Research focus
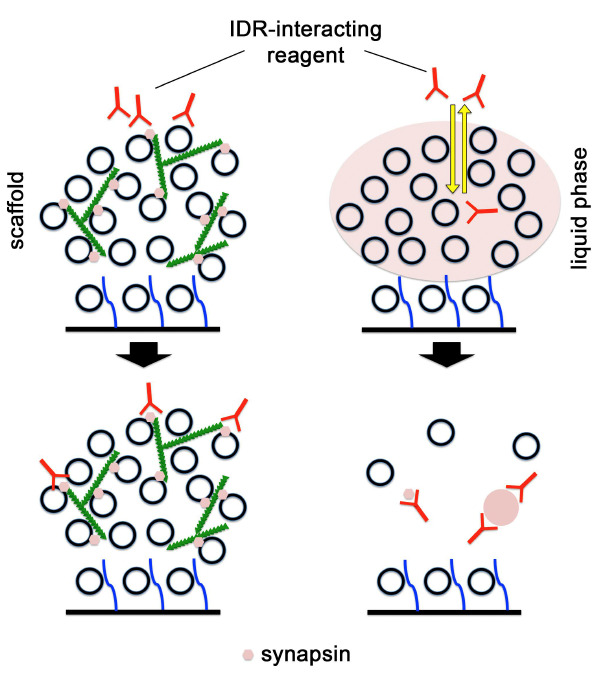
Liquid-liquid phase separation in cells has emerged as a common principle for the organization of membrane-less compartments. In nerve terminals in the central nervous system, liquid-liquid phase separation has been implicated in the organization or assembly of several functional compartments.
We are aiming at characterizing the signaling mechanisms that regulate the liquid-liquid phase transitions in the synaptic vesicle cycle, mitochondrial functions, and degradation pathways at nerve terminals to clarify how do they become affected at the early stages of neurodegenerative diseases and which molecular steps lead to protein aggregate formation.
To address our goals we use several model systems such as giant reticular spinal axon in lamprey, Drosophila neuromuscular junction, and mammalian neurons, in combination with molecular biology, genetics, cellular imaging techniques, and intracellular recordings.
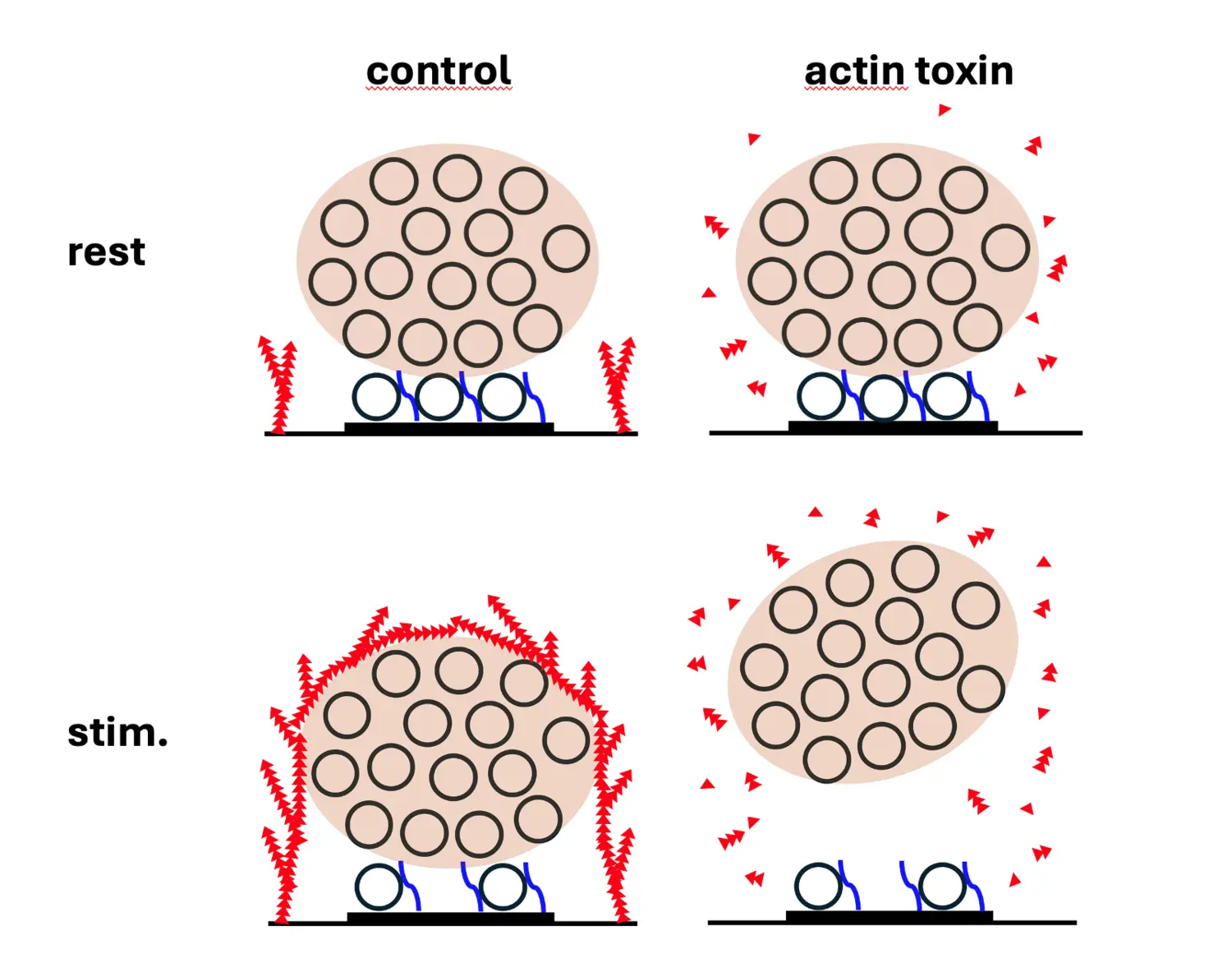
We believe that our experiments will pave the way for the identification of therapeutic targets for treatments of neurodegenerative disorders.
