll group
- Nanomedicine and Spatial Biology – Teixeira Lab
- Global Disaster Medicine - Health Needs and Response – Johan von Schreeb's research group
- Translational cardiac and skeletal muscle physiology – Daniel Andersson's research group
- Neuroradiology – Staffan Holmin's research group
- Tumour genomics – Lauri Aaltonen's research group
- Neurobiology of Motor Actions – Abdel El Manira group
- Neuroinflammation in Alzheimer disease – Marianne Schultzberg research group
- Palliative Medicin – Linda Björkhem-Bergman's research group
- Nucleotide metabolism and molecular pharmacology – Sean Rudd's Group
- Cardiovascular Inflammation – Research group Peder Olofsson
- Mesenchymal stem cells – Katarina Le Blanc group
- Advancing precision immunotherapy in lung cancer through clinical, molecular, and AI-driven insights – Luigi De Petris' Team
- Autoimmunity and cancer – Research group Marie Wahren-Herlenius
- Atherothrombosis research – Team Nailin Li
- Precision cancer medicine – Olli Kallioniemi's group
- Research group Stephan Mielke
- Genetics of autoimmune diseases – Team Leonid Padyukov
- Musculoskeletal disorders; planetary health & biopsychosocial approaches – Nina Brodin's research group
- Vascular Functions in Metabolic and CNS Diseases – Ulf Eriksson group
- Sarcoma Genomics – Felix Haglund de Flon's Group
- Team Helene Alexanderson
- Injury and Repair in the Immature Brain – Blomgren group
- Statistical methods for cancer patient survival – Therese Andersson's research group
- Individual differences in cognition – Agneta Herlitz’ research group
- Research groups in health economy, healthcare management and policy
- Research groups in reproductive medicine and gynaecology
- Research groups in structural biology
- Research groups in artificial intelligence
- Work environment, health and productivity – Christina Björklund's research group
- Predictive medicine – Mattias Rantalainen's research group
- Epigenetic regulation of leukemia and normal blood development – Andreas Lennartsson's research group
- Epigenetic mechanisms underlying metabolic-inflammatory diseases – Eckardt Treuter's research group
- The IMPACT research group – Marie Löf
- Stockholm Center for Health Economics (StoCHE) – Emelie Heintz's team
- Towards personalized physiology in Intensive Care – Team Johan Mårtensson
- Ninib Baryawno research group
- Neurohormonal Basis of Physiological and Maladaptive Expressions of Survival Behaviors – Stefanos Stagkourakis group
- Medical Oncology in breast, prostate and bowel cancers – Pehr Lind's Research Group
- Arthroplasty – Olof Sköldenberg’s research group
- Anna Undeman Asarnoj team
- Neuroinflammatory disorders – Anders Svenningsson's research group
- Global infections – Research group Anna Färnert
- EcoMind and biological pathways in cognitive aging – Debora Rizzuto group
- Circuits controlling Action and their Evolution – Sten Grillner group
- Mikael Rydén & Niklas Mejhert lab
- Understand Fundamental Immune Mechanisms to Develop New Treatments for Lung Diseases – Tim Willinger group
- Clinical Brain Imaging Methods – Anna Falk Delgado's research group
- Hyperbaric medicine – Peter Lindholm's research group
- Eye movements and vision – Tony Pansell's research group
- Lung toxicology – Lena Palmberg's research group
- The NeuroCardioMetabol Group – Thomas Nyström and Cesare Patrone
- Environmental and genetic factors influence on neurodevelopment – Sandra Ceccatelli group
- Skin wound healing – Research group Ning Xu Landén
- From development to disease: decoding stem cell programs for CNS repair – Erik Sundström-Xiaofei Li group
- Neurodegenerative diseases and Aging – Daniel Ferreira Research Group
- Deep learning models of cancer mechanisms – Avlant Nilsson's Group
- Clinical Physiology – Marcus Carlsson's research group
- Biomaterials for Therapeutics and Regenerative Medicine – Molly Stevens group
- Molecular Mechanisms of Viral Oncogenesis – Maria G. Masucci's Group
- Björn Reinius group
- Quantitative principles in tumour biology – Jean Hausser's Group
- Bone biology – Sara Windahl Research group
- Research group Volkan Özenci
- Genetics and mechanisms of metals – Karin Broberg's research group
- Immunodeficiency Diseases – Lisa Westerberg Group
- Structural studies of fertilisation and zona pellucida module proteins – Luca Jovine's research group
- Tissue regeneration, focusing on fat cells (adipocytes) and obesity – Kirsty Spalding's Group
- Robert Månsson group
- Developmental and Regenerative Stem Cell Medicine – Lanner Lab
- Suicide and mental health lab – Vladimir Carli and Gergö Hadlaczky's group
- Medical, genetic and molecular decoding of neurodevelopmental disorders – Kristiina Tammimies' research group
- Working hours, Recovery and Safety in working life. – Anna Dahlgren's research group
- Research groups in hematology
- SOMIND: Somatic, Mental, and Neurodevelopmental Conditions – Agnieszka Butwicka's research group
- Research groups in drug abuse and addiction
- Research groups in biomaterials science
- Mikael Adner's research group
- Chemical and functional neuroanatomy of pain and stress systems – Tomas Hökfelt group
- Spider silk biology for biomedical applications – Anna Rising's research group
- Cancer Epidemiology and Causal Inference – Research group Maria Feychting
- Protein biochemistry for development of novel medical treatments – Janne Johansson's research group
- Healthcare financing and organization – Clas Rehnberg's group
- Space and environmental physiology – Rodrigo Fernandez-Gonzalo group
- Novel treatment approaches for neuroblastoma – Malin Wickström Näsman research group
- Cancer immune and gene therapy – Rolf Kiessling's Team
- Breast cancer precision medicine – Marike Gabrielson's research group
- Trauma – Max Gordon's research group
- Obstetrics and Gynecology at KI DS
- Atopic dermatitis – Research group Emma Johansson
- Reproductive endocrinology and metabolism – Angelica Lindén Hirschberg's research group
- Genetic basis for B and T cell recognition and function – Gunilla Karlsson Hedestam Group
- Cognitive Aging and Mental Health – Erika Jonsson Laukka Group
- Molecular and circuit neuropharmacology – Gilberto Fisone group
- The role of immunity in shaping adipose tissue development and metabolism – Jurga Laurencikiene group
- Chronic myeloid malignancies – Johanna Ungerstedt group
- Studies of tissue microbiology and immunology – Mattias Svensson group
- Cellular composition of the human brain and brain cancer – Sten Linnarsson group
- NAVIGATE research group – Neurology, ActiVity, Imaging, GAit, Technology and Exercise
- Global Health Pharmacology and Therapeutics (GH-Pharma) – Eleni Aklillu's research group
- Real-world-data in Multiple Sclerosis – Anna Glaser's research group
- Suicidology and transcultural psychiatry – Marie Dahlin's research group
- Experimental and Clinical Neuroendocrinology – Maria Petersson's research group
- Cutting edge translational research of common and rare skin disease – Research group Jakob Wikström
- Alzheimer’s Disease Risk Factors & Sex-Specific Mechanisms – Silvia Maioli's research group
- Frontal lobes and cognitive failure – Wahlund's research group
- Clinical Pharmacology – Research group Rickard Malmström
- Molecular Vascular Medicine – Team Lars Maegdefessel
- Molecular endocrine pathology – Christofer Juhlin's Group
- Presynaptic mechanisms – Lennart Brodin group
- Malignant melanoma – Hildur Helgadottir's Group
- Precision strategies to eradicate heterogeneous tumor cells in pediatric cancer – Kasper Karlsson's Group
- Respiratory and systemic immune responses in human pulmonary viral infection and inflammation – Research group Anna Smed Sörensen
- Genetic toxicology – Kristian Dreij's group
- Identifying new molecular targets for the treatment of motor neuron diseases – Eva Hedlund's research
- Epigenetics - basic mechanisms and disease – Karl Ekwall's research group
- Forensic medicine – Henrik Druid's Group
- Ageing and Health – Karin Modig's research group
- Perception Neuroscience – Johan Lundström's research group
- Computational medicine and bioinformatics – Trung Nghia Vu's research group
- Toxicology – Bertrand Joseph´s Research group
- Etiology, prevention and treatment of eating disorders – Ata Ghaderi's research group
- Research groups in medical ethics and history of science
- Research groups in occupational and environmental health
- Research groups in surgery
- Research groups in biophysics
- Research Group Johan Ärnlöv
- Narrative in health and social care – Research group
- Cellular and molecular mechanisms underlying intervertebral disk formation and Diversity and plasticity of adipose tissue cell niche – Meng Xie's research team
- Neuroimmunology of cancer – Sébastien Talbot
- Pediatric Systems Immunology – Petter Brodin's research group
- Microvascular oxidative stress in the triad of cardiovascular, metabolic and renal disease – Mattias Carlström group
- Cardio-renal epidemiology – Juan-Jesus Carrero's research group
- Human Translational Genetics – Stefano Romeo Group
- Clinical Pancreatology – Miroslav Vujasinovic research group
- Organoids, Cell identity, Development and Regeneration – Amanda Andersson Rolf's Group
- D-WHIN: Danderyd Wrist and Hand INitiative – Research group
- Gynecological research – at Danderyd Hospital
- Research area Medicine at KI DS
- Movement, Activity and Health – Eva Broström's research group
- Multidimensional health in old age – Amaia Calderón-Larrañaga group
- Molecular epidemiology of aging – Sara Hägg's research group
- Long noncoding RNA in the adipocyte – Alastair Kerr Research Group
- Leukemia niche – Hong Qian group
- Disease mechanisms in severe acute bacterial infections – Anna Norrby-Teglund group
- Gastrointestinal pediatric surgery – Tomas Wester's research group
- Developmental biology and regenerative medicine – Igor Adameyko's research group
- Experimental audiology – Barbara Canlon's research group
- Translational Immunology in Neuroinflammation – Olivia Thomas' research group
- Sickness absence, health and living conditions – Emilie Friberg's research group
- Health Systems and Policy – Cecilia Stålsby Lundborg's research group
- Endocrinology and autoimmune diseases – Research group Sophie Bensing
- Health economics of Alzheimer disease and other dementias – Linus Jönsson Group
- Epidemiology: Diet, contaminants and health – Agneta Åkesson's research group
- Neuronal circuits of anxiety – Janos Fuzik group
- Paediatric rheumatology – Research group Helena Erlandsson Harris
- Cardiovascular Epidemiology – Karin Leander research group
- Molecular cardiology – Research group Francesco Cosentino
- Björn Högberg group
- Regeneration mechanisms – Andras Simon's Group
- Assessing and managing chemical risks – Linda Schenk's research group
- Autoimmunity and adaptive immunity in rheumatic disease – Research group Vivianne Malmström
- Rheumatic systemic diseases – Team Marie Holmqvist
- Heart failure with reduced and preserved ejection fraction. Clinical and translational aspects. – Research group Lars Lund
- Gene Regulation and Alternative Splicing in development and disease – Ziegenhain Group
- Psychiatric epidemiology – Paul Lichtenstein's research group
- Structural basis of RNA biogenesis – Martin Hällberg's research group
- Hematopoietic Stem Cell Biology Group – Sten Eirik W. Jacobsen Lab
- Psychiatric Genetic Epidemiology – Sarah Bergen's research group
- Renal Research Novum – Bengt Lindholm Group
- Research groups in biochemistry
- Psychological treatment: internet and exposure – Brjánn Ljótsson's research group
- Research groups in immunology
- Research groups in ophthalmology
- Research groups in urology and nephrology
- Research groups in sociology and migration
- Integrative physiology – Juleen Zierath and Anna Krook
- Hormonal signalling and cancer – Cecilia Williams research group
- Cell Biology of Cancer – Staffan Strömblad's research group
- Chronic inflammation with focus on rheumatology and cancer – Research group Per-Johan Jakobsson
- Health services and social work research – Lena Von Koch's research group
- Nitrate-nitrite-NO pathway in health and disease – Eddie Weitzberg's research group
- Medical sensors and diagnostics – Team Onur Parlak
- Uncovering the molecular and physical principles governing early embryonic division and nuclear organization – Jan Ellenberg Group
- Hemostasis and Thrombosis – Mika Skeppholms research group
- CAIR-LAB: Clinical AI Research Laboratory – group
- Research area orthopedics at KI DS
- Amyotrophic Lateral Sclerosis - ALS – Caroline Ingre's research group
- Saving the brain in critically ill neonates – Ulrika Ådén's research group
- Regulatory Transcriptomics – Kutter group
- NeuroimAging – Grégoria Kalpouzos group
- Section of Pharmacogenetics – MIS-lab
- Biological and clinical aspects of the myelodysplastic syndrome – Magnus Tobiasson group
- Human innate lymphoid cell lab – Jenny Mjösberg group
- Viruses and their interactions with the immune system – Sara Gredmark Russ group
- Protein degradation pathways – Helin Norberg's research group
- Translational Auditory Neuroscience – Christopher Cederroth's research group
- Neuroradiology: Neurodegeneration and Neuroinflammation – Tobias Granberg's research group
- Tumor Immunology and Immunotherapy Group – Dhifaf Sarhan
- Causal inference in epidemiologic research – Arvid Sjölander's research group
- The role of autophagy in Aβ metabolism and neurodegeneration in Alzheimer’s disease – Per Nilsson group
- Cholinergic Pathobiology & PET-Imaging – Darreh Shori Group
- PROCOME – Hanna Öfverström's and Marta Roczniewska's group
- Molecular brain imaging of neurodegenerative disorders – Andrea Varrone's research group
- Alcohol and Drug Epidemiology – Mats Ramstedt's research group
- Applied psychiatric research and innovation – Christian Rück's research group
- Psychotherapy Research – Tobias Lundgren's research group
- Human olfaction – Mats J. Olsson's research group
- Tissue stem cells and aging – Pekka Katajisto's Group
- SOSVASC – David Lindström's research group
- Yenan Bryceson group
- Epidemiology of sarcoidosis and systemic lupus erythematosus – Team Elizabeth Arkema
- Viral genomics and metagenomics – Tobias Allander Group
- Defining the metabolic signatures of tissue resident human ILCs – Chris Tibbitt team
- Human papillomavirus which causes cervical cancer – Research Group Karin Sundström
- Research groups in bioinformatics and systems biology
- Work and Organizational Psychology – Marika Melin's research group
- Gilad Silberberg group
- Research groups in oto-rhino-laryngology and logopaedics
- Immunoregulation of environmental-induced airway inflammation in severe asthma and bronchiectasis – Apostolos Bossios's research group
- Multimorbidity and frailty in older adults – Davide Liborio Vetrano group
- Psychiatric and pharmaco-epidemiology – Zheng Chang's research group
- Charting normal and malignant hematopoiesis – Team Joakim Dahlin
- Molecular mechanisms governing oxygen homeostasis in development, evolution, and disease – Kragesteen's research group
- Precision Medicine – Infrastructure team
- Research groups in medical epigenetics and epigenomics
- Spinal circuits for whole-body motor coordination – Laurence Picton group
- Clinical HIV epidemiology and comorbidity – Christina Carlander Research group
- Nanoscale lipid flux architecture – Veijo Salo’s Research Group
- Molecular mechanisms of drug action – Shane Wright's team
- Pediatric and respiratory epidemiology – Catarina Almqvist Malmros' research group
- Sexual and Reproductive Health – Marie Klingberg-Allvin research group
- Angiogenesis, Cancer, Metabolic disease, Cardiovascular disease, Eye disease – Yihai Cao Group
- Cardiovascular and psychosocial health in resilience and healthy brain aging – Chengxuan Qiu Group
- Translational Psychiatry – Catharina Lavebratt's research group
- Rare metabolic liver diseases, coagulation and hepatocellular carcinoma – Stål and Wahlin research group
- Blood Engineering – Vanessa Lundin group
- Development, regulation and function of human innate lymphoid cells throughout life – Jakob Michaëlsson group
- Clinical Genetics – Richard Rosenquist Brandell's research group
- Oxysterols – Ingemar Björkhem and Ulf Diczfalusy research
- Molecular exercise physiology – Carl Johan Sundberg's research group
- Large-scale Network Connectivity in the Human Brain – Peter Fransson's research group
- Environmental physiology
- The Systems Virology Lab – Ujjwal Neogi
- Cancer Cell Invasion – Marco Gerling's research group
- Molecular brain imaging, neuropsychopharmacology, depression and anxiety disorders – Johan Lundberg's research group
- Organization and operation of postural neuronal networks in health and disease – Tatiana Deliagina group
- Paediatrics – Anna Lindholm Olinder's research group
- Petzold group
- Molecular and epidemiological studies of ascending aortic aneurysms – Research group Hanna Björck
- Prevention, Intervention and Mechanisms in Public Health (PRIME Health) – Cecilia Magnusson's research group
- The interplay between circadian 3D genome organization and metabolism in complex diseases – Anita Göndör's Group
- Reconstructive Plastic Surgery and Global Surgery – Jenny Löfgren's research group
- Real-world effectiveness of psychopharmacological treatment – Jari Tiihonen's Research Group
- Center for Heart Failure and Arrhythmia – Gianluigi Savarese
- Sports Medicine – Anders Stålman's research group
- Women's Mental Health Epidemiology – Donghao Lu Lab
- The SMC complexes, chromosome dynamics and stability – Camilla Björkegren's Group
- Extracellular vesicles in immunity – Research group Susanne Gabrielsson
- Precision Cancer Medicine in Lung Cancer - Preclinical, Translational & Clinical Research – Simon Ekman's research group
- RNA-guided DNA repair and cancer – Marianne Farnebo's research group
- Laboratory of translational fertility preservation – Kenny Rodriguez-Wallberg's group
- Research groups in cancer and oncology
- Common Mental Disorders and Behavioral Medicine in Primary Care – Erik Hedman's research group
- Neuronal membrane trafficking – Oleg Shupliakov group
- Research groups in paediatrics
- Biostatistics With Focus on Statistical Methods in Cancer Epidemiology and Screening – Keith Humphreys' research group
- Research groups in nanotechnology
- Gastroenterology – clinical epidemiology and clinical trials – Research group Ola Olén
- Precision engineering of T cells in vivo for therapies of cancer – William Nyberg team
- Protein Misfolding and Prevention by Molecular Chaperones in Cancer and Neurodegenerative Disease – Gefei Chen's research group
- Social Perception – Arvid Guterstam's research group
- Neuroepidemiology – Kyla McKay and Katarina Fink's research group
- Experimental growth research – Lars Sävendahl's research group
- Neuronal circuits of the Basal Ganglia – Maya Ketzef group
- Psychological Interventions - innovation, improvement and implementation – Viktor Kaldo's research group
- Susanna Ranta team
- Head and neck pathology – Anders Näsman's Team
- Residual conditions after polio and neuromuscular diseases – Eva Melin's research group
- Sepsis, Critical Illness, and Perioperative Optimization – Maria Cronhjort’s Research Group
- Selenoproteins and redox biology – Elias Arnér research group
- Gynecologic Cancer – Henrik Falconer's research group
- B cell biology, from immunodeficiency to cancer – Pan Hammarström Lab
- Developmental Cognitive Neuroscience – Torkel Klingberg group
- Rare Diseases – Ann Nordgren och Anna Lindstrand's research group
- Research on steatotic liver disease – Hagström Group
- Stem Cell Size – Jette Lengefeld Team
- Natural Killer Cell Biology and Cell Therapy – Kalle Malmberg group
- Mast Cell Biology – Research group Gunnar Nilsson
- Traumatic Brain Injuries and Neuromonitoring – Eric Thelin's research group
- Medical Statistics team
- Augmented Neurosurgery – Erik Edström and Adrian Elmi Terander's research group
- Molecular neurophysiology – Karima Chergui's research group
- Injuries Social Aetiology and Consequences (ISAC) – Lucie Laflamme's research group
- Unconventional T cells in human pathology – Andrea Ponzetta team
- Inflammation and immune regulation during infection – Research group Christopher Sundling
- Orthopaedics – Anders Enocson's research group
- Endothelial dysfunction in atherosclerotic cardiovascular disease – Research Group John Pernow
- Clinical epidemiology of multiple sclerosis and inflammatory joint diseases – Team Thomas Frisell
- Cancer stem cells and clonal structure in acute lymphoblastic leukemia – Martin Enge's Group
- Mitochondrial metabolism in health and disease – Anna Wredenberg group
- Biomolecular Medicine and Advanced therapies- focus on delivery of RNA therapeutics – Research group Samir EL Andaloussi
- Antimicrobial resistance – Christian Giske research group
- Tissue immunology – Research group Helen Kaipe
- Health Economics and Economic Evaluation – Niklas Zethraeus' group
- Mental Health Neural Dynamics Lab – Kristoffer Månsson's research group
- Stem Cell Biology – Jonas Muhr's Group
- VIVAC research group: Vaccines and Immunotherapies against Viruses And Cancer – Matti Sällberg
- The passage of the mRNP particle through the nuclear pore – Bertil Daneholt's research
- Therapeutic Immunology and Transfusion Medicine – Michael Uhlin’s research group
- Research groups in cancer and oncology
- Nutritional neuroscience – Janina Seubert's Research Group
- Tobias Karlsson's group
- Research groups in pharmacology and toxicology
- Precision Psychiatry – Lu Yi's research group
- Research groups in medicinal chemistry
- Agneta Richter-Dahlfors group
- Cancer epidemiology with focus on breast cancer and applied biostatistics – Anna Johansson's research group
- Autism, ADHD and other neurodevelopmental conditions – Sven Bölte's research group
- Twinstudies on health conditions and sickness absence – Pia Svedberg's research group
- Psychiatric comorbidity and intergenerational transmission of mental health problems – Erik Pettersson's research group
- Translational neuroblastoma and medulloblastoma research – John Inge Johnsen research group
- Genetic and pharmacological epidemiology – Zeberg laboratory
- REACH – digital treatment and prevention – David Moulaee Conradsson's research group
- Mucosal immunology lab – Charlotte Thålin research group
- Neurovascular diseases – Christina Sjöstrand research group
- Carin Håkansta's research group – Digital work environment and health
- Proteomes of Metabolism – Florian Rosenberger's Research Group
- Juan Du Lab
- Single-cell and spatial omics to investigate mechanisms underlying multiple sclerosis and disease-associated oligodendroglia – Gonçalo Castelo-Branco Group
- Pediatric Neuroimmunology and Epilepsy – Ronny Wickström's research group
- Explores various cell signaling phenomena and their impact on critical biological and medical processes using advanced light microscopy – Per Uhlén Group
- Neural stem cells – Anna Falk group
- Diabetes and Associated Complications – Sergiu-Bogdan Catrina's research group
- Myelodysplastic syndromes – Eva Hellström Lindberg group
- Immune responses to human viral infections and cancer – Hans-Gustaf Ljunggren group
- Health services research in neurological conditions – Charlotte Ytterberg's research group
- Neurosurgery – Mikael A. Svensson's research group
- Westman neuroimaging group
- Stemcells and Inflammation – Lou Brundin's research group
- Pharmacological nitric oxide research – Jon Lundberg's research group
- Epidemiology of Psychiatric Conditions, Substance use and Social Environment (EPiCSS) – Emilie Agardh's & Renee Gardner's Research Group
- Autoimmune neurology – Jakob Theorell team
- Reducing antibiotic resistance – Research group Pontus Nauclér
- Orthopaedics – Karl Eriksson's research group
- Endocrine Surgery – Robert Bränström's research group
- Paediatrics – Inger Kull's research team
- Cancer proteogenomics - from methods to clinical applications – Janne Lehtiö's Group
- Primary Immunodeficiency, Innate Immunity and Antimicrobial peptides – Peter Bergman Research Group/The AMP-group
- Clinical Virology and Immunology – Annika Karlsson's research group
- Jussi Taipale Group
- Global Child Health and the Sustainable Development Goals – Tobias Alfvén's research group
- Topoisomerases, chromatin biology and cancer – Laura Baranello's group
- Stem cells and neural development – Johan Ericson's Group
- Vascular morphogenesis and function in health and disease – Lars Jakobsson group
- Epidemiology and Public Health Intervention Research (EPHIR) – Jette Möller's research group
- Epithelial stem cells in development and disease – Maria Genander's group
- Research Group Gustaf Edgren
- Drug discovery, pancreatic beta-cell regeneration – Olov Andersson's Group
- Unit of Biostatistics – Matteo Bottai
- Research groups in cardiology and cardiovascular diseases
- Sleep, cognition and health – John Axelsson's research group
- Research groups in infectious medicine
- Research groups in physiology and anatomy
- Epidemiology of cardiometabolic and cognitive aging – Ida Karlsson's research group
- How the overview of research fields is made
- Precision Cancer Control Group – Martin Eklund's research group
- Liver and monocyte remodelling in non-alcoholic fatty liver disease and cardiovascular disease – Rongrong Fan's research group
- Early childhood development and neurodevelopmental conditions – Terje Falck-Ytter's research group
- Mental health and social integration (Mente) – Ellenor Mittendorfer-Rutz's research group
- Health outcomes in autistic people – Mark Taylor's research group
- Mechanisms of protein aggregation and inhibition – Axel Abelein group
- Accelerating drug discovery using molecular modeling and machine learning – Andreas Luttens' group
- Pediatric Healthcare Science – Cecilia Bartholdson research group
- Digital Strategies for AF Detection and Outcome Prevention – Emma Svennberg Team
- Stroke – Annika Lundströms research group
- Lifestyle, Prevention and Health – Ylva Trolle Lagerros research group
- Antibody-mediated neuroinflammation – Susanna Brauner's research group
- Computational Biology, Virology, and Immunology – Murrell Lab
- Environment, nutrition and health – Anna Bergström's research group
- Genetic and epigenetic factors in asthma and allergy – Cilla Söderhäll's research group
- Cardiovascular epidemiology – Team Bruna Gigante
- Neurobiology of Stress and Treatment Response – Juan Pablo Lopez group
- Vascular Surgery – Ulf Hedin's research group
- Annika Bergquist group
- Acute myeloid leukemia – Sören Lehmann group
- Human tissue-resident NK cells – Niklas Björkström group
- Neuropsychoimmunology – Sophie Erhardt's research group
- Diabetes epidemiology – Sofia Carlsson's research group
- Stroke - Acute Intervention and Secondary Prevention – Niaz Ahmed's research group
- Molecular muscle physiology and pathophysiology – Lanner Lab
- Community nutrition and physical activity (CoNPA) – Liselotte Schäfer Elinder's research group
- Inflammatory bowel disease (IBD) – Research group Eduardo Villablanca
- CRISPR-based drug target discovery in cancer and autoimmunity – Research group Fredrik Wermeling
- Emergency care – Kristian Ängeby's research group
- Clinical Cancer Epidemiology – Research group Karin Ekström Smedby
- Cancer prevention and screening – Johannes Blom's research group
- Somatosensation – Patrik Ernfors group
- DNA replication & Cancer Genetics – Lemmens Group
- Laboratory testing for alcohol and drugs of abuse – Anders Helander research
- Anaesthesia and Intensive care – Rebecka Rubenson Wahlin/Anna Schandl's research group
- Jonas Fuxe Group
- SCF ubiquitin ligases, cell cycle, transcription and cancer development – Olle Sangfelt's Group
- Toxicological Mechanisms – Emma Wincent's research group
- Stem Cells in Tissue Homeostasis and Regenerative Medicine – Jonas Frisén's Group
- Urology – Anna Lantz's research group
- The New World of Work – Theo Bodin's research group
- Midbrain dopaminergic neuron development
- Research group Michael Fored
- Neurobiology of pain & Therapeutics – Saida Hadjab Group
- Risk assessment – Mattias Öberg's research group
- Research groups in cell and molecular biology
- Developmental Psychology: Digital Media and ADHD – Lisa Thorell's research group
- Research groups in education and pedagogy
- Research groups in biostatistics and probability theory
- ESSI – Emotion regulation, Self-injury, Suicide, and Intervention – Johan Bjureberg's research group
- Medical ethics – Gert Helgesson's group
- Treatment of substance use disorders – Johan Franck's Research Group
- Sex hormones and sex differences in diseases of the brain – Ivan Nalvarte's research group
- Cilia in the brain – and their connections to human brain disorders – Peter Swoboda's research group
- Evidence-based methods in autism and ADHD – Tatja Hirvikoski's research group
- Long-Term Outcomes after Perioperative and Intensive Care – Max Bell research group
- Translational and clinical research in acute myeloid leukemia – Team Martin Jädersten
- CEREBRA – Susanne Palmcrantz's research group
- Prognostication, and early detection of recurrence, in upper tract urothelial carcinoma (UTUC) – Marianne Brehmer research group
- Loh
- Surgery and Urology at KI DS
- Protein expression, stability, and redox regulation – Saei Lab
- Translational Pharmacology – Kent Jardemark's team
- Global and Sexual Health (GloSH) – Anna Mia Ekström's research group
- Neuro-infections & Neuroinflammation – Federico Iovino Group
- Clinical Chemistry and Blood Coagulation – Jovan Antovic's research group
- Translational research on human microbial infections and its consequences – Anders Sönnerborg's group
- Human tissue-resident NK cells in homeostasis and disease – Nicole Marquardt team
- Etiology and pathogenesis of type 1 diabetes – Malin Flodström-Tullberg group
- Oxygen sensing, cancer and intratumor heterogeneity – Schlisio Lab
- Genetic Epidemiology of Neuroinflammatory Disorders – Ingrid Kockum's research group
- Retina – Anders Kvanta's research group
- Glaucoma – Pete Williams' research group
- Leadership in healthcare and academia – Mia von Knorring's group
- Luminal gastroenterology – Research group Charlotte Hedin
- Identifying molecular signals in the genital mucosa that determine susceptibility to sexually transmitted infections – Research group Kristina Broliden
- Coronary heart disease – Research group Per Svensson
- Educational development – Matti Nikkola's group
- Post-transcriptional regulation in mitochondria – Joanna Rorbach group
- Cellular diversity is generated during development, neuronal identity – Johan Holmberg's Group
- Lipid handling in health and cardiometabolic disease – Research group Carolina Hagberg
- Renal glomerulus biology and diseases – Jaakko Patrakka group
- Research group Caroline Palm Apergi
- Precision pathology and tumor heterogeneity – Johan Hartman's Group
- Lifelong Learning in Health Care Contexts – Terese Stenfors' group
- Translational Arthritis Research – Team Bence Réthi
- Stem cells, Developmental Biology, Heart Disease, Heart Development, Genetics, Biotechnology – Kenneth R. Chien's group
- Urological cancer – Lars Egevad's Group
- The ubiquitin/proteasome system in neurodegenerative diseases and cancer – Nico Dantuma's Group
- Experimental alcohol- and drug dependence research – Vladana Vukojević research group
- Chronic inflammatory disease epidemiology – Research group Johan Askling
- Statistical and bioinformatics analyses of high-throughput molecular data – Yudi Pawitan's research group
- Urban environment and children´s and youth health – Olena Gruzieva's research group
- Research groups in laboratory medicine
- Research groups in medical biotechnology
- Research groups in physiotherapy
- Breast cancer epidemiology – Kamila Czene's research group
- Research groups in human computer interaction
- Research group Liv Eidsmo
- Preventive Medicine – Susanna Larsson's research group
- Transition to adulthood for individuals with neurodevelopmental conditions – Ulf Jonsson's team
- Function and Health in Respiratory and Cardiovascular Conditions – Malin Nygren-Bonnier's research group
- Laboratory of lymphocyte biology – Team Taras Kreslavskiy
- Global Reproductive Outreach & Wellness (GROW) – Michael Wells
- Mats Marshall Heyman research group
- SLE/APS/Vasculitis group – Research group Aleksandra Antovic
- Speech-Language Pathology – Liza Bergström's research group
- Arrhythmia – Johan Engdahl's research group
- Research at Ersta Hospital – Anders Thorell's group
- Surgical Nursing Research, Innovation and Surgical Education, SuN-Rise – Ami Fagerdahl's research group
- Tissue immunosurveillance by cytotoxic lymphocytes – Takuya Sekine team
- Physical activity and Sports medicine with focus on prevention – Hagströmer research group
- Receptor biology and signaling – Schulte Lab
- Neurobiology of Obesity – Alessandro Furlan group
- Upper GI Surgery – Jesper Lagergren's research group
- Sepsis and COVID-19 – Kristoffer Strålin Group
- Harnessing Neutrophils for Precision Cell Therapy against solid tumors – Roland Fiskesund Team
- Human T cell immunity to evolving viruses and cancers – Marcus Buggert group
- Neuroradiology - MRI physics – Stefan Skare's research group
- Wilhelm lab
- Inflammatory responses in cancer and autoimmunity – Mikael Karlsson's Group
- Working life, ergonomics, psychosocial factors, and health – Research group Daniel Falkstedt
- Aging and health – Welmer's research group
- NK Cell Recognition – Klas Kärre Group
- Computational Breast Imaging – Fredrik Strand's Group
- Drug treatment – Erik Eliasson research group
- Neural differentiation as a strategy for neuroblastoma treatment – Marie Arsenian Henriksson Group
- Cancer immunotherapy – Mattias Carlsten group
- Cell-based immune therapy for cancer – Andreas Lundqvist's Group
- Basic molecular structure of human skin – Lars Norlén's research
- Reproductive, Perinatal and Pediatric Epidemiology – Research group Olof Stephansson
- Research group Erik Melén
- Research group Joel Nordin
- MINT – Klas Karlgren's team
- Translational breast cancer research – Theodoros Foukakis' Group
- Genes function and molecular bases of diseases – Research group Magdalena Paolino
- Homeostasis and tissue repair mechanisms in the mammalian system, fibroblasts, stem cells – Christian Göritz Research Group
- The Perinatal Epidemiology Lifecourse Lab – Team Neda Razaz
- Experimental Cancer Medicine (ECM)
- Research group Therese Djärv
- Molecular and cellular neuroendocrinology – Tibor Harkany group
- Neuropsychiatric disorders – Håkan Karlsson group
- Research groups in dermatology and venereal diseases
- Evidence-based practice: prevention, intervention and implementation – Pia Enebrink's research group
- Research groups in radiology and medical imaging
- Research groups in psychiatry
- Maria Eriksdotter research group
- Research groups in precision medicine
- Breast cancer surgery – Jana de Boniface's research group
- ICare – Ann Langius-Eklöf's research group
- Immunology and Chronic Disease – Johan Frostegård's research group
- Health in Everyday Life (HELD) – Susanne Guidetti's research group
- Center for Resuscitation Science – Jacob Hollenberg's research group
- Applied Developmental Neurobiology – Carl Sellgren research group
- Reproductive Health/Reproductive Medicine – Kristina Gemzell Danielsson's research group
- Malin Holzmann's research group
- Radiation therapy/ Radiation Oncology – Pehr Lind Group
- Ute Römling group
- Perioperative quality – Jan Jakobssons research group
- Medical Education and Implementation – Anna Kiessling's research group
- Particle toxicology – Anda Gliga's research group
- ALGOSH: Algorithmic Management at Work
- Development of autonomic control – Herlenius Research
- Maintenance and expression of mtDNA in disease and ageing – Nils-Göran Larsson Group
- Neuropharmacology - Movement Disorders – Per Svenningsson's research group
- Somatosensation & Gargalesis – Konstantina Kilteni group
- Signal Transduction – Ismael Valladolid Acebes' research group
- Microbiome and Infections – Piotr Nowak Research Group
- Immunological tolerance and transfusion immunology – Petter Höglund group
- A precision medicine framework for tailored host‑directed therapies in tuberculosis – Susanna Brighenti group
- Pain and Brain Imaging – Karin Jensen's research group
- Susanne Nylén group
- Hand surgery – Maria Wilckes Research group
- Environmental impact on pulmonary host defence and chronic airflow obstruction – Anders Lindén's research group
- Rehabilitation, collaboration and aging – Elisabeth Rydwik's research group
- Tuberculosis Aerobiology – Antonio Rothfuchs group
- Development of new molecules of biomedical interest through cell-based phenotypic screens – Oscar Fernandez-Capetillo group
- Diversity within the T cell response, T cell memory – Research group Carmen Gerlach
- Clinical and translational melanoma research – Hanna Eriksson's Group
- Meiotic chromosome segregation – Christer Höög's research group
- B cells and autoantibodies in rheumatic disease – Team Caroline Grönwall
- Precision medicine in lymphoid malignancies including CLL; from new targets to real-world assessments – Anders Österborg's Group
- Molecular cancer research – Anthony Wright
- Genome integrity and nucleolar stress in cancer – Jiri Bartek group
- Translational Cardiology (@TransCardio) – Research group Magnus Bäck
- Gene regulation, genome-wide experimental and computational techniques – Rickard Sandberg's Group
- The Helleday Laboratory focuses on harnessing defects in the DNA damage response and metabolism to develop novel therapies
- Small RNAs in cancer development – Weng-Onn Lui's Team
- Geriatric pharmacoepidemiology – Kristina Johnell's research group
- Molecular neurodevelopment and neuro-oncology – Ola Hermanson group
- Research groups in anesthesiology and intensive care
- Gastrointestinal epidemiology – Jonas Ludvigsson's research group
- Health inequalities and minority stress – Richard Bränström's research group
- Research groups in microbiology
- Research groups in orthopaedics
- Hereditary hematological malignancies – Bianca Tesi team
- Mechanisms of establishment of blood vessels – Lars Holmgren's Group
- Substance use prevention – Johanna Gripenberg's research group
- Perioperative care – Ulrica Nilsson's research group
- Mechanisms behind HIV-1 latency and rebound – Peter Svensson's group
- ENGAGE- Enacting health and change through everyday activity – Patomella group
- Diagnosis and treatment of children with asthma and allergy: from molecular diagnosis to intervention – Jon Konradsen's research group
- Translational research in childhood cancer and histiocytic diseases – Nikolas Herold Research group
- Coronary artery disease – research at Danderyd hospital
- Mechanisms of malignant hematopoiesis – Pedro Moura team
- Itziar Martinez Gonzalez group
- CoNFIND Cognition and Fatigue – Marika Möllers research group
- Venous thromboembolism and emergency medicine – Eli Westerlund's research group
- Reproductive endocrinology and metabolism – Elisabet Stener-Victorin's research group
- Tissue engineering – Magdalena Fossum team
- Inflammation and metabolism – Nicolas Pillon's team
- Neurobiology of Sensory Systems – François Lallemend group
- Experimental Traumatology Research Unit
- Genetic Modification of NK cells for Optimized Functions against Cancer – Arnika Wagner team
- NK cells in the development of adaptive immune responses – Benedict Chambers team
- Molecular pain research – Camilla Svensson's research group
- Brain Connectomics – Joana Pereira's research group
- Genomic Science and RNA Biology – Vicent Pelechano group
- Immunotherapy – Robert Harris' research group
- Infectious diseases "Venhälsan" – Jaran Eriksen's research group
- Hjerling-Leffler group
- Respiratory and invasive infections and microbial pathogenesis – Birgitta Henriques-Normark Laboratory
- Multimodal Brain Imaging – Daniel Lundqvist's research group
- Obsessive - Compulsive and Related Disorders Across the Lifespan – David Mataix-Cols research group
- With a focus on proteins for better cancer treatments – Pär Nordlund's Group
- Precision cancer medicine and precision radiotherapy in lung cancer - from molecular mechanisms/biomarkers to clinical trials – Kristina Viktorsson's team
- Cancer Bioinformatics – Nick Tobin's Team
- Lipoproteins and the Immune System – Research group Stephen Malin
- Equity and Health Policy (EHP) – Ann Liljas' research group
- Effects of hypoxia in physiological and pathological contexts, Hypoxia Inducible Factors (HIF) – Randall Johnson's Group
- Chemistry II – Jesper Z. Haeggström group
- Genomic analysis of parasites and viruses, metagenomic sequencing – Björn Andersson's Group
- Translational control of cancer – Ola Larsson's Group
- Gastrointestinal Surgery KI SÖS – Gabriel Sandblom's research group
- Understanding the mechanisms behind hantavirus-mediated pathogenesis – Jonas Klingström group
- The Gynaecological research team – Elisabeth Epstein
- Understanding how life develops – Emma Andersson's Group
- Statistical methods in epidemiology – Paul Dickman's research group
- Neurons and Neural Networks – Ole Kiehn group
- Research groups in developmental biology
- Behavioral, Resilience and Digital Health in Pain – Rikard Wicksell’s research group
- Research groups in neurology
- Research groups in epidemiology, global public health and social medicine
- Nordic Brain Network – Miia Kivipelto's research group
- Resilience and mental health – Serhiy Dekhtyar group
- Forensic behavioral and neuroscience – Katarina Howner's Research Group
- Immune Engineering – Team Leo Hanke
- Chemical and physical exposure in the work environment and health – Jenny Selander's research group
- Mechanobiology of cardiac regeneration – Elif Eroglu's Group
- Tumors of the female genital organs – Miriam Mints' research group
- Psychoneuroimmunology – Mats Lekander's research group
- Upper gastrointestinal surgical – research at Danderyd Hospital
- Research groups in odontology
- Neuroimmunovascular biology research – Harald Lund's research group
- Bacteriophages – Andrea Fossati's research group
- Division of Neurogeriatrics – Sophia Schedin Weiss's research group
- Rehabilitation medicine at KI DS
- Karin Flood team
Molecular pain research – Camilla Svensson's research group
Chronic pain is a major health problem affecting about 20 per cent of the Swedish population, resulting in markedly reduced quality of life for the individual.

 Photo: Image by Konstantin Kolosov from Pixabay
Photo: Image by Konstantin Kolosov from PixabayMake a donation to our research
Your support means a lot to the success of research. This allows us to go further in our efforts to improve human health through research and education.
For example, you can make a donation via Swish.
Our research
Nearly 1 in 5 adults suffer from chronic pain. Drug development has hitherto been insufficient and there are currently few effective treatments for persistent pain. Without adequate relief, individuals with persistent pain often endure physical and psychosocial problems resulting in a dramatically reduced quality of life. Pain is not only devastating for the individual but also imparts large societal and economic costs in the form of medical care, sick leave and lost productivity. Thus, it is critical to increase our understanding of how chronic pain is regulated, in order to identify new treatment strategies and novel drug targets for pain relief.
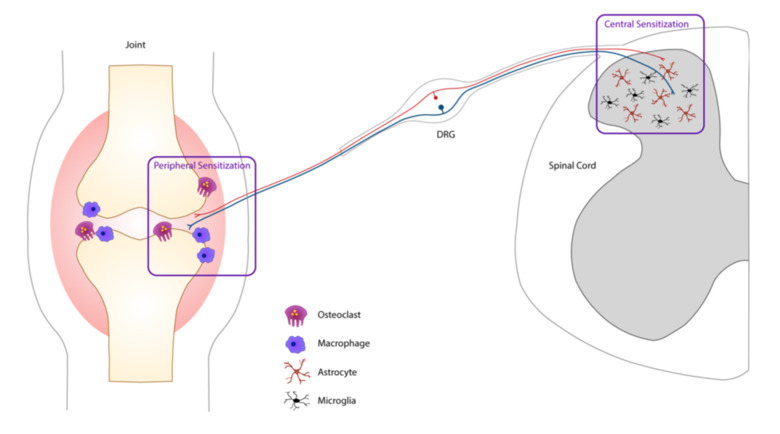
Pain is one of the most problematic symptoms of rheumatic diseases, such as rheumatoid arthritis (RA) osteoarthritis (OA) and fibromyalgia (FM). RA is an autoimmune disease characterized by joint inflammation along with bone and cartilage destruction that affects around 1% of the population. In RA, current anti-inflammatory treatments effectively control joint-inflammation (and inflammatory pain) in many patients. Unfortunately, well-controlled joint inflammation does not mean pain relief. RA patients frequently present with debilitating pain prior to joint-inflammation, which often persists even when the disease is medically controlled or in remission. Effective analgesic therapies for the “non-inflammatory” pain are lacking. We identified that several RA-associated antibodies, isolated from RA patients, induce pain-like behavior without classical signs of inflammation when transferred to mice.
FM is a highly prevalent disease affecting 2-4% of the population, which predominantly affects women (7/10). Hallmarks of FM include widespread musculoskeletal pain, stiffness, and physical/cognitive fatigue. There is growing evidence of central nervous system (CNS) disturbances in FM, however, our lack of understanding the underlying pathophysiology has prevented development of effective therapies. Further, available treatments are often ineffective and frequently associated with adverse effects. FM is currently not considered an autoimmune disease, but we have discovered that transfer of antibodies (immunoglobulin G, IgG) purified from FM patients’ blood to mice gives rise to an increased sensitivity to deep pressure and cold and a reduced activity without causing an overt tissue inflammation.
The insight that autoantibodies may generate pain in the absence of visible inflammation led us to the hypothesis that not only in RA, but also in FM, there are undiscovered links between antibodies and chronic pain. Several projects in the lab are centered on investigating mechanisms by which antibodies contribute to long-lasting pain, with a focus on the molecular and cellular processes responsible for pronociceptive properties of antibodies, and the consequences of their actions.
Publications
Selected publications
- Article: BRAIN BEHAVIOR AND IMMUNITY. 2019;75:60-71Palada V; Ahmed AS; Finn A; Berg S; Svensson CI; Kosek E
- Article: PAIN. 2019;160(1):224-236Fernandez-Zafra T; Gao T; Jurczak A; Sandor K; Hore Z; Agalave NM; Su J; Estelius J; Lampa J; Hokfelt T; Wiesenfeld-Hallin Z; Xu X; Denk F; Svensson CI
- Article: NEUROPEPTIDES. 2018;69:53-59Sandor K; Krishnan S; Agalave NM; Krock E; Salcido JV; Fernandez-Zafra T; Khoonsari PE; Svensson CI; Kultima K
- Editorial: NEURON. 2018;97(4):729-731Hunt MA; Nascimento DSM; Farinotti AB; Svensson CI
- Review: JOURNAL OF COMPARATIVE NEUROLOGY. 2015;523(10):1505-1528Su J; Gao T; Shi T; Xiang Q; Xu X; Wiesenfeld-Hallin Z; Hokfelt T; Svensson CI
- Article: PAIN. 2014;155(9):1802-1813Agalave NM; Larsson M; Abdelmoaty S; Su J; Baharpoor A; Lundback P; Palmblad K; Andersson U; Harris H; Svensson CI
- Article: ARTHRITIS & RHEUMATOLOGY. 2012;64(12):3886-3896Bas DB; Su J; Sandor K; Agalave NM; Lundberg J; Codeluppi S; Baharpoor A; Nandakumar KS; Holmdahl R; Svensson CI
- Book chapter: METHODS IN MOLECULAR BIOLOGY. 2012;p. 249-260Christianson CA; Corr M; Yaksh TL; Svensson CI
Full list of publications
Commentaries about our work
https://www.jci.org/articles/view/150382
https://news.ki.se/autoantibodies-a-possible-contributor-to-fibromyalgia
https://www.verywellhealth.com/autoimmunity-neuroinflammation-in-fibromyalgia-5197944
https://www.painresearchforum.org/news/180579-antibodies-patients-cause-fibromyalgia-symptoms-mice
https://www.sciencealert.com/mouse-study-suggests-fibromyalgia-really-is-an-autoimmune-disorder
https://www.expressen.se/premium/halsa/nya-studien-ger-hopp-for-personer-med-fibromyalgi/
https://www.aftonbladet.se/halsa/a/Ryzmer/nya-ron-ger-hopp-at-drabbade-av-fibromyalgi
https://www.nature.com/articles/s41584-019-0269-8
https://news.ki.se/rheumatoid-arthritic-pain-could-be-caused-by-antibodies
Funding
Selected awards and funding
2020 UCSD Pharmacology Department Annual Outstanding Alumnus Award
2019 ERC Consolidator grant ANTIBODYPAIN
2019 Wallenberg Academy Scholar, Knut and Alice Wallenberg Foundation
2018 Wallenberg Academy Fellow prolongation, Knut and Alice Wallenberg Foundation
2012 Wallenberg Academy Fellow, Knut and Alice Wallenberg Foundation
2012 Söderberg Fellow in Medicine, Ragnar Söderberg Foundation
2010 Future Research Leader 4, Swedish Foundation for Strategic Research
2009 Early Career Award, International Association for Studies of Pain
2006 McDuffie Honor Award, American Arthritis Foundation
2021 FOREUM “Pain and Fatigue”
2020 FOREUM “Sex and gender issues”
2020 EIC-FETPROACT: BoneFix (lead by Michael Malkoch, KTH)
2019 EU-ITN: TOBeATPAIN (lead by Marcia Malcangio, KCL)
2019 SSF E-NeuroPharma (lead by Magnus Berggren, Linköping University)
2018 EU-ITN: BONEPAIN.II (lead by Anne-Marie Heegaard, Copenhagen University)
2014 EU-ITN: BONEPAIN (lead by Anne-Marie Heegaard, Copenhagen University)
2013 Vetenskapsrådet Bidrag till framstående yngre forskare
Staff and contact
Group leader
All members of the group
- Kirill AgashkovPostdoctoral Researcher
- Sven ArvidssonPhd Student
- Alex Bersellini FarinottiResearch Specialist
- Cajsa ClassonPostdoctoral Researcher
- Matthew Adam HuntPostdoctoral Researcher
- Emilie LinderothLaboratory Coordinator
- Andrea Lovincic BabicPhd Student
- Khosiyat MakhmudovaPhd Student
- Daniele OlmedaResearch Assistant
- Mohd Sami Ur RasheedPostdoctoral Studies
- Katalin SandorSenior Research Specialist
- Macarena Tejos BravoAffiliated to Research
- Gustaf WängbergBioinformatician
- Sijing YeAffiliated to Research
- Zhening ZhangPhd Student
- Qianzi ZhouResearch Assistant

Previous group members
- Sara Sadek - Bachelor student
- Merna Bitar - Master student
- Nicole Nova - Master student
- Max Larsson - Researcher
- Kristina Ängeby-Möller - Doctoral student
- Teresa Fernandez-Zafra - Doctoral student
- Payam Emami - Postdoc
- Shibu Krishnan - Postdoc
- Tianle Gao - Postdoc
- Anahita Morbagha - Bachelor student
- Fatima Rhordo - Master student
- Christina Christianson - Visiting PhD student
- Johanna Pettersson - Master student
- Joshua Gregory - Postdoc
- Pia Sandberg - Bachelor Student
- Hanna Steurer - Bachelor Student
- Ebba Gregory - Doctoral student
- William Nyberg - Master student
- Simone Codeluppi - Asisstant professor
- Magdalena Kegel - Master student
- Kim Kultima - Associated
- Ada Delaney - Postdoc
- Tianle Gao - Postdoc
- Camilla Ultenius - Postdoc
- Anya Finn - Doctoral student
- Jungo kato - Postdoc
- Jie Su - Doctoral student
- Katarzyna Rogoz - Postdoc
- Duygu Belkis Baş - Postdoc
- Gustaf Wigerblad - Doctoral student
- Sally AbdelMoaty - Postdoc
- Ahmad Shareef - Research assistant
- Jaira Villarreal Salcido - Master student
- Diana Nascimento - Postdoc
- Nilesh Agalave - Postdoc (previously PhD student)
- Azar Baharpoor - Biomedical scientist
- Yuki Nomura - Asisstant professor
- Vinko Palada - Postdoc
- Tinna Ängeby Möller - PhD Student
- Freija ter Heegde - Postdoc
- Louisa Brieskorn - Master student
- Francisca Castillo - Research assistant
- Resti Rudjito - PhD student
- Femke de Krom - Master student
- Chrisitan Martin Gil - Visiting pPhD student
- Ivan Voloshyn - Research assistant
- Zhenggang Wang - Visiting PhD student
- Emerson Krok - Postdoc
- Freja Holländer - Masterstudent
- Paul Benjamin - Masterstudent
- Juan Antonio Vazquez Mora - Masterstudent
- Julia Döring - Masterstudent
- Sigita Venckute - Master student and Laboratory assistant
- Federico Picciau - Intern
- Carlos Eduardo Morado Urbina - PhD student
- Annelie Lifvergren - Bachelor student
- Elena PDurán González - Visiting PhD student
- Linda Kaupp - Master Student
- Lydia Moll - PhD Student
- Joana Menezes - PhD student
- Alexandra Kuliszkiewicz - Research infrastructure specialist
- Nils Simon - PhD student
Group activities
Research retreats and conferences


Our projects
The research in our laboratory is centered on mechanisms that regulate pain signaling, with a particular focus on exploring the role of autoantibodies, nerve growth factor (NGF) and pro/anti inflammatory factors in pain signal transmission in both peripheral and central nervous systems. We are currently using several experimental pain models and clinical data/tissues from patient cohorts with the aim to pin point novel targets for the development of new pain therapeutics.
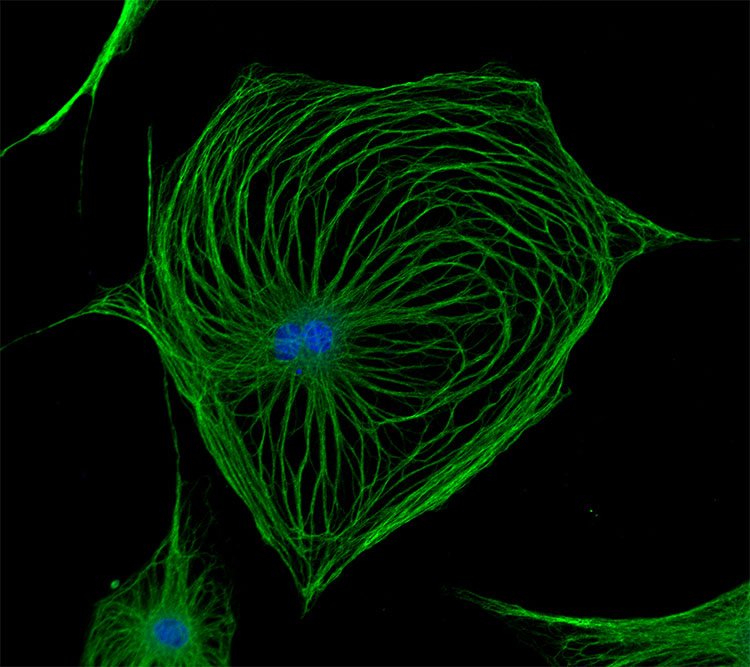
Fibromyalgia
Fibromyalgia is a common condition that affects 2-4% of the population and predominantly affects women. Fibromyalgia is characterized by chronic widespread pain, hypersensitivity to stimuli like touch, light and sound, chronic fatigue, and cognitive difficulties. Despite the widespread prevalence, diagnostic tests do not exist, and pain management strategies have little efficacy. In turn, individuals with fibromyalgia have reduced quality of life.
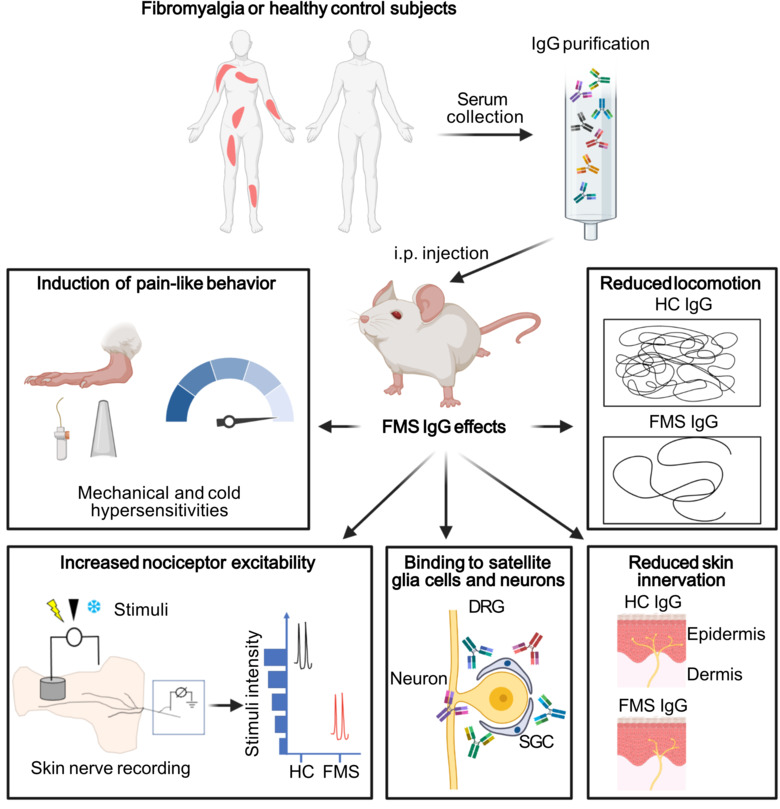
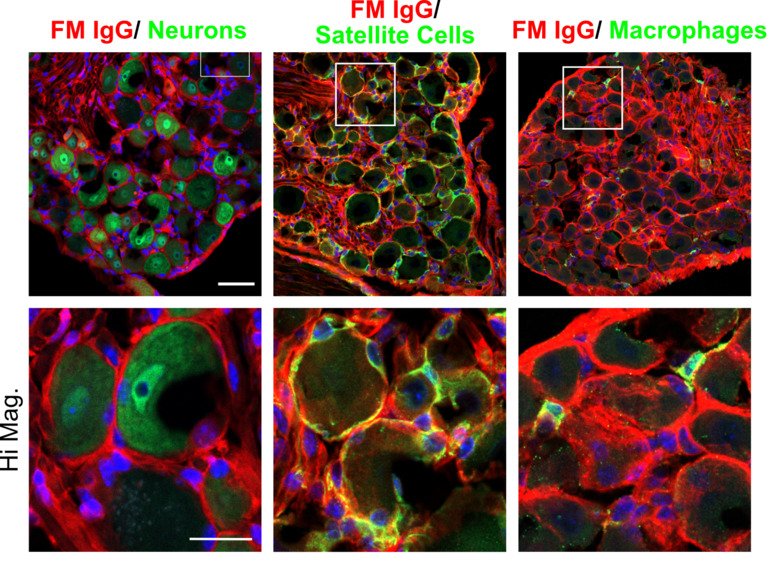
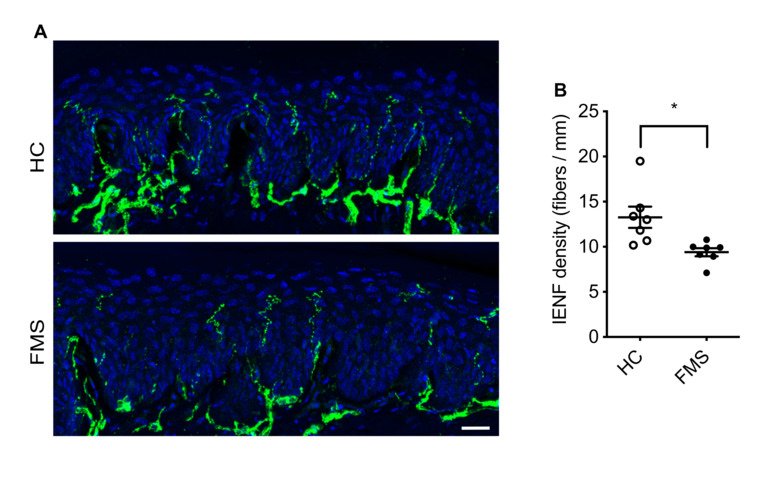
Our group aims to change this by identifying new therapeutic strategies and developing diagnostic tests for fibromyalgia. We recently found that antibodies from individuals with fibromyalgia induce pain-like behavior in mice (increased sensitivity to mechanical stimulation and cold) and reduce their locomotor activity, while antibodies from healthy individuals do not alter the behavior of the mice. We discovered that the fibromyalgia antibodies accumulate in the dorsal root ganglia and bind to the satellite glia cells that surround the cell bodies of sensory neurons. We also identified that mice with FM antibodies have reduced innervation of the skin. We are currently investigating how the interaction between FM patient derived antibodies and satellite glial cells results in increased excitability of pain fibers and reduced innervation of the skin.
Taken together our discoveries suggest that autoantibodies are likely an important component of fibromyalgia that has yet to be fully explored. We are now investigating cellular and molecular aspects of how fibromyalgia autoantibodies cause pain, changes in activity and innervation. We are also trying to identify what antigen the fibromyalgia autoantibodies are binding to. We hope that our work will lead the way to new pain management strategies for fibromyalgia and that by identifying what the autoantibodies bind, we will be able to develop antibody based diagnostic tests for fibromyalgia.
For more information:
Passive transfer of fibromyalgia symptoms from patients to mice.
Goebel A, Krock E, Gentry C, Israel MR, Jurczak A, Urbina CM, Sandor K, Vastani N, Maurer M, Cuhadar U, Sensi S, Nomura Y, Menezes J, Baharpoor A, Brieskorn L, Sandström A, Tour J, Kadetoff D, Haglund L, Kosek E, Bevan S, Svensson CI, Andersson DA
J Clin Invest 2021 07;131(13)
Autoantibody-mediated pain mechanism
Anti–modified protein antibodies (AMPAs)
Antibodies binding citrullinated epitopes of e.g. vimentin, alpha-enolase and fibrinogen (so called anti-citrullinated protein antibodies, ACPA) are the most disease specific RA autoantibodies, present in 70% of the patients. In the past, autoantibodies in RA have predominantly been coupled to pain as an intermediary factor during inflammation, which enhance activation of immune cells that subsequently release pain-inducing factors such as cytokines and prostaglandins. In sharp contrast, data from our laboratory demonstrated for the first time that injection of total IgG purified from RA patients, or isolated ACPA IgG, mediate long-lasting pain-like behavior in mice without causing classical signs of inflammation.
Prior to their clinical diagnosis many RA patients experience pain prior to signs of joint inflammation. Similarly, ACPA and additional members of the anti-modified protein antibodies (AMPAs, which show reactivity towards e.g. carbamylation, acetylation or malondialdehyde) family can be present in the blood for many years before clinical manifestation and diagnosis. We aim to better understand how AMPAs and other RA-associated antibodies drive pain in the absence of overt joint inflammation. We have found that osteoclasts play an important role; blocking their activation in mice injected with ACPA and other RA-associated antibodies prevents the development of pain-related behaviors. This reveals a fascinating interplay between the immune system, the nervous system and bone homeostasis. We are also currently exploring how different cell types such as osteoblasts, fibroblasts, macrophages, satellite glia cells, CNS microglia and astrocytes contribute to AMPA-mediated pain.
For more information:
Autoantibodies to citrullinated proteins induce joint pain independent of inflammation via a chemokine-dependent mechanism.
Wigerblad G, Bas DB, Fernades-Cerqueira C, Krishnamurthy A, Nandakumar KS, Rogoz K, Kato J, Sandor K, Su J, Jimenez-Andrade JM, Finn A, Bersellini Farinotti A, Amara K, Lundberg K, Holmdahl R, Jakobsson PJ, Malmström V, Catrina AI, Klareskog L, Svensson CI
Ann Rheum Dis 2016 Apr;75(4):730-8
Antibody-induced pain-like behavior and bone erosion: links to subclinical inflammation, osteoclast activity, and acid-sensing ion channel 3-dependent sensitization.
Jurczak A, Delay L, Barbier J, Simon N, Krock E, Sandor K, Agalave NM, Rudjito R, Wigerblad G, Rogóż K, Briat A, Miot-Noirault E, Martinez-Martinez A, Brömme D, Grönwall C, Malmström V, Klareskog L, Khoury S, Ferreira T, Labrum B, Deval E, Jiménez-Andrade JM, Marchand F, Svensson CI
Pain 2021 Nov;():
Pathogenic Citrulline-Multispecific B Cell Receptor Clades in Rheumatoid Arthritis.
Titcombe PJ, Wigerblad G, Sippl N, Zhang N, Shmagel AK, Sahlström P, Zhang Y, Barsness LO, Ghodke-Puranik Y, Baharpoor A, Hansson M, Israelsson L, Skriner K, Niewold TB, Klareskog L, Svensson CI, Amara K, Malmström V, Mueller DL
Arthritis Rheumatol 2018 12;70(12):1933-1945
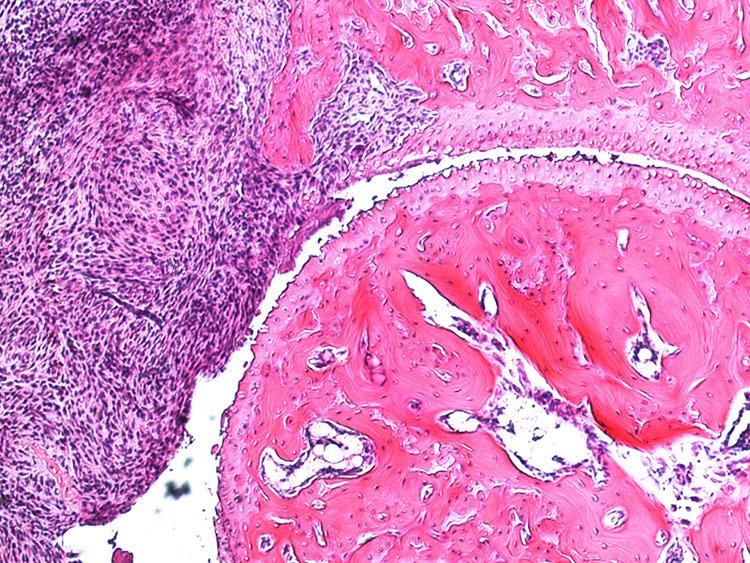
Collagen antibody-induced arthritis (CAIA)
In the collagen antibody-induced arthritis (CAIA) model mice injected with collagen type II antibodies exhibit joint inflammation and other pathologies similar to those experienced by patients with rheumatoid arthritis. This model was based on the presence of anti-collagen type II antibodies in patients with RA and has become commonly used in studies aimed at examining the pathology of rheumatoid arthritis. Prior to our work, however, this model had not been used for assessment of arthritis-induced pain (nociception). We found that in this model mice developed mechanical hypersensitivity prior to any signs of joint inflammation, which persisted for several weeks after the resolution of joint inflammation. This mimics the onset of pain in RA patients as well as its persistence in individuals whose other symptoms are well controlled. Using behavioral, electrophysiological, molecular and imaging techniques along with transgenic animals, we are currently focusing on studying the mechanisms underlying pain prior to RA onset and pain that persists during phases with low disease activity.
Hypersensitivity prior to joint inflammation (early phase CAIA)
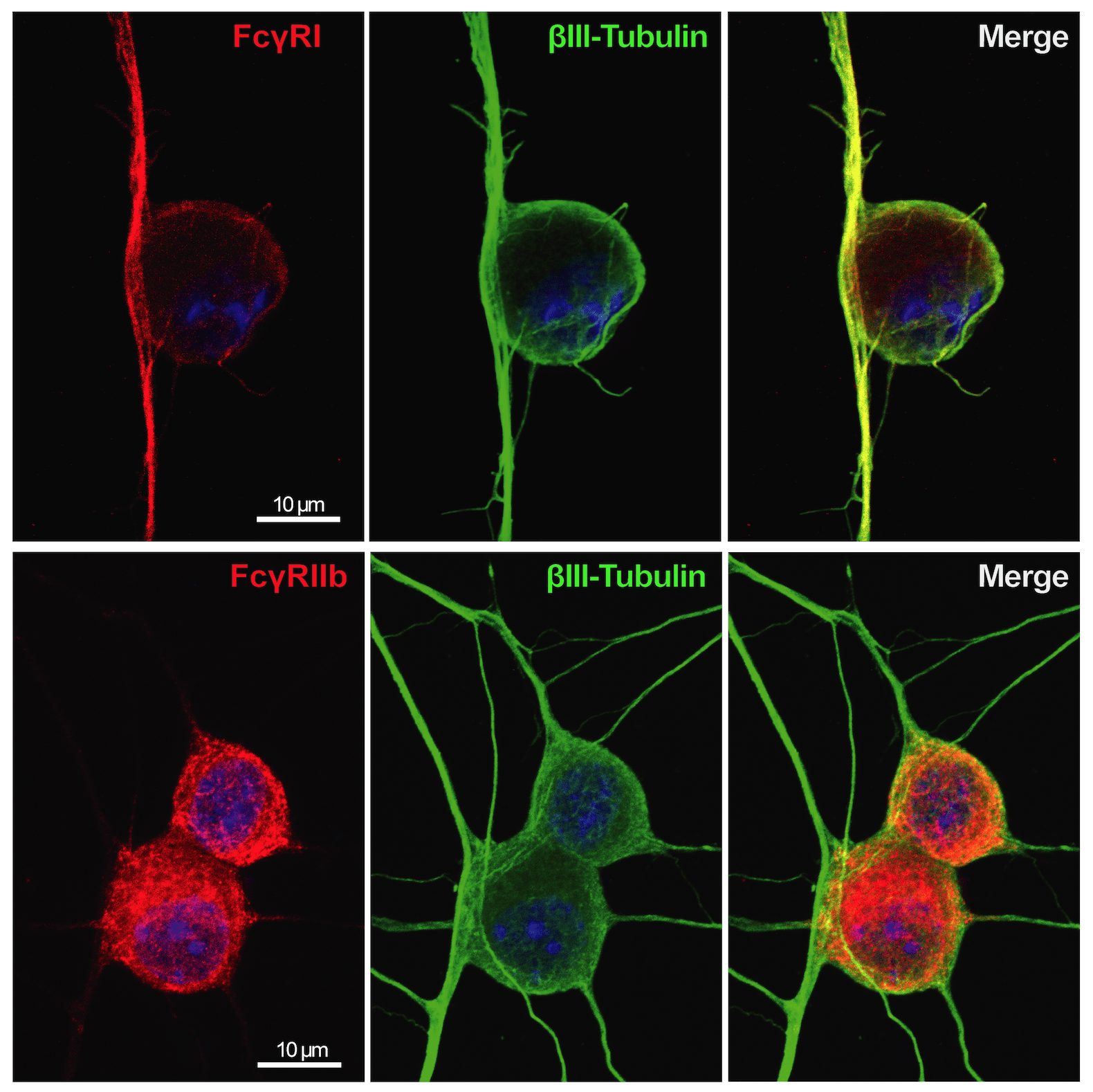
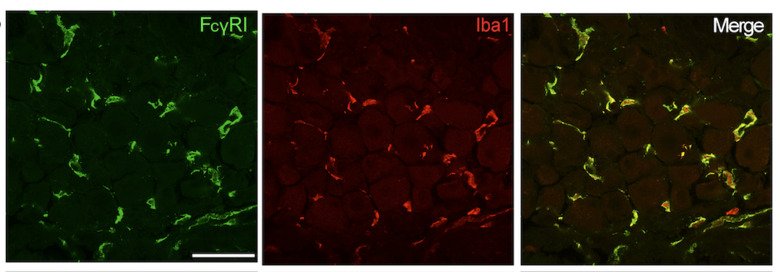
Immune complex (IC) is composed of aggregates of antibodies bound to their soluble antigens. ICs regulate inflammatory processes by activation of Fc receptors (FcR) expressed on various inflammatory cells and the complement system. Importantly, we and others have shown that sensory neurons express FcγRI and FcγRIIb and that IgG in IC formation can directly activate nociceptors, independent of inflammatory cells and the complement system. Our studies show that the FcγRI is responsible for the early pain-like behavior we see in the CAIA model. We are currently investigating the role of FcγRs expressed on DRG cells as an additional novel pain mechanism in RA (and other autoimmune diseases) in which autoantibodies may act as pain-inducing molecules independent of the inflammatory process during active disease. This project has also a strong translational potential as we discovered the presence of FcγRIII on human adult DRGs. This could lead to novel therapeutic strategies for pain not only for RA, but also for other autoimmune diseases where there are accumulation of IgG and formation of immune complexes in innervated tissues, such as the DRG.
For more information:
Cartilage-binding antibodies induce pain through immune complex-mediated activation of neurons.
Bersellini Farinotti A, Wigerblad G, Nascimento D, Bas DB, Morado Urbina C, Nandakumar KS, Sandor K, Xu B, Abdelmoaty S, Hunt MA, Ängeby Möller K, Baharpoor A, Sinclair J, Jardemark K, Lanner JT, Khmaladze I, Borm LE, Zhang L, Wermeling F, Cragg MS, Lengqvist J, Chabot-Doré AJ, Diatchenko L, Belfer I, Collin M, Kultima K, Heyman B, Jimenez-Andrade JM, Codeluppi S, Holmdahl R, Svensson CI
J Exp Med 2019 08;216(8):1904-1924
Chronic hypersensitivity (inflammatory and late phases of CAIA model)
We have previously identified several potential factors that could drive persistent pain-like behavior in the CAIA model, such as high mobility group box 1 (HMGB1) and lysophosphatidic acid (LPA).
HMGB1 is an important molecule in the pathogenesis of RA. We have reported the involvement of both peripheral and spinal HMGB1 in CAIA-induced pain-like behavior in both inflammatory and late phases. Interestingly, the pronociceptive role of HMGB1 is redox- and sex-dependent, thus highlighting the importance of these two parameters when developing novel therapeutics.
LPA is a bioactive phospholipid that had been linked to neuropathic pain in animal models and to joint inflammation in RA patients and RA models. We found that blocking LPA signaling reverses CAIA-induced pain-like behavior during both the inflammatory and late phases, independent of joint inflammation. We determined that LPA signaling between satellite glia cells (SGCs), the resident DRG glia, and neurons can regulate pain-like behavior. These findings highlight the importance of local signaling events within the DRG itself and that LPA and SGCs are potential antinociceptive targets in persistent RA-associated pain.
Currently, we are investigating if changes in bone metabolism contribute to long-lasting pain-like behavior in the CAIA model. Even following the resolution of joint inflammation, the “post-inflammatory” phase, there are residual signs of bone destruction present. Our current work suggests that osteoclasts display an interesting pronociceptive role in the CAIA model that is not only linked to their resorptive actions, but also coupled to their modulation of bone vascularization and innervation via release of factors such as netrin-1. In collaborations with Netris Pharma (France), we are examining the potential of anti-netrin-1 antibody, currently in Phase II clinical trials for cancer, as a possible therapeutic for the treatment of chronic pain.
In addition, we are exploring the antinociceptive mechanism of baricitinib, a disease modifying anti-rheumatic drug used for the treatment of RA, in collaboration with Eli Lilly (USA). Baricitinib is known to reduce joint inflammation and disease activity by targeting the JAK/STAT signaling pathway. Interestingly, it has been shown that baricitinib also targets molecules involved in the clathrin-mediated endocytosis, such as AAK1. We are exploring whether clathrin-mediated endocytosis is an integral aspect of chronic pain signaling in the CAIA model and if this pathway represents an additional pain-reducing mechanism of baricitinib.
Role of NGF in RA-induced pain
NGF has been implicated in joint pain and interestingly a missense mutation in the Ngf gene, identified in a Norrbottnian Swedish family, results in a loss of deep pain sensation in bones and joints. We are examining the role of NGF in joint health using genetically modified mice where NGF is replaced with the human mutated NGF. While a role of NGF in pain signaling has been confirmed in patients with bone cancer and osteoarthritis, as well as animal models, the role of NGF in RA pain and bone erosion is unknown. The overall aim of this project is to explore and understand the link between NGF, bone homeostasis and pain using both evoked pain and non-evoked pain tests to assess deep pain sensation, as well as molecular and imaging techniques.
Mechanisms of chronic pain in osteoarthritis
Pain experienced by patients with osteoarthritis (OA) is highly variable and linked to sex . Identification of molecular mechanisms that underlie sex-dependent differences could provide personalised approaches to patient care. In collaboration with Eva Kosek (Department of Clinical Neuronscience, KI), we are involved in studies addressing this and the neuroimmune components of pain in OA. Joint instability and repeated mechanical joint loading in mice provide clinically relevant models of painful OA. Using such models, we have projects in the lab that are exploring molecular and cellular mechanisms behind chronic pain in OA. One of them has a sex-focus, the project is led by our collaborator Tonia Vincent (Oxford University, UK). Epidemiological studies of OA indicate that pain in females often coincides with lower levels of structural damage and is more often complicated by evidence of central sensitization. Few studies to date have examined molecular mechanisms that may explain this apparent sexual dimorphism in OA. Identifying such pathways could provide a number of clinically useful outcomes: (i) new treatments tailored according to the sex of the patient, (ii) biomarkers (potentially sex-dependent) of painful disease that could be used in clinical evaluation and for personalised approaches to pain management (iii) opportunities to combine treatments to reduce side effects of existing pain therapies.
Macrophage heterogeneity and function in the DRG
Macrophages are resident immune cells that inhabit every organ of the body including the central and peripheral nervous systems. Macrophages can have diverse functions that are tailored to their tissue microenvironment and multiple macrophage subsets have been found in individual organs, including lungs, brain and liver. In this project we are characterizing the distinct subsets of macrophages residing in the DRG from a molecular and functional perspective during homeostasis and in conditions of chronic pain, primarily in models of RA and FM. In addition, just like the blood-brain barrier, there is a blood-nerve barrier, which can prevent the entry of pathogens and neurotoxic compounds into the peripheral nerves. This barrier is significantly more permeable in the DRG, as demonstrated in rodents by the high accumulation of intravenously injected dyes in the DRG parenchyma. One aspect of this project is focused on delineating the interaction between macrophages and the vasculature system in the regulation of tissue homeostasis and development of chronic pain.
Application of new technologies for pain relief and nerve regeneration
In collaboration with Magnus Berggren, Daniel Simon and colleagues (Department of Science and Technology, Linköping University), we are part of two projects that are focused on the development of new technologies for precise spatial and temporal delivery of drugs or other compounds to neurons in vitro and in vivo. In the Ionotronix project we are developing an ionic pump that, after being positioned for example around the sciatic nerve, can be used to deliver local anesthetic drugs in situ, to obtain their potent pain-relief action while avoiding systemic side effects. Moreover, we are exploring the biocompatibility and tolerance of conductive biopolymers, with the ultimate goal of utilizing these substances for in vivo nerve modulation and regeneration.