Research focus
Parkinson’s disease (PD) is the second most frequent neurodegenerative disorder and is diagnosed based on the appearance of tremor, rigidity, and bradykinesia, secondary to the loss of midbrain dopamine neurons projecting to the basal ganglia. In addition to the cardinal motor symptoms, PD is accompanied by a wide range of comorbidities, which include olfactory and sleep disturbances, affective disorders and cognitive impairment.
These conditions are often refractory to and may even be exacerbated by standard dopamine replacement therapies against PD. In addition, treatment with L-DOPA or dopamine receptor agonists is often accompanied by the development of motor complications, or dyskinesia, and obsessive-compulsive disorders, including pathological gambling and addictive-like behaviour.
Our work is based on the characterization of animal models reproducing these various disorders, combined with multiscale analyses to identify molecular, cellular and circuit abnormalities as potential targets for therapeutic interventions.
Non-motor symptoms in Parkinson’s disease
A major research line centres on the non-motor comorbidities commonly observed in PD patients, which often appear during the prodromal phase of the disease. We have set up a toxin-based mouse model of early-stage PD, which reproduces several of these conditions.
For a detailed description, see Guide to the Generation of a 6-Hydroxydopamine Mouse Model of Parkinson’s Disease for the Study of Non-Motor Symptoms.
We are currently using this model to investigate signaling and circuit abnormalities linked to anxiety and depression. Part of these studies are carried out within the AND-PD consortium, supported by the Horizon2020 EU program.
Other projects focus on circadian rhythm and sleep disorders, which are commonly observed in both de novo and medicated PD patients. Our mouse model of PD is characterized by disrupted rest/activity and endogenous circadian rhythm (more information at: https://doi.org/10.1038/tp.2017.58).
In a similar mouse model, we recently identified important changes in sleep architecture and micro-structure, indicative of excessive daytime sleepiness and sleep fragmentation. This study provides a behavioral framework for the identification of mechanisms involved in PD-related sleep disorders and of potential therapeutic interventions.
Molecular mechanisms of L-DOPA-induced dyskinesia
We have identified several signal transduction components of the cAMP, ERK and mTOR cascades causally linked to the dystonic and choreic uncontrollable movements (dyskinesia) developed in response to administration of L-DOPA.
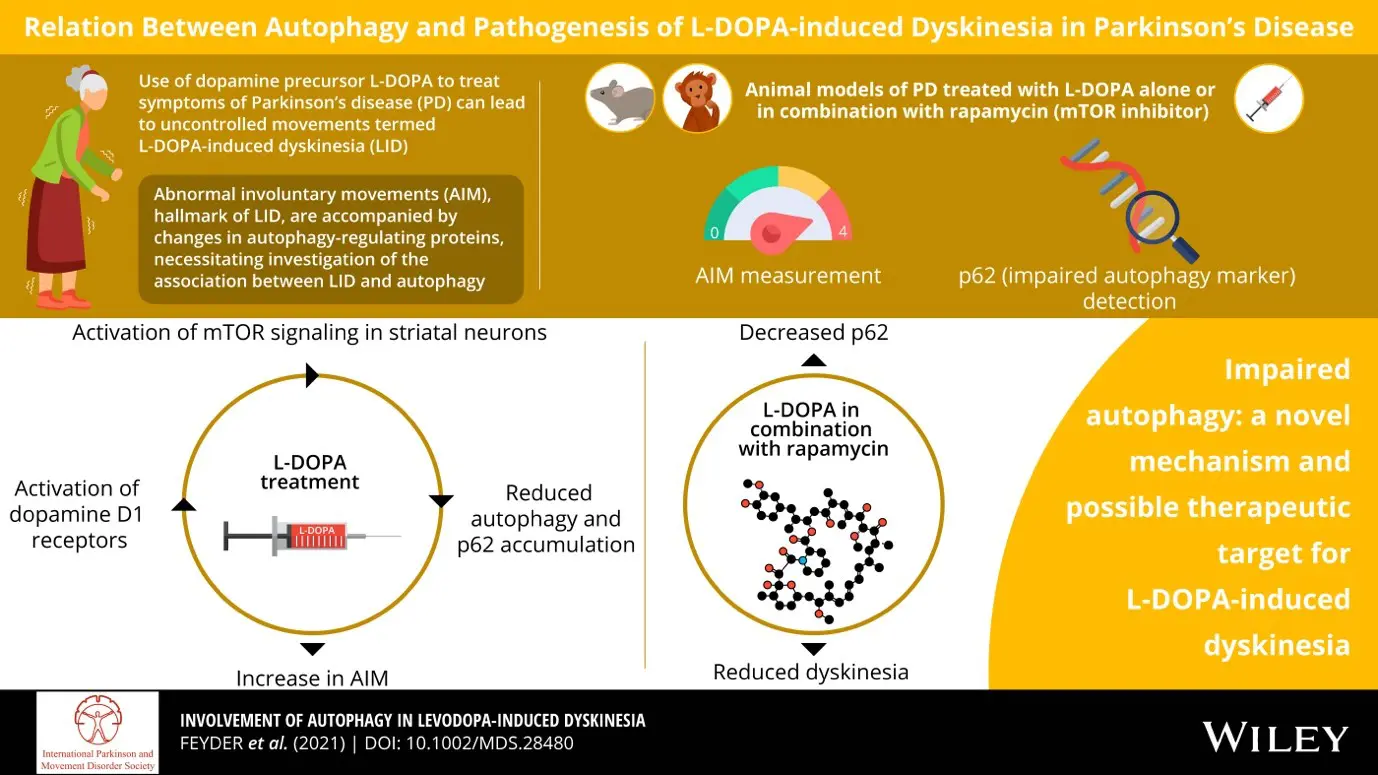
Based on this conceptual background, we provided evidence in support of the involvement of impaired autophagy in L-DOPA-induced dyskinesia. Our study suggests that autophagy promoting drugs, including those approved for the treatment of cancer and metabolic disorders, may also be introduced in the clinic as anti-dyskinetic agents.
Obsessive compulsive disorders in Parkinson’s disease
In another project, we optimized a mouse model of PD displaying obsessive-compulsive disorders associated with dopamine replacement therapy. In particular, we reproduced an addictive-like condition affecting Parkinsonian patients and commonly referred to as dopamine dysregulation syndrome. We are currently utilizing this model to identify mechanisms at the basis of L-DOPA-induced psychiatric complications.