Team Innate immune system in infection, inflammation, and cancer
Our team studies the innate immune system in acute and chronic inflammatory diseases in children and adults.
The team’s focus
The innate immune system is instrumental in infection, inflammation, and cancer, yet the characterization of disease-specific functional states, metabolic programs, modes of intercellular communication, as well as their translation into therapeutic interventions and biomarkers remains incomplete. Our goal is to identify disease-specific immune signatures that enable improved diagnostics, and personalized therapeutic strategies, with relevance for global health and pandemic preparedness.
Our team investigates how innate immune mechanisms drive disease pathogenesis across inflammatory skin diseases, high-burden viral infections, and inflammatory neoplasias. We focus on myeloid cell populations, especially granulocytes, to define how disease-specific innate immune states shape inflammation, tissue damage, immune–neural interactions, and long-term pathology.
Through interdisciplinary collaborations and a systems immunology framework, we integrate advanced immune profiling, spatial multi-omics, and extracellular vesicle analyses to translate mechanistic insights into innovative diagnostic and therapeutic strategies.
Team leader
Magdalini Lourda
Senior Research SpecialistTeam Members

Rossana Maria Felicia Serlenga
MMedSc studentPrevious team members
- Avinash Padhi, PhD. Postdoc.
- Sabia Miah, MSc student. Master’s degree project.
- David Fusté Vegas, BSc student. Erasmus+ traineeship.
- Lara Antonia Kaland Thielcke, BSc student. Research assistant.
- Mira Akber, MSc. Research trainee project. Research assistant.
Media
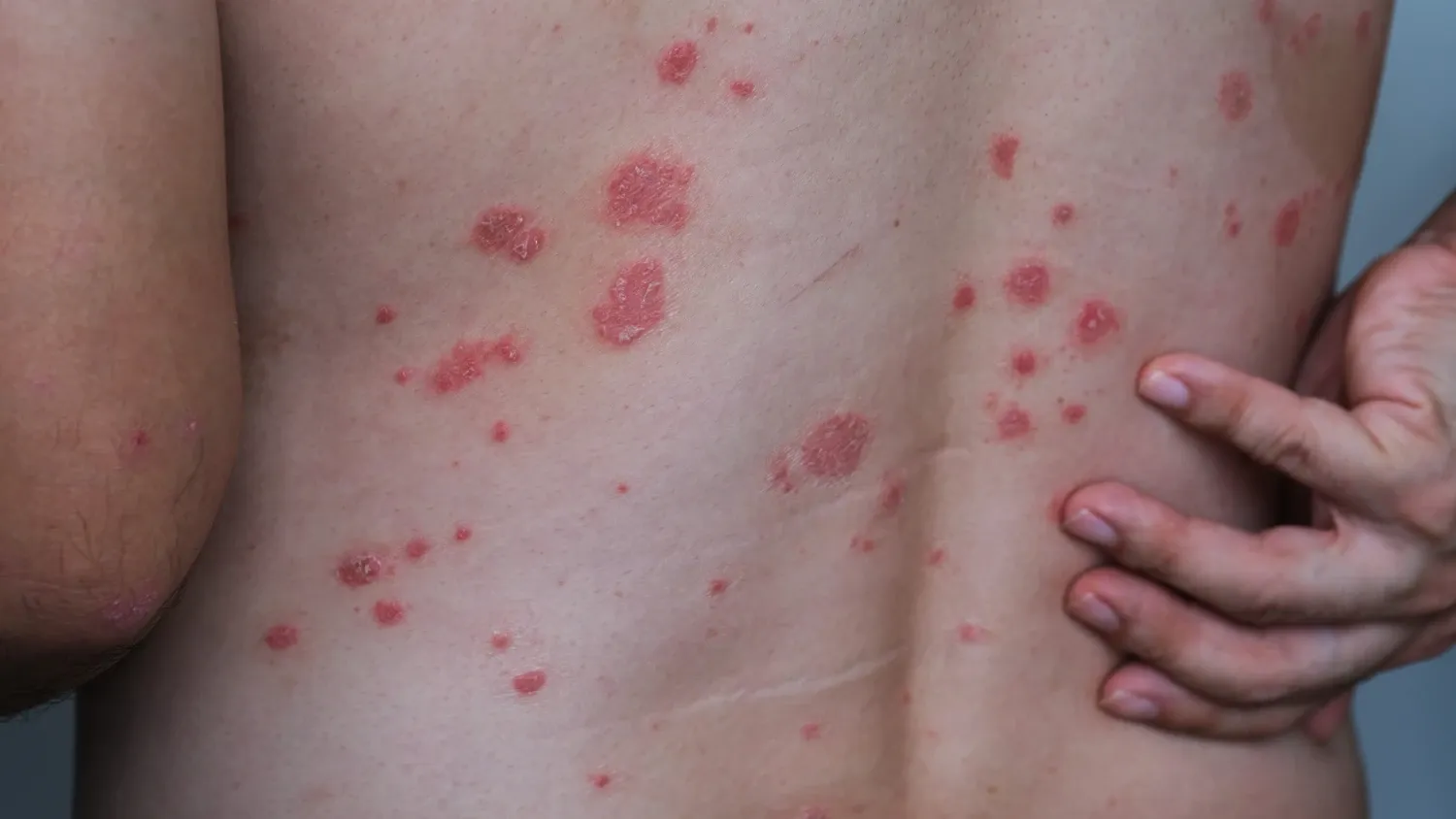 Photo: Getty Images
Photo: Getty ImagesNewly found immune cells link strep throat to psoriasis
A common strep throat infection can trigger guttate psoriasis by altering the behaviour of key immune cells, according to a new study from Karolinska Institutet published in eBioMedicine. The findings suggest how an infection can lead to sudden skin inflammation, particularly in children and young adults.
Current Research Projects
Innate immune mechanisms underlying psoriasis pathogenesis
Psoriasis is a chronic inflammatory skin disease driven by aberrant innate immune activation and prominent neutrophil infiltration. Neutrophils sustain inflammation through cytokine release, metabolic reprogramming, extracellular trap formation, and communication with keratinocytes and sensory neurons, contributing to disease chronicity and pruritus.
We investigate how granulocyte heterogeneity and altered innate immune signalling drive psoriasis across clinical phenotypes. A central focus is the metabolism–inflammation interplay as a regulator of granulocyte migration and function. In parallel, we study extracellular vesicles (EVs) as mediators of immune–stromal–neuronal communication, aiming to identify biomarkers of disease severity, itch, and treatment response using high-dimensional spectral flow cytometry, single-cell RNA sequencing, spatial multi-omics, and EV profiling.
Mapping granulocyte-driven pathways in severe viral infections
Severe viral infections such as COVID-19 and dengue expose critical vulnerabilities in host innate immune responses. Our prior work in COVID-19 identified marked granulocyte heterogeneity and severity-associated functional states. Building on this, we investigate how granulocyte–platelet interactions and thromboinflammatory pathways contribute to systemic inflammation and severe outcomes.
Extending these insights to dengue, we study granulocyte responses associated with disease severity in adults (within the COMBATconsortium) and children (within the PRISM project). By integrating high-dimensional immune profiling with proteomics, transcriptomics, and clinical data, we aim to identify early functional and metabolic signatures linked to progression to severe disease and to define clinically relevant risk markers across viral infections.
Innate immune triggers and neurodegeneration in Langerhans cell histiocytosis (LCH)
LCH is a rare inflammatory disease in which oncogenic mutations intersect with innate immune dysregulation, leading to heterogeneous clinical outcomes. Lesions are characterized by complex myeloid infiltrates, including pathogenic dendritic cells and eosinophils, reflecting profound immune activation. A major unmet need is identifying the immune triggers of disease initiation and progression, particularly those driving central nervous system involvement and neurodegeneration in children.
Our research focuses on how innate immune activation, eosinophil-associated inflammation, and EV-mediated signalling contribute to disease initiation, progression, and neurodegeneration. By integrating immune profiling, EV analyses, and advanced model systems, we aim to identify mechanisms and biomarkers that can be targeted to halt or prevent neurodegeneration.
Our research in media
News articles
Newly found immune cells link strep throat to psoriasis
Innate immune response may predict COVID-19 severity
Blodprov kan förutspå hur sjuk du blir i covid-19 (only in Swedish)
Research funding
- The Swedish Childhood Cancer Fund
- Dr Åke Olsson foundation for haematological research
- Gösta A Karlssons 60-årsfond
- Karolinska Institute Foundations & Donations
- Karolinska Institute Doctoral funding
Open positions
We collaborate closely with clinicians, immunologists, virologists, computational scientists and other researchers in Sweden and internationally.
Researchers who are interested in applying for external funding (e.g., Marie Skłodowska-Curie Actions Postdoctoral Fellowships, Swedish Research Council or similar schemes) and would like to develop their proposal within our research environment are welcome to contact the Team leader at: magdalini.lourda@ki.se with a CV and a brief description of their research interests.
Selected publications
High-dimensional single-cell analyses reveal neutrophil heterogeneity in guttate psoriasis.
Padhi A, Ambikan AT, Andriopoulos P, Sinha I, Akber M, Zheng W, Rekha RS, Palma Medina L, Henter JI, Svensson M, Eidsmo L, Norrby-Teglund A, Neogi U, Bergman P, Lysell J, Lourda M
eBioMedicine. 2026 March;125:106172
Host Plasma Microenvironment in Immunometabolically Impaired HIV Infection Leads to Dysregulated Monocyte Function and Synaptic Transmission Ex Vivo.
Mikaeloff F, Gelpi M, Escós A, Wang T, Gupta S, Olofsson A, Akusjärvi SS, Schuster S, Naval P, Sood V, Nikouyan N, Knudsen AD, Vestad B, Høgh J, Hov JR, Benfield T, Trøseid M, Pawar V, Rucevic M, Benfeitas R, Végvári Á, O'Mahony L, Savai R, Björkström NK, Lourda M, de Magalhães JP, Weiss S, Mardinoglu A, Varshney MK, Karlsson AC, Syed YA, Nielsen SD, Neogi U
Adv Sci (Weinh). 2025 Apr;12(16):e2416453
Intestinal stroma guides monocyte differentiation to macrophages through GM-CSF.
Kvedaraite E, Lourda M, Mouratidou N, Düking T, Padhi A, Moll K, Czarnewski P, Sinha I, Xagoraris I, Kokkinou E, Damdimopoulos A, Weigel W, Hartwig O, Santos TE, Soini T, Van Acker A, Rahkonen N, Flodström Tullberg M, Ringqvist E, Buggert M, Jorns C, Lindforss U, Nordenvall C, Stamper CT, Unnersjö-Jess D, Akber M, Nadisauskaite R, Jansson J, Vandamme N, Sorini C, Grundeken ME, Rolandsdotter H, Rassidakis G, Villablanca EJ, Ideström M, Eulitz S, Arnell H, Mjösberg J, Henter JI, Svensson M
Nat Commun 2024 Feb;15(1):1752
Myeloid cells from Langerhans cell histiocytosis patients exhibit increased vesicle trafficking and an altered secretome capable of activating NK cells.
Hagey DW, Kvedaraite E, Akber M, Görgens A, Javadi J, Von Bahr Greenwood T, Björklund C, Åkefeldt SO, Hannegård-Hamrin T, Arnell H, Dobra K, Herold N, Svensson M, El Andaloussi S, Henter JI, Lourda M
Haematologica 2023 Sep;108(9):2422-2434
Role of myeloid cells in system-level immunometabolic dysregulation during prolonged successful HIV-1 treatment.
Svensson Akusjärvi S, Krishnan S, Ambikan AT, Mikaeloff F, Munusamy Ponnan S, Vesterbacka J, Lourda M, Nowak P, Sönnerborg A, Neogi U.
AIDS 2023 June;37(7):1023-1033
Multi-omics personalized network analyses highlight progressive disruption of central metabolism associated with COVID-19 severity.
Ambikan AT, Yang H, Krishnan S, Svensson Akusjärvi S, Gupta S, Lourda M, Sperk M, Arif M, Zhang C, Nordqvist H, Ponnan SM, Sönnerborg A, Treutiger CJ, O'Mahony L, Mardinoglu A, Benfeitas R, Neogi U
Cell Syst 2022 Aug;13(8):665-681.e4
Screening for neurodegeneration in Langerhans cell histiocytosis with neurofilament light in plasma.
Sveijer M, von Bahr Greenwood T, Jädersten M, Kvedaraite E, Zetterberg H, Blennow K, Lourda M, Gavhed D, Henter JI
Br J Haematol 2022 Aug;198(4):721-728
High-dimensional profiling reveals phenotypic heterogeneity and disease-specific alterations of granulocytes in COVID-19.
Lourda M, Dzidic M, Hertwig L, Bergsten H, Palma Medina LM, Sinha I, Kvedaraite E, Chen P, Muvva JR, Gorin JB, Cornillet M, Emgård J, Moll K, García M, Maleki KT, Klingström J, Michaëlsson J, Flodström-Tullberg M, Brighenti S, Buggert M, Mjösberg J, Malmberg KJ, Sandberg JK, Henter JI, Folkesson E, Gredmark-Russ S, Sönnerborg A, Eriksson LI, Rooyackers O, Aleman S, Strålin K, Ljunggren HG, Björkström NK, Svensson M, Ponzetta A, Norrby-Teglund A, Chambers BJ,
Proc Natl Acad Sci U S A 2021 Oct;118(40):e2109123118
Complete publication list
https://staff.ki.se/people/magdalini-lourda
