Precision Genome Engineering
CFG provides flexible and affordable gene-editing services in cells. We offer knock-outs, knock-ins, and point mutations in your cell type of choice.
Service Area Precision Genome Engineering
Precision genome engineering allows the defined and precise modification of a genomic locus. CFG performs such modifications in cell lines, primary cells and ESCs, and designs CRISPR reagents for the creation of genetically modified mice. Application examples are the creation of disease-representative model systems, of reporter cell lines for high-throughput screens or other applications, the validation of hits from such screens, creation of isogenic cell lines, and many more. CFG has a rich toolbox for creating knock-outs, knock-ins, small mutations, or large deletions, including HDR-based editing, base-editing and prime-editing.
Our workflow
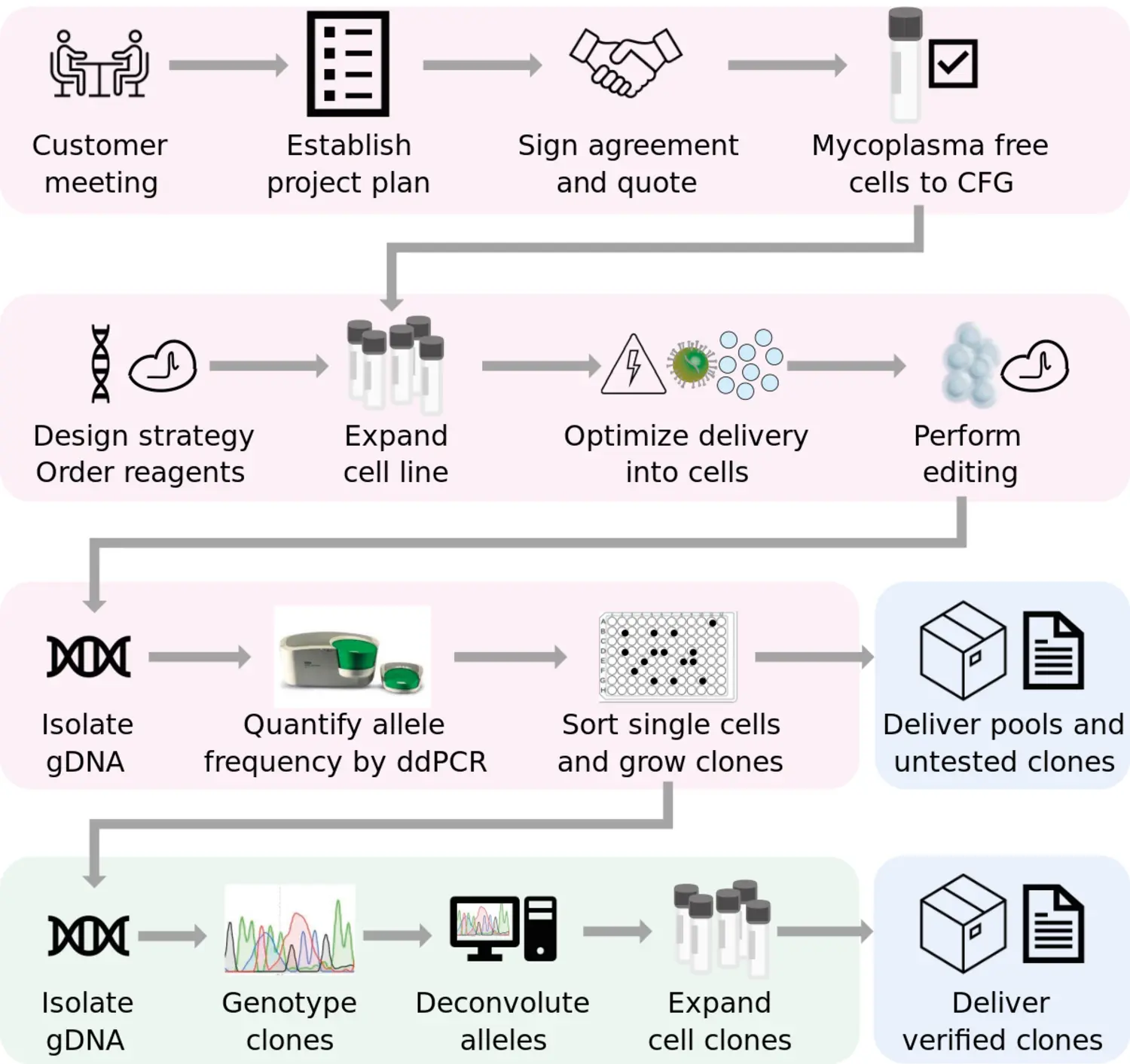
A typical project begins with an initial meeting where we establish a detailed project plan together with the client. Following this, we provide a project quote, and upon mutual agreement, both parties sign a project agreement.
The customer is responsible for delivering two vials of mycoplasma-tested cells per cell line. We then design an optimized CRISPR editing strategy, culture the cells, and, if necessary, optimize the delivery method for the CRISPR system. Once the editing is performed, we harvest genomic DNA and quantify allele frequency using droplet digital PCR.
Single-cell clones are sorted and can either be delivered to the customer for genotyping or genotyped by us. If we handle the genotyping, we will provide two validated clones per edit. These clones will also be tested to ensure they are mycoplasma-free before delivery.
Strategy design
We determine the optimal guide sequences and recombination templates (dsDNA, ssDNA, ssODN). We then custom design a genome engineering strategy for each project, with an emphasis on facilitating downstream screening for correctly edited cell clones.
Optimization of delivery method and delivery into target cells
With input from the client, we determine the best way of delivery of the editing components into cells. We most commonly use electroporation (Neon NxT Transfection System, ThermoFisher Scientific).
Evaluating editing efficiency by droplet digital PCR
Quantification of the editing frequency in a cell pool is an important step in the process, and currently requires dedicated instrumentation. We either use amplicon sequencing or droplet digital PCR (ddPCR) to directly quantify the frequency of edited alleles in a population of cells. These assays determine whether editing efficiency is sufficient to proceed to clone growing.
ddPCR responsible

Anneke Navis
Research Infrastructure SpecialistThis system is also available for researchers to use following a brief introduction. Please note that a user fee applies.
Clone growing and genotyping
As CRISPR editing results in a genetically diverse pool of cells, clone growing is essential for obtaining cells with a correct genotype. We sort single cells in 96-well plates by FACS or apply limiting dilution. Clones can be either expanded and genotyped by the customer, or CFG can take over clone growing and genotyping (contingent upon available capacity). We generally genotype by Sanger or amplicon sequencing (Oxford Nanopore Technologies).
Transgenic mouse models
CFG has a long-standing collaboration with the Karolinska Center for Transgene Technologies (KCTT). CFG’s CRISPR expertise combined with KCTT’s mouse expertise provides a seamless service pipeline from CRISPR strategy design to transgenic animals. CFG provides strategy and reagents, and KCTT injects or electroporates mouse zygotes with the designed CRISPR-Cas components and repair templates for the one-step creation of genetically modified mice.
