Photo: Dhifaf Sarhan
Photo: Dhifaf SarhanResearch line I: Studies of sexual immune dimorphism in the TM
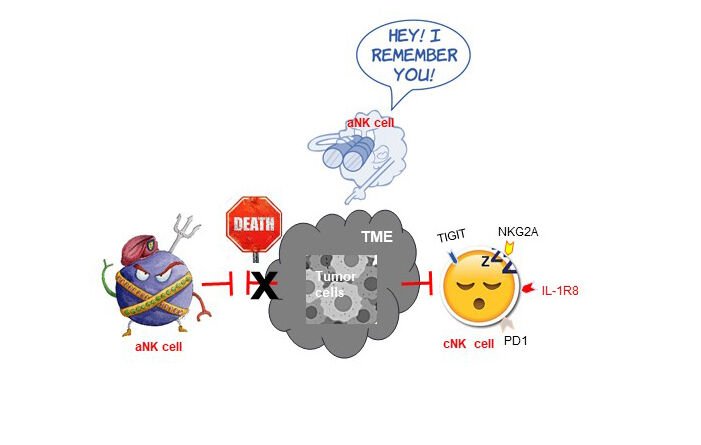
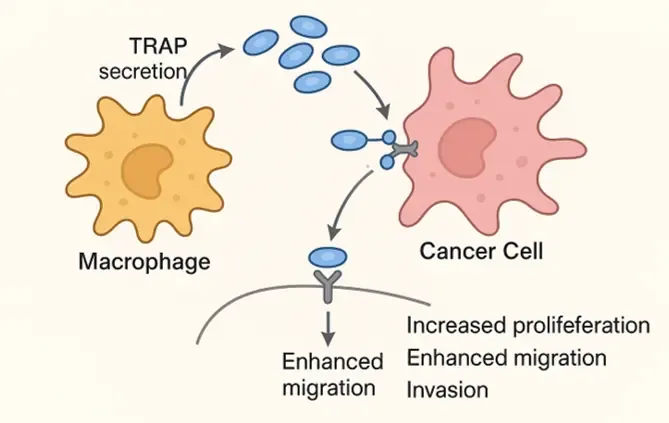
The Tumor Immunology & Immunotherapy Group aim to fill the knowledge gap about differences in sex-specific immune responses and how these contribute to the response to immunotherapy.