Tomography
Electron tomography enables visualization of proteins and organelles in situ; within the cell without isolation and purification. This method can be used under cryo conditions for cells, or at room temperature to look at fibers or other non-symmetrical particles.
Explore
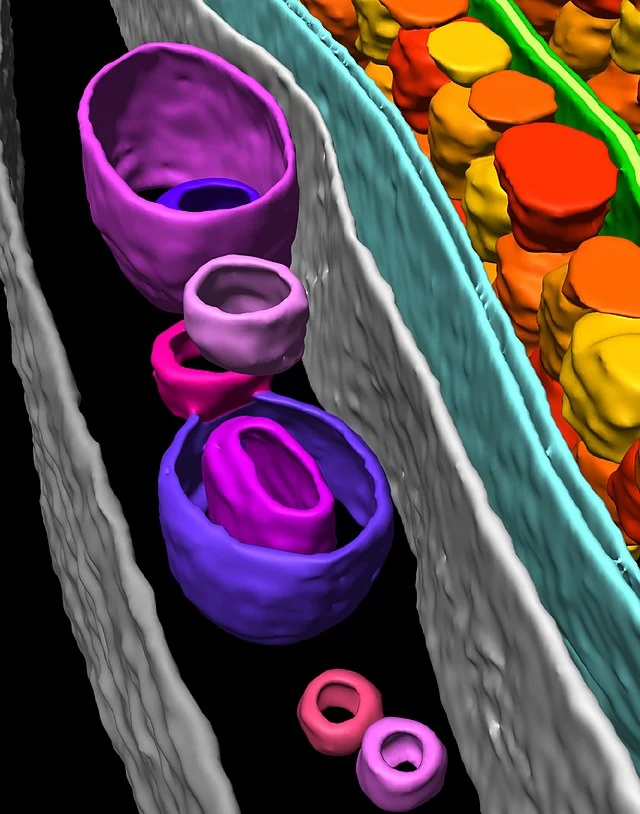
Electron tomography of cells and biological material is a powerful 3D visualization method that can deliver both structural information about individual proteins as well as their spatial arrangements within the cell. Cryo-electron tomography bridges the gap between light microscopy and near-atomic resolution techniques like single particle analysis, achieving resolutions on the scale of 1-4 nm.
This method requires taking a tilt series of an object, followed by alignment, and then segmentation of objects of interest. Cell samples for tomography can be prepared using plunge freezing (Vitrobot) or high pressure freezing (Leica EM ICE). If plunge frozen cells or regions of cells are electron transparent (200 nm), they can immediately be used for tomography in our cryo-TEM (Krios). However, often cells need to be thinned and lamella created (Aquilos 2) before the sample is thin enough for tomography. Furthermore, cryo-fluorescence microscopy is often used with this method to identify the locations of fluorescently tagged features of interest in a method known as cryo-CLEM (sp8 cryo-confocal).
The workflow may vary dramatically depending on your target but here are a few examples using the 3D-EM facility’s instrumentation:
- Vitrobot ► Krios
- Vitrobot ► Aquilos ► Krios
- EM ICE ► sp8 cryo confocal ► Aquilos ► Krios
Data collection softwares available for the Krios: Tomography, Serial EM
Processing softwares available: WARP, Amira
Additional resources
Cryo-electron tomography
Doerr, A.
Nat Methods 14, 34 (2017)
Biophysical Techniques for Characterization of Cells.
V. Lučić, et al.
Comprehensive Biophysics (2012).
Getting Started with In Situ Cryo-Electron Tomography.
Serwas D, Davies KM
Methods Mol Biol 2021 ;2215():3-23
