Our research
Our central theme is to understand the immunological effects of extracellular vesicles (EVs). EVs are natural nanovesicles released from all cells and are present in all body fluids. We work both with clinical studies and in different disease models. Our work has revealed that EVs are major players in lung diseases such as asthma and sarcoidosis. We are also studying the immune response to dendritic cell derived EVs, in order to develop EV based immunotherapies for cancer.
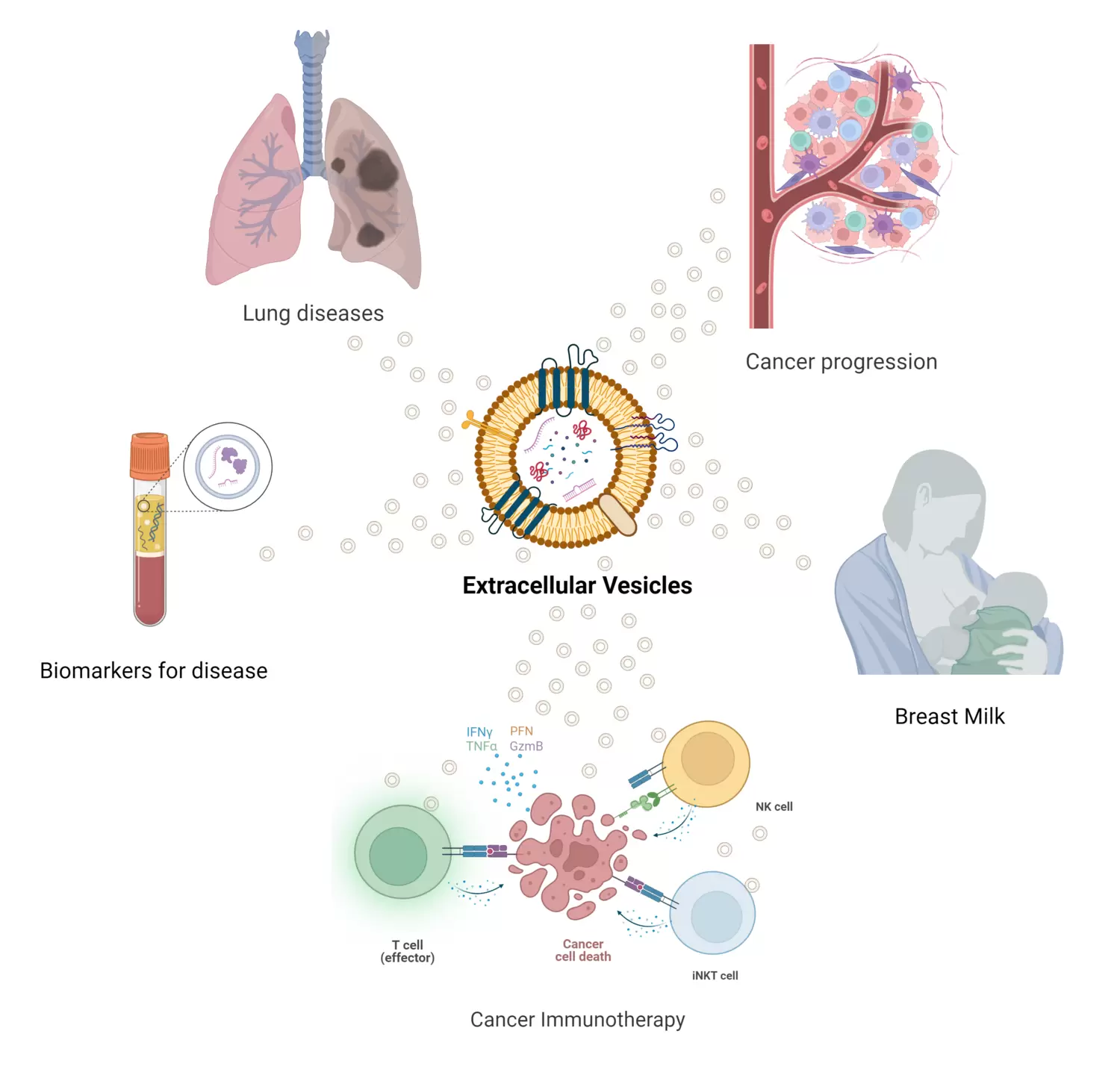
Projects
Understanding the role of EVs in lung diseases
EVs are packed with information such as RNA, proteins and lipids. Since our discovery of EVs in bronchoalveolar lavage fluid in 2003, we have demonstrated that EVs can have pro-inflammatory functions in lung diseases. Now we investigate the molecular basis for this, in order to develop new treatments for asthma, sarcoidosis and Chronic Obstructive Lung Disease (COPD).
EVs as a driver for cancer progression
Cancer cells that mutate to produce EVs that inhibit immune responses, promote blood vessel formation or cell migration have an advantage over other cells. We are analyzing EVs from cancer patients to find new therapeutic targets in cancer.
EVs as biomarkers for disease
EVs are ideal as biomarkers for disease, as they are released from all cells and represent a snapshot of the state of the originating cell, they can therefore represent a liquid biopsy. We develop new methods for EV detection and we analyze patient samples in order to develop biomarkers for early detection, disease monitoring or treatment guidance in both lung diseases and cancer.
EVs as cancer immunotherapy
Currently we are working on different ways to boost immunogenicity of EVs. In different model systems, we engineer EVs to induce cancer-specific immune responses, thereby developing new EV-based cancer treatments, with more specificity and less side effects than current treatments.
EVs in breast milk
Breast milk contains a lot of immune information delivered to the newborn baby. We have discovered that milk contains EVs, and now we are both trying to understand their immunological role, and work to use them in therapeutic applications in e.g. allergies.
The research group

