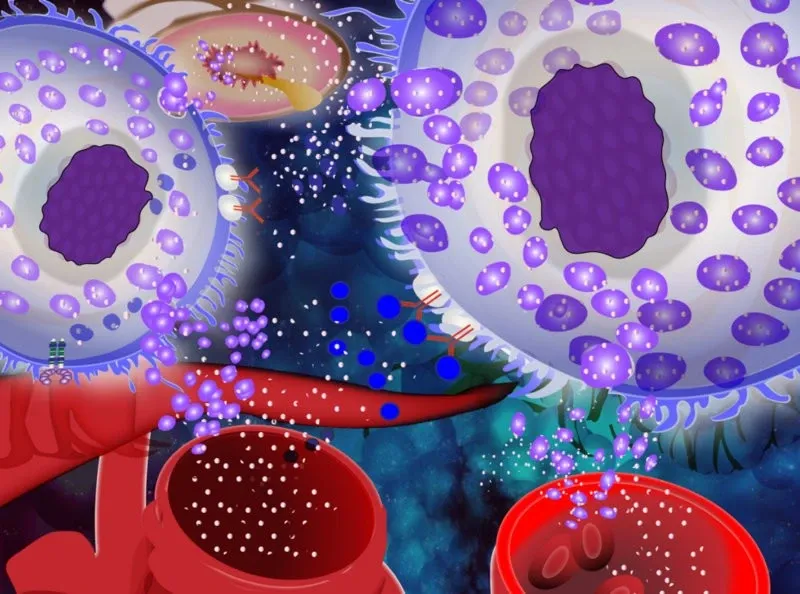
Research focus
Mast cells are (in)famous for their role as effector cells causing the symptoms in allergy and anaphylactic chock. However, we don’t have mast cells to cause problems for us but to protect us. They are an essential part of our host defense system, sensing both endogenous and exogenous “danger signals”, and react to these by initiating an inflammatory response.
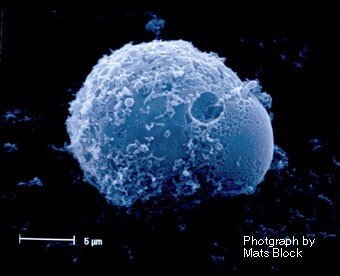
Mast cells have a unique capacity to recognize different molecules and to react to these by releasing specific sets of mediators. Given the great capacity to release powerful mediators, such as histamine proteases, lipid mediators and cytokines, mast cells are also associated with a number of different diseases, where these cells and their mediators are part of the pathology. Examples of diseases where mast cells are involved are asthma, chronic inflammatory skin diseases, cancer and systemic mastocytosis.
The focus for our research is to understand the role of mast cells in health and disease and to study regulatory mechanisms for mast cell activation and mediator release.
Systemic Mastocytosis: Identification of novel biomarkers for anaphylaxis and immune tolerance in mastocytosis
Anaphylaxis is a life-threatening, systemic allergic reaction. Patients with systemic mastocytosis have a significantly higher risk of experiencing an anaphylactic shock than the general population. Our overall goal is to better understand mechanisms of anaphylaxis and immune tolerance to improve treatment options that can prevent anaphylaxis in the future. We screen for specific diagnostic and prognostic biomarkers of anaphylaxis and immune tolerance in mastocytosis patients.
Further, we study the molecular mechanisms underlying the altered protein levels of the identified biomarkers. Understanding these regulatory mechanisms might help to find better treatment options for mastocytosis patients as well as for anaphylactic patients without a clonal mast cell disease.
Projects
Systemic Mastocytosis
Theo Gülen, Joakim Dahlin, Katarina Lyberg, Jennine Grootens
Systemic mastocytosis (SM) is a rare and sometimes fatal disorder, in which the mast cell lineage development is dysfunctional. The disease is associated with an accumulation of mast cells in the body and symptoms caused by the release of mast cell mediators. The long-term goal is to find markers that have a prognostic value for the disease, as well as to identify specific targets that can be used for clinical therapeutic interventions. Several projects are ongoing:
- Identification of dysfunctional regulatory pathways in SM.
- Investigation of developmental checkpoints in the mast cell lineage that might be corrupted.
- Identification of biomarkers that can be used for diagnostic and/or prognostic purposes.
Studies of risk factors for the development of anaphylaxis
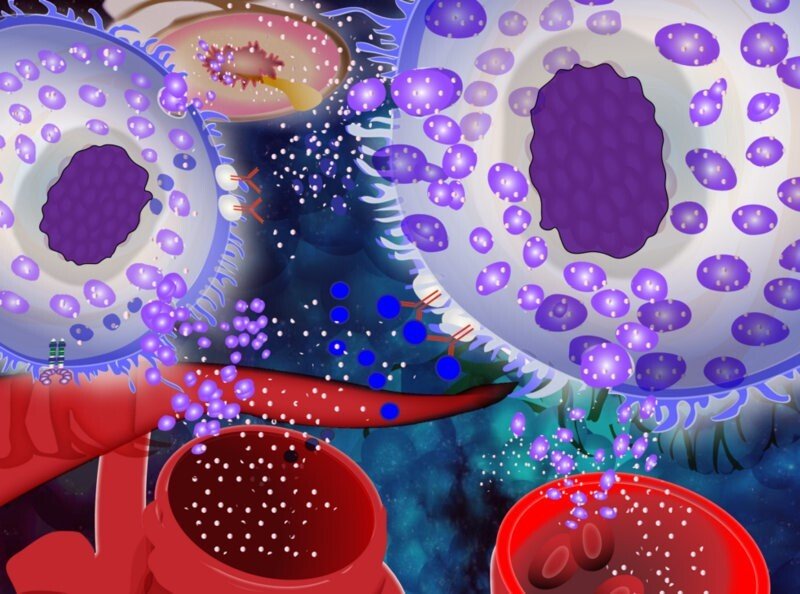
Regulatory functions of microRNAs in mast cells
In this project we study microRNA-based regulatory mechanisms underlying mast cell-activating diseases. The main objective is to identify microRNAs and their target genes regulating mast cell functions, including growth and reactivity. The project is conceived with two specific aims:
- To identify genes and pathways regulated by identified microRNAs in mast cells
- To evaluate plasma microRNAs as biomarkers and diagnostic tools in mast cell-activating diseases
MicroRNAs belong to a big class of small, non-coding RNAs that regulate gene expression by targeting mRNAs for translational repression and/or degradation. These molecules are emerging as important modulators of cellular pathways such as growth, proliferation, apoptosis and inflammatory responses. Mast cell activation occurs in a variety of acute and chronic inflammatory states, playing an important role in the pathogenesis of several different types of diseases. Some of these diseases, such as allergy, asthma, anaphylaxis, urticaria and mastocytosis, are particularly associated with mast cell activation and the symptoms are related to mast cell mediators, and in some cases hyperplasia. However, the etiology and why mast cells are prone to be activated are for many of these diseases not fully deciphered. One area that has attracted a lot of attention recently is the role of microRNA in regulating cellular functions and the use of circulating miRNAs as biomarkers. However, the current knowledge about the role of microRNAs) in regulating mast cell functions is very limited.
Role of interleukin-33 in the development and exacerbation of allergic asthma
Ying Lei, Anna Nilsson
The airway epithelium is considered an essential controller of inflammatory immune responses to allergens, viruses and environmental pollutants that contribute to asthma pathogenesis. Recent experimental and clinical studies have implicated the novel epithelial cells-derived cytokines interleukin-33 (IL-33) in the pathogenesis of respiratory allergy and asthma. The interactions between IL-33 and mast cells and type 2 cytokine–producing innate lymphoid cells (ILC2s) could be promising therapeutic targets for allergic asthma. We sought to investigate the interplays between mast cells, ILC2 cells and IL-33, and their contribution to allergic lung responses by applying our asthma models in both wild type and genetically modified mice. The long-term goal of the study is to translate the conclusions from our well-established in vivo models to subjects with asthma.
Human lung mast cell heterogeneity
Joakim Dahlin, Avinash Ravindran
Lung mast cells are infamous for their devastating role in allergic asthma. Human mast cells are classically divided into two distinct subsets based on their expression of the proteases chymase and tryptase. In this project we further explore the heterogeneity of human lung mast cells, with the ultimate goal to find the characteristic subset of the mast cells causing asthma exacerbations.
The interaction between innate lymphoid cells of type 2 and mast cells in allergic airway inflammation – Avinash Ravindran
Both mast cells and innate lymphoid cells of type 2 have an important role for the development of allergic airway inflammation. The focus of this project is to study the interaction between mast cells and innate lymphoid cells of type 2, and its implication for acute and chronic airway inflammation.
Activation pathways and mast cell mediators. Inhibition of mast cell activation responses
Maria Ekoff
The main focus of this project is to identify substances with the ability to inhibit mast cell release of inflammatory mediators. Mast cells are long-lived effector cells of the immune system perhaps best known for their involvement in allergic diseases. Upon activation, the mast cell releases a variety of inflammatory substances (histamine, leukotrienes, proteases and cytokines) that cause the symptoms we associate with allergy. Today allergy is treated primarily with anti-histamines, i.e. treating the symptoms rather than the cause. A more direct approach would be to prevent/reduce mast cell release. Since mast cell mediators also play a part in many inflammatory diseases, this approach could be used as a novel strategy for treatment also in these settings. A more long-term goal is to perform large scale screenings to find substances or combinations thereof to reduce/abolish mast cell activation. Effective candidate-substances are then tested more extensively in vitro, ex vivo (biopsies) and in vivo using animal models.