Our research
Obesity is increasing in an epidemic manner in most countries and constitutes a public health problem by enhancing the risk for diseases such as diabetes, fatty liver disease and atherosclerosis. Together these diseases form a cluster referred to as the metabolic syndrome.
An important factor behind obesity complications is the fat cell (adipocyte). Adipocytes release large amounts of free fatty acids which regulate insulin action and the metabolism of glucose and lipids in skeletal muscle and liver. They also secrete hormones, inflammatory proteins and other substances with peripheral effects on blood vessels, appetite, energy homeostasis, blood pressure and glucose as well as lipid metabolism. Thus, disturbances in the normal functioning of fat cells have significant consequences on the health of an individual. Despite the importance of the fat mass very little is known about the maintenance of fat cells in humans, how different fat depots are regulated and how, or if, this is altered in obesity.
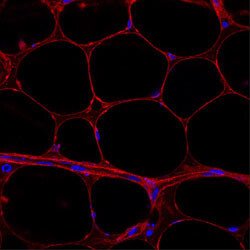
Lipid turnover and cell age are studied using radiocarbon dating. By studying cell turnover in a variety of different adipose depots (such as various subcutaneous adipose depots as well as visceral depots) we aim to better understand the regulation of the fat mass in humans. Understanding the dynamics of adipocyte turnover may shed new light on potential treatments for obesity.
News archive
 Photo: iStock
Photo: iStockNy studie visar varför vi går upp i vikt när vi blir äldre
– Fetma och fetma-relaterade sjukdomar har blivit ett globalt problem. Därför har det aldrig varit mer relevant än nu att förstå hur lipider fungerar och vad som reglerar storleken på fettmassan hos människor, säger Kirsty Spalding.
 Photo: Martin Stenmark
Photo: Martin StenmarkBored at work? Fat chance
Who cares about the fate of an annoying fat cell? Kirsty Spalding has taken on the task with a great deal of energy and a genuine thirst for knowledge.
 Photo: Stefan Zimmerman
Photo: Stefan ZimmermanInterview with Kirsty Spalding
Kirsty studies how fat tissue contribute to diseases such as metabolic diseases (Kirsty studerar hur fettväven bidrar till olika sjukdomar inkluderande metabola sjukdomar). Intervjun görs på engelska av Kerstin Brismar.

