Research focus
We showed that transplantation of embryonic dopamine neurons can counteract Parkinson’s disease in an animal model, and demonstrated functional effects of a repair strategy for complete spinal cord injury in an adult mammal.
Present focus is aging, neurodegenerative diseases, particularly Parkinson’s disease, the role of the Nogo system in the formation of lasting memories and treatment strategies for spinal cord injury. We have recently found that oral treatment with the clinically used cancer drug Imatinib improves recovery in an animal model of traumatic spinal cord injury. Recent studies of the mtDNA mutator mouse have shown that maternally inherited mtDNA mutations can cause mild aging phenotypes and aggravate premature aging phenotypes in mice carrying mutated PolgA genes.
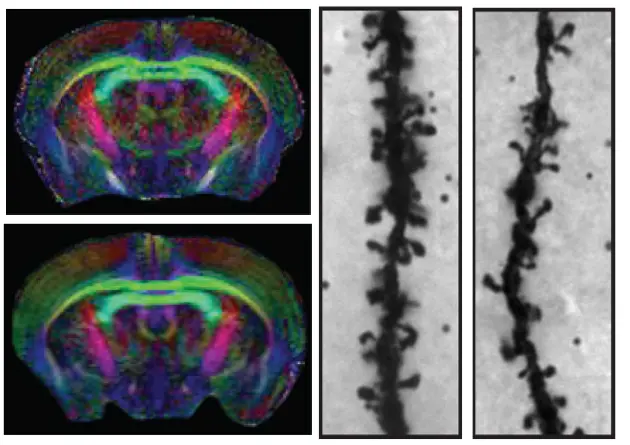
We have recently shown how several genes encoding proteins belonging to the nerve growth inhibitory Nogo signaling system are regulated to influence structural synaptic plasticity and the formation of lasting memories. Ongoing studies using transgenic animals aim at understanding molecular mechanisms behind the formation of lasting memories and causes of memory disorders, as seen e.g. in aging.
Overexpression of Nogo receptor 1 (NgR1) in forebrain neurons does not affect gross brain anatomy but inhibits the formation of dendritic spines.
