Research
Lung cancer is a common disease characterized by high mortality rates. From a clinico-pathological point of view, lung cancer can be divided into small-cell lung cancer (SCLC, 15%) and non-small cell lung cancer (NSCLC, 85%). The former is an aggressive tumor form where no major advances in treatment have been obtained during the last decades. On the contrary, immunotherapy (IT) based on check-point inhibitors represents a real breakthrough in NSCLC.
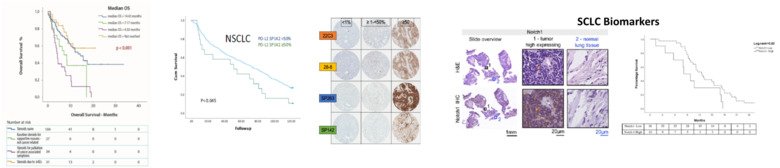
Although targeting the programmed cell death protein 1 (PD1) pathway with monoclonal antibodies has reached clinically meaningful effects in NSCLC, still little is known about which patients benefit most from this approach.
Aims: To improve patient selection for IT in NSCLC based on clinical and molecular biomarkers (BM). To integrate individual treatment pathways with clinical and BM data to increase personalized medicine possibilities even in SCLC.
Design and methods: The NSCLC IT program is based on the analysis of retrospective patient material (n=200) with additional focus on cases with brain metastasis (n=50), a setting where IT is particularly challenging. Our findings will be further validated on a cohort of similar patients on a national basis through the Swedish National Cancer Registry. In addition, liquid biopsies from patients receiving IT in clinical practice (n=200) will also be collected and analyzed for circulating BM (such as proteins on exosomes). Within the SCLC program, we analyze tumor tissues (n=100) in search of subgroup of patients that by a given immune profile (expression of PD-L1, PD1 and STING) might most likely benefit from IT. In addition, we will extract exosomes from liquid biopsies from 50 SCLC and matched cases with NSCLC and benign lung diseases, respectively, in search for SCLC-specific signatures.
Finally, in collaboration with researchers at KTH, we are applying Artificial intelligence models to a cohort of 544 SCLC treated ad our institution, in order to map individual treatment pathways and examine the impact of decision support tools, in precision medicine and precision dosing, on treatment outcomes for this disease. All studies will have direct impact on healthcare, supporting patient selection and exploring novel treatment strategies.
