
- >
- >
- >
Signal Transduction – Ismael Valladolid Acebes' research group
We focus on Signal transduction in the pancreatic beta-cell. Pancreatic beta-cell signal transduction is complex and involves a well-regulated interaction of a number of signals generated by the metabolism of glucose and the activation of a variety of receptor-operated pathways. Our future research will tell to what extent these various signalling pathways are really regulatory pathways under in vivo conditions, or rather serve as signalling pathways maintaining normal beta-cell function.
Calendar Signal Transduction
 Photo: Roger Brolén
Photo: Roger BrolénSignal Transduction Research group
The group conducts research at the Rolf Luft Research Center for Diabetes and Endocrinology.
Research Areas
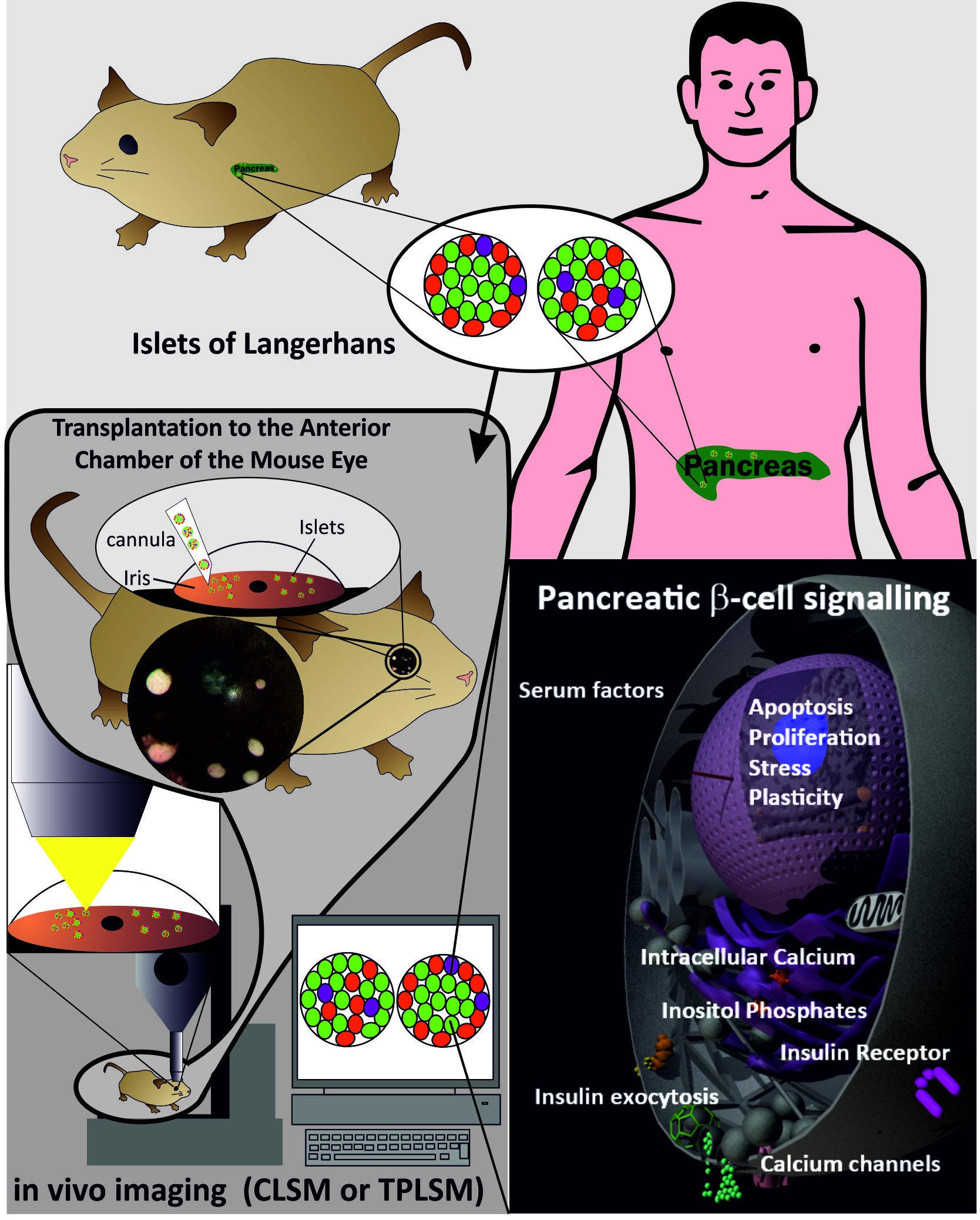
The coupling of glucose metabolism to electrical activity remains central in all models of beta-cell stimulus-secretion coupling. The resting membrane potential of the beta-cell is set by the ATP-sensitive potassium (KATP) channel. Incubation of the pancreatic beta-cell with stimulatory glucose concentrations leads to the activation of a cascade of reactions which ends in the exocytosis of stored insulin. This complex of processes starts with the uptake of glucose by the beta-cell high-Km/low affinity glucose transporter GLUT2 and proceeds with the conversion of glucose into glucose-6-phosphate by the beta-cell isoform of glucokinase. Metabolism of glucose in glycolysis and the Krebs cycle results in the generation of ATP. Elevation in the ATP/ADP ratio leads to closure of the KATP, which in turn results in depolarization of the plasma membrane.
The subsequent opening of voltage-gated L-type Ca2+ channels leads to an increase in the cytoplasmic free Ca2+ concentration, [Ca2+]i, which promotes insulin secretion. With regard to the actions of the plethora of additional factors like neurotransmitters, islet generated factors and systemic growth factors; they are in most cases mediated by membrane receptors coupled to either G-proteins or tyrosine kinases, many of which subsequently activate the phosphoinositide-derived second messenger cascades. Among other things, the role of signalling through these receptor-operated effector systems is the focus of our work.
Pancreatic beta-cell signal transduction is complex and involves a well-regulated interaction of a number of signals generated by the metabolism of glucose and the activation of a variety of receptor-operated pathways. Our future research will tell to what extent these various signalling pathways are really regulatory pathways under in vivo conditions or rather serve as signalling pathways maintaining normal beta-cell function.
Subgroups
Publications
All publications from group members
- Journal article: ADVANCES IN BIOLOGICAL REGULATION. 2026;99:101121Troster P; Visa M; Berggren P-O
- Journal article: NATURE COMMUNICATIONS. 2025;16(1):10047Troster P; Visa M; Valladolid-Acebes I; Kohler M; Berggren P-O
- Journal article: MICROSYSTEMS & NANOENGINEERING. 2025;11(1):213Tian X; Kavand H; Kohler M; Jessika J; Gjaci R; Visa M; Berggren P-O; Stemme G; van der Wijngaart W; Herland A; Roxhed N
- Doctoral thesis: 2025Wang Z
- Journal article: DEVELOPMENTAL CELL. 2025;60(18):2385-2387Xiong Y; Berggren P-O
- Article: NANO LETTERS. 2025;25(31):11860-11869Wang Z; Zhu X; Zhou Y; Zhang T; Shi Y; Zheng X; Yang S-N; Li W; Berggren P-O
- Letter: SIGNAL TRANSDUCTION AND TARGETED THERAPY. 2025;10(1):247Recio-Lopez P; Berggren P-O; Visa Majoral M; Valladolid-Acebes I; Juntti-Berggren L
- Article: JOURNAL OF CLINICAL INVESTIGATION. 2025;135(15):e177601Xiong Y; Dicker A; Visa M; Ilegems E; Berggren P-O
- Article: INTERNATIONAL JOURNAL OF MOLECULAR SCIENCES. 2025;26(3):1383Wang Z; Ou Y; Zhu X; Zhou Y; Zheng X; Zhang M; Li S; Yang S-N; Juntti-Berggren L; Berggren P-O; Zheng X
- Article: ADVANCED SCIENCE. 2025;12(3):e2407572Youhanna S; Kemas AM; Wright SC; Zhong Y; Klumpp B; Klein K; Motso A; Michel M; Ziegler N; Shang M; Sabatier P; Kannt A; Sheng H; Oliva-Vilarnau N; Buettner FA; Seashore-Ludlow B; Schreiner J; Windbergs M; Cornillet M; Bjorkstrom NK; Hulsmeier AJ; Hornemann T; Olsen JV; Wang Y; Gramignoli R; Sundstrom M; Lauschke VM
- Review: BIOMEDICINES. 2024;12(11):2422Valladolid-Acebes I
- Conference publication: ACTA PHYSIOLOGICA. 2024;240:26-27Dolensek J; Klemen MS; Kopecky J; Paradiz E; Kercmar LJ; Bombek LK; Pohorec V; Rupnik MS; Valladolid-Acebes I; Stozer A
- Article: DIABETOLOGIA. 2024;67(8):1663-1682Visa M; Berggren P-O
- Review: PHYSIOLOGICAL REVIEWS. 2024;104(3):881-929Yang S-N; Shi Y; Berggren P-O
- Article: NANO LETTERS. 2024;24(24):7548-7556Li W; Liu S; Wang Z; Gou L; Ou Y; Zhu X; Zhou Y; Zhang T; Liu J; Zheng X; Berggren P-O; Liu J; Zheng X
- Article: HEPATOLOGY. 2024;79(6):1337-1351Oliva-Vilarnau N; Beusch CM; Sabatier P; Sakaraki E; Tjaden A; Graetz L; Buettner FA; Dorotea D; Nguyen M; Bergqvist F; Sundstrom Y; Mueller S; Zubarev RA; Schulte G; Tredup C; Gramignoli R; Tietge UJF; Lauschke VM
- Article: ACTA PHYSIOLOGICA. 2024;240(5):e14128Addinsall AB; Cacciani N; Moruzzi N; Akkad H; Maestri A; Berggren P-O; Widegren A; Bergquist J; Tchkonia T; Kirkland JL; Larsson L
- Article: JOVE-JOURNAL OF VISUALIZED EXPERIMENTS. 2024;(205)Lazzeri-Barcelo F; Ciardo P; Leibiger B; Leibiger IB; Berggren P-O; Moruzzi N
- Article: FRONTIERS IN NEUROSCIENCE. 2024;18:1328815Willekens SMA; Morini F; Mediavilla T; Nilsson E; Oradd G; Hahn M; Chotiwan N; Visa M; Berggren P-O; Ilegems E; Overby AK; Ahlgren U; Marcellino D
- Article: FRONTIERS IN CELL AND DEVELOPMENTAL BIOLOGY. 2024;12:1380564Lipovsek S; Dolensek J; Daris B; Valladolid-Acebes I; Vajs T; Leitinger G; Stozer A; Skelin Klemen M
- Article: NATURE COMMUNICATIONS. 2024;15(1):767Lazzeri-Barcelo F; Oliva-Vilarnau N; Baniol M; Leibiger B; Bergmann O; Lauschke VM; Leibiger IB; Moruzzi N; Berggren P-O
- Article: ADVANCED MATERIALS. 2024;36(1):e2306686Kavand H; Visa M; Kohler M; van der Wijngaart W; Berggren P-O; Herland A
- Article: CELL TRANSPLANTATION. 2024;33:09636897241241995Branstrom R; van Krieken PP; Frobom R; Juhlin CC; Shabo I; Leibiger B; Leibiger IB; Berggren P-O; Aspinwall CA
- Doctoral thesis: 2023Zhao K
- Conference publication: DIABETOLOGIA. 2023;66(SUPPL 1):S220-S221Ballesteros-Pla C; Sevillano J; Sanchez-Alonso M; Limones M; Pita J; Pizarro Delgado J; Izquierdo A; Gomez GM; Valladolid-Acebes I; Herradon G; Alvarez MR
- Article: BIOCHEMICAL PHARMACOLOGY. 2023;215:115755Oliva-Vilarnau N; Vorrink SU; Buettner FA; Heinrich T; Sensbach J; Koscielski I; Wienke D; Petersson C; Perrin D; Lauschke VM
- Doctoral thesis: 2023Tun SBB
- Article: FRONTIERS IN BIOENGINEERING AND BIOTECHNOLOGY. 2023;11:1147244Voznesenskaya A; Berggren P-O; Ilegems E
- Editorial: FRONTIERS IN PHYSIOLOGY. 2023;14:1228926Bergamini C; Bonora E; Moruzzi N
- Article: BIOMOLECULES. 2023;13(6):885Yu J; Leibiger B; Yang S-N; Shears SBB; Leibiger IBB; Berggren P-O; Barker CJJ
- Review: SEMINARS IN LIVER DISEASE. 2023;43(02):149-162Adori M; Bhat S; Gramignoli R; Valladolid-Acebes I; Bengtsson T; Uhlen M; Adori C
- Article: BIOMEDICINES. 2023;11(3):807Zhao K; Shi Y; Yu J; Yu L; Kohler M; Mael A; Kolton A; Joyce T; Odorico J; Berggren P-O; Yang S-N
- Article: CELLULAR SIGNALLING. 2023;102:110535Ma J; Liu Y; Valladolid-Acebes I; Recio-Lopez P; Peng G; Li J; Berggren P-O; Juntti-Berggren L; Tong N
- Article: BIOMEDICAL OPTICS EXPRESS. 2023;14(1):54-64Park WY; Kim J; Le H; Kim B; Berggren P-O; Kim KH
- Article: BIOFACTORS. 2023;49(1):153-172Recio-Lopez P; Valladolid-Acebes I; Hadwiger P; Hossbach M; Krampert M; Prata C; Berggren P-O; Juntti-Berggren L
- Article: METHODS IN MOLECULAR BIOLOGY. 2023;2592:21-36Moede T; Tun SBB; Leibiger IB; Berggren P-O
- Article: OBESITY RESEARCH & CLINICAL PRACTICE. 2023;17(1):86-90Rodriguez A; Korzeniowska K; Szarejko K; Borowski H; Mysliwiec M; Brzezinski M; Czupryniak L; Berggren P-O; Radziwill M; Soszynski P
- Review: ADVANCES IN BIOLOGICAL REGULATION. 2023;87:100919Moruzzi N; Leibiger B; Barker CJ; Leibiger IB; Berggren P-O
- Article: FRONTIERS IN PLANT SCIENCE. 2022;13:1062658Akter S; Afrin S; Kim J; Kang J; Razzak MA; Berggren P-O; Hwang I
- Article: ADVANCED HEALTHCARE MATERIALS. 2022;11(21):e2200782Wang Z; Li W; Gou L; Zhou Y; Peng G; Zhang J; Liu J; Li R; Ni H; Zhang W; Cao T; Cao Q; Su H; Han Y-P; Tong N; Fu X; Ilegems E; Lu Y; Berggren P-O; Zheng X; Wang C
- Article: INTERNATIONAL JOURNAL OF MOLECULAR SCIENCES. 2022;23(20):12338Kabra UD; Moruzzi N; Berggren P-O; Jastroch M
- Article: LIFE SCIENCE ALLIANCE. 2022;5(12):e202201505Moruzzi N; Valladolid-Acebes I; Kannabiran SA; Bulgaro S; Burtscher I; Leibiger B; Leibiger IB; Berggren P-O; Brismar K
- Article: SCIENTIFIC DATA. 2022;9(1):558Hahn M; Nord C; van Krieken PP; Berggren P-O; Ilegems E; Cheddad A; Ahlgren U
- Article: JOURNAL OF CELLULAR AND MOLECULAR MEDICINE. 2022;26(18):4847-4858Wang C; Du X; Fu F; Li X; Wang Z; Zhou Y; Gou L; Li W; Li J; Zhang J; Liao G; Li L; Han Y-P; Tong N; Liu J; Chen Y; Cheng J; Cao Q; Ilegems E; Lu Y; Zheng X; Berggren P-O
- Book chapter: ELECTRON MICROSCOPY. 2022;p.Skelin Klemen M; Dolenšek J; Valladolid-Acebes I; Stožer A; Lipovšek S
- Article: NUCLEIC ACIDS RESEARCH. 2022;50(13):7783-7799Aulicino F; Pelosse M; Toelzer C; Capin J; Ilegems E; Meysami P; Rollarson R; Berggren P-O; Dillingham MS; Schaffitzel C; Saleem MA; Welsh GI; Berger I
- Article: BIOCHEMISTRY AND BIOPHYSICS REPORTS. 2022;30:101260Branstrom R; Berglund E; Frobom R; Leibiger IB; Leibiger B; Aspinwall CA; Larsson O; Berggren P-O
- Article: CELL REPORTS. 2022;39(11):110936Saghaleyni R; Malm M; Moruzzi N; Zrimec J; Razavi R; Wistbacka N; Thorell H; Pintar A; Hober A; Edfors F; Chotteau V; Berggren P-O; Grassi L; Zelezniak A; Svensson T; Hatton D; Nielsen J; Robinson JL; Rockberg J
- Article: ACS APPLIED MATERIALS & INTERFACES. 2022;14(22):25173-25182Li W; Wang C; Wang Z; Gou L; Zhou Y; Peng G; Zhu M; Zhang J; Li R; Ni H; Wu L; Zhang W; Liu J; Tian Y; Chen Z; Han Y-P; Tong N; Fu X; Zheng X; Berggren P-O
- Article: CELL CALCIUM. 2022;104:102566Troster P; Berggren P-O
- Article: CELLULAR AND MOLECULAR LIFE SCIENCES. 2022;79(6):286Shi Y; Zhao K; Yang G; Yu J; Li Y; Kessels MM; Yu L; Qualmann B; Berggren P-O; Yang S-N
- Article: MOLECULAR THERAPY. 2022;30(4):1754-1774Du W; Liu G; Shi N; Tang D; Ferdek PE; Jakubowska MA; Liu S; Zhu X; Zhang J; Yao L; Sang X; Zou S; Liu T; Mukherjee R; Criddle DN; Zheng X; Xia Q; Berggren P-O; Huang W; Sutton R; Tian Y; Huang W; Fu X
- Article: SCIENCE TRANSLATIONAL MEDICINE. 2022;14(638):eaba9112Ilegems E; Bryzgalova G; Correia J; Yesildag B; Berra E; Ruas JL; Pereira TS; Berggren P-O
- Article: NUTRIENTS. 2022;14(7):1369Rodriguez A; Korzeniowska K; Szarejko K; Borowski H; Brzezinski M; Mysliwiec M; Czupryniak L; Berggren P-O; Radziwill M; Soszynski P
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2022;119(11):e2113074119Zheng X; Ho QWC; Chua M; Stelmashenko O; Yeo XY; Muralidharan S; Torta F; Chew EGY; Lian MM; Foo JN; Jung S; Wong SH; Tan NS; Tong N; Rutter GA; Wenk MR; Silver DL; Berggren P-O; Ali Y
- Article: METABOLISM OPEN. 2022;13:100167Olesen K; Moruzzi N; Bulatovic I; Folmes C; Jeon R; Felldin U; Terzic A; Simonson OE; Le Blanc K; Osterholm C; Berggren P-O; Schiffer T; Rodin S; Tilevik A; Grinnemo K-H
- Article: CELL TRANSPLANTATION. 2022;30:9636897211066508Zhao K; Shi Y; Yu J; Yu L; MaeI A; Li Y; Kolton A; Joyce T; Odorico J; Berggren P-O; Yang S-N
- Book chapter: VOLTAGE-GATED CALCIUM CHANNELS. 2022;p. 425-448Yang S-N; Shi Y; Zhao K; Yang G; Yu J; Berggren P-O
- Article: CELL TRANSPLANTATION. 2022;31:9636897221098038Tun SBB; Chua M; Tan GSW; Leibiger I; Ali Y; Barathi VA; Berggren P-O
- Article: INTERNATIONAL JOURNAL OF MOLECULAR SCIENCES. 2021;23(1):62Recio-Lopez P; Valladolid-Acebes I; Berggren P-O; Juntti-Berggren L
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2021;581:110-117Zheng J; Chen X; Wu L; Zhou Y; Wang Z; Li J; Liu Y; Peng G; Berggren P-O; Zheng X; Tong N
- Preprint: RESEARCH SQUARE. 2021Shi Y; Zhao K; Yang G; Yu J; Li Y; Kessels MM; Yu L; Qualmann B; Berggren P-O; Yang S-N
- Article: ACS NANO. 2021;15(11):18237-18249Li W; Wang C; Lv H; Wang Z; Zhao M; Liu S; Gou L; Zhou Y; Li J; Zhang J; Li L; Wang Y; Lou P; Wu L; Zhou L; Chen Y; Lu Y; Cheng J; Han Y-P; Cao Q; Huang W; Tong N; Fu X; Liu J; Zheng X; Berggren P-O
- Article: NATURE COMMUNICATIONS. 2021;12(1):6558Sabatier P; Beusch CM; Saei AA; Aoun M; Moruzzi N; Coelho A; Leijten N; Nordenskjold M; Micke P; Maltseva D; Tonevitsky AG; Millischer V; Villaescusa JC; Kadekar S; Gaetani M; Altynbekova K; Kel A; Berggren P-O; Simonson O; Grinnemo K-H; Holmdahl R; Rodin S; Zubarev RA
- Article: SCIENTIFIC REPORTS. 2021;11(1):21539Expression of truncated Kir6.2 promotes insertion of functionally inverted ATP-sensitive K+ channelsHeitz BA; Branstrom R; Yang W; Huang Y; Moede T; Leibiger IB; Leibiger B; Chen LQ; Yu J; Yang S-N; Larsson O; Saavedra SS; Berggren P-O; Aspinwall CA
- Editorial: CELL TRANSPLANTATION. 2021;30:9636897211045320Abdulreda MH; Berggren P-O
- Journal article: HUMAN CELL. 2021;34(5):1289-1300Hossein-Khannazer N; Torabi S; Hosseinzadeh R; Shahrokh S; Aghdaei HA; Memarnejadian A; Kadri N; Vosough M
- Conference publication: DIABETOLOGIA. 2021;64(SUPPL 1):96-97Recio-Lopez P; Berggren P-O; Juntti-Berggren L; Valladolid-Acebes I
- Article: SCIENCE ADVANCES. 2021;7(30):eabg5733Adori C; Daraio T; Kuiper R; Barde S; Horvathova L; Yoshitake T; Ihnatko R; Valladolid-Acebes I; Vercruysse P; Wellendorf AM; Gramignoli R; Bozoky B; Kehr J; Theodorsson E; Cancelas JA; Mravec B; Jorns C; Ellis E; Mulder J; Uhlen M; Bark C; Hokfelt T
- Article: ANTIOXIDANTS. 2021;10(7):1070Dallner G; Bentinger M; Hussain S; Sinha I; Yang J; Schwank-Xu C; Zheng X; Swiezewska E; Brismar K; Valladolid-Acebes I; Tekle M
- Article: DIABETOLOGIA. 2021;64(7):1626-1641Korpos E; Kadri N; Loismann S; Findeisen CR; Arfuso F; Burke GWIII; Richardson SJ; Morgan NG; Bogdani M; Pugliese A; Sorokin L
- Article: NUTRIENTS. 2021;13(7):2281Gu W; Anker CCB; Christiansen CB; Moede T; Berggren P-O; Hermansen K; Gregersen S; Jeppesen PB
- Article: METABOLITES. 2021;11(6):387Leibiger B; Moede T; Valladolid-Acebes I; Paschen M; Visa M; Leibiger IB; Berggren P-O
- Article: PRIMARY CARE DIABETES. 2021;15(3):522-527Taloyan M; Momtaz S; Steiner K; Ostenson C-G; Salminen H
- Article: JOURNAL OF CELLULAR AND MOLECULAR MEDICINE. 2021;25(10):4800-4813Moruzzi N; Lazzeri-Barcelo F; Valladolid-Acebes I; Moede T; Paschen M; Leibiger B; Berggren P-O; Leibiger IB
- Review: FRONTIERS IN ENDOCRINOLOGY. 2021;12:652853Ilegems E; Berggren P-O
- Article: LIFE SCIENCE ALLIANCE. 2021;4(4):e202000723Luu TT; Schmied L; Nguyen N-A; Wiel C; Meinke S; Mohammad DK; Bergo M; Alici E; Kadri N; Ganesan S; Hoglund P
- Article: SCIENCE ADVANCES. 2021;7(11):eabc2931Valladolid-Acebes I; Avall K; Recio-Lopez P; Moruzzi N; Bryzgalova G; Bjornholm M; Krook A; Alonso EF; Ericsson M; Landfors F; Nilsson SK; Berggren P-O; Juntti-Berggren L
- Article: INTERNATIONAL JOURNAL OF MOLECULAR SCIENCES. 2021;22(4):1813Mir-Coll J; Moede T; Paschen M; Neelakandhan A; Valladolid-Acebes I; Leibiger B; Biernath A; Ammala C; Leibiger IB; Yesildag B; Berggren P-O
- Review: INTERNATIONAL JOURNAL OF MOLECULAR SCIENCES. 2021;22(2):932Valladolid-Acebes I; Berggren P-O; Juntti-Berggren L
- Article: CANCERS. 2021;13(2):298Wagner AK; Gehrmann U; Hiltbrunner S; Carannante V; Luu TT; Naslund TI; Brauner H; Kadri N; Karre K; Gabrielsson S
- Article: CELLR4. 2021;9:e3093Abdulreda MH; Berggren PO
- Article: DIABETES. 2021;70(1):111-118Barker CJ; Tessaro FHG; Ferreira SDS; Simas R; Ayala TS; Kohler M; Rajasekaran SS; Martins JO; Dare E; Berggren P-O
- Article: GLOBAL HEALTH ACTION. 2020;13(1):1795439Timm L; Harcke K; Karlsson I; Sidney Annerstedt K; Alvesson HM; Stattin NS; Forsberg BC; ostenson C-G; Daivadanam M
- Article: SCIENTIFIC REPORTS. 2020;10(1):20145Glucokinase intrinsically regulates glucose sensing and glucagon secretion in pancreatic alpha cellsMoede T; Leibiger B; Sanchez PV; Dare E; Kohler M; Muhandiramlage TP; Leibiger IB; Berggren P-O
- Article: ELIFE. 2020;9:e56914Xiong Y; Scerbo MJ; Seelig A; Volta F; O'Brien N; Dicker A; Padula D; Lickert H; Gerdes JM; Berggren P-O
- Article: VIRAL IMMUNOLOGY. 2020;33(9):594-599Niklasson B; Klitz W; Juntti-Berggren L; Berggren P-O; Lindquist L
- Article: STEM CELLS TRANSLATIONAL MEDICINE. 2020;9(10):1190-1202Boberg E; von Bahr L; Afram G; Lindstrom C; Ljungman P; Heldring N; Petzelbauer P; Legert KG; Kadri N; Le Blanc K
- Article: JOURNAL OF THROMBOSIS AND HAEMOSTASIS. 2020;18(10):2685-2700Tan S; Li S; Min Y; Gistera A; Moruzzi N; Zhang J; Sun Y; Andersson J; Malmstrom RE; Wang M; Berggren P-O; Schlisio S; Liao W; Ketelhuth DFJ; Ma C; Li N
- Review: DIABETOLOGIA. 2020;63(10):2064-2075Moede T; Leibiger IB; Berggren P-O
- Article: TRANSPLANTATION. 2020;104(10):2048-2058Yao M; Watanabe M; Sun S; Tokodai K; Cerami A; Brines M; Ostenson C-G; Ericzon B-G; Lundgren T; Kumagai-Braesch M
- Article: COMMUNICATIONS BIOLOGY. 2020;3(1):541Hahn M; van Krieken PP; Nord C; Alanentalo T; Morini F; Xiong Y; Eriksson M; Mayer J; Kostromina E; Ruas JL; Sharpe J; Pereira T; Berggren P-O; Ilegems E; Ahlgren U
- Article: ADVANCED SCIENCE. 2020;7(15):2000248Oliva-Vilarnau N; Vorrink SU; Ingelman-Sundberg M; Lauschke VM
- Article: DIABETOLOGIA. 2020;63(8):1603-1615Kalinovich A; Dehvari N; Aslund A; van Beek S; Halleskog C; Olsen J; Forsberg E; Zacharewicz E; Schaart G; Rinde M; Sandstrom A; Berlin R; Ostenson C-G; Hoeks J; Bengtsson T
- Article: ELIFE. 2020;9:e58065Chitirala P; Chang H-F; Martzloff P; Harenberg C; Ravichandran K; Abdulreda MH; Berggren P-O; Krause E; Schirra C; Leinders-Zufall T; Benseler F; Brose N; Rettig J
- Article: NEUROPHARMACOLOGY. 2020;170:108070Narvaez M; Andrade-Talavera Y; Valladolid-Acebes I; Fredriksson M; Siegele P; Hernandez-Sosa A; Fisahn A; Fuxe K; Borroto-Escuela DO
- Article: CELL REPORTS. 2020;31(9):107699Osman AM; Sun Y; Burns TC; He L; Kee N; Oliva-Vilarnau N; Alevyzaki A; Zhou K; Louhivuori L; Uhlen P; Hedlund E; Betsholtz C; Lauschke VM; Kele J; Blomgren K
- Letter: HAEMATOLOGICA. 2020;105(6):E310-E314Boberg E; Kadri N; Winterling J; Davies LC; Bjorklund A; Msghina M; Iacobaeus E; Le Blanc K
- Article: PLOS PATHOGENS. 2020;16(5):e1008244Duru AD; Sun R; Allerbring EB; Chadderton J; Kadri N; Han X; Peqini K; Uchtenhagen H; Madhurantakam C; Pellegrino S; Sandalova T; Nygren P-A; Turner SJ; Achour A
- Article: SCANDINAVIAN JOURNAL OF CLINICAL & LABORATORY INVESTIGATION. 2020;80(3):230-235Sandin A; Olofsson C; Stralfors A; Ekberg NR; Brismar K; Juntti-Berggren L; Eggertsen G
- Article: EUROPEAN JOURNAL OF IMMUNOLOGY. 2020;50(4):494-504Luu TT; Wagner AK; Schmied L; Meinke S; Freund JE; Kambayashi T; Ravens I; Achour A; Bernhardt G; Chambers BJ; Hoglund P; Kadri N
- Article: NANO LETTERS. 2020;20(3):1517-1525Kim J; Kim J; Ku M; Cha E; Ju S; Park WY; Kim KH; Kim DW; Berggren P-O; Park J-U
- Article: JCI INSIGHT. 2020;5(3):129694Liang T; Qin T; Kang F; Kang Y; Xie L; Zhu D; Dolai S; Greitzer-Antes D; Baker RK; Feng D; Tuduri E; Ostenson C-G; Kieffer TJ; Banks K; Pessin JE; Gaisano HY
- Article: ACS BIOMATERIALS SCIENCE & ENGINEERING. 2020;6(2):1186-1195Johansson U; Shalaly ND; Hjelm LC; Ria M; Berggren P-O; Hedhammar M
- Article: ROYAL SOCIETY OPEN SCIENCE. 2020;7(1):191171Grubelnik V; Markovic R; Lipovsek S; Leitinger G; Gosak M; Dolensek J; Valladolid-Acebes I; Berggren P-O; Stozer A; Perc M; Marhl M
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2020;117(1):448-453Yu J; Shi Y; Zhao K; Yang G; Yu L; Li Y; Andersson E-M; Ammala C; Yang S-N; Berggren P-O
- Article: PEDIATRIC ENDOCRINOLOGY, DIABETES, AND METABOLISM. 2020;26(1):22-26Brzeziński M; Korzeniowska K; Szarejko K; Radziwiłł M; Myśliwiec M; Anyszek T; Czupryniak L; Berggren P-O; Soszyński P
- Article: FASEB JOURNAL. 2020;34(1):945-959Jacob S; Kohler M; Troster P; Visa M; Garcia-Prieto CF; Alanentalo T; Moede T; Leibiger B; Leibiger IB; Berggren P-O
- Article: CELL TRANSPLANTATION. 2020;29:963689720913256-096368972091325Tun SBB; Chua M; Hasan R; Kohler M; Zheng X; Ali Y; Abdulreda MH; Juntti-Berggren L; Barathi VA; Berggren P-O
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2019;116(52):26816-26822Shi Y; Park KS; Kim SH; Yu J; Zhao K; Yu L; Oh KW; Lee K; Kim J; Chaggar K; Li Y; Dolphin AC; Catterall WA; Ryu SH; Yang S-N; Berggren P-O
- Article: BMJ OPEN. 2019;9(12):e028757Taloyan M; Amri A; Steiner KH; Lamian F; Ostenson C-G; Salminen H
- Article: JOURNAL OF CLINICAL MEDICINE. 2019;8(12):E2102-2102Iacobaeus E; Kadri N; Lefsihane K; Boberg E; Gavin C; Andren AT; Lilja A; Brundin L; Le Blanc K
- Review: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2019;317(6):E1108-E1120Chittezhath M; Gunaseelan D; Zheng X; Hasan R; Tay VSY; Lim ST; Wang X; Berggren P-O; Bornstein S; Boehm B; Ruedl C; Ali Y
- Article: NUTRIENTS. 2019;11(11):E2658-2658Timm L; Daivadanam M; Lager A; Forsberg B; Ostenson C-G; Alvesson HM
- Review: NEUROENDOCRINOLOGY LETTERS. 2019;40(4):175-183Juhlin CC; Skoglund S; Juntti-Berggren L; Karlberg M; Calissendorff J
- Article: NATURE METABOLISM. 2019;1(11):1110-1126Menegaz D; Hagan DW; Almaca J; Cianciaruso C; Rodriguez-Diaz R; Molina J; Dolan RM; Becker MW; Schwalie PC; Nano R; Lebreton F; Kang C; Sah R; Gaisano HY; Berggren P-O; Baekkeskov S; Caicedo A; Phelps EA
- Article: SCIENTIFIC REPORTS. 2019;9(1):14636Balan M; Trusohamn M; Ning FC; Jacob S; Pietras K; Eriksson U; Berggren P-O; Nyqvist D
- Article: FASEB JOURNAL. 2019;33(10):10668-10679Hwang H-J; Yang YR; Kim HY; Choi Y; Park K-S; Lee H; Ma JS; Yamamoto M; Kim J; Chae YC; Choi JH; Cocco L; Berggren P-O; Jang H-J; Suh P-G
- Article: FRONTIERS IN IMMUNOLOGY. 2019;10:2249Gavin C; Meinke S; Heldring N; Heck KA; Achour A; Iacobaeus E; Le Blanc K; Kadri N; Hoglund P
- Corrigendum: NATURE COMMUNICATIONS. 2019;10(1):4218Arrojo e Drigo R; Jacob S; Garcia-Prieto CF; Zheng X; Fukuda M; Hoa TTN; Stelmashenko O; Martins Pecanha FL; Rodriguez-Diaz R; Bushong E; Deerinck T; Phan S; Ali Y; Leibiger I; Chua M; Boudier T; Song S-H; Graf M; Augustine GJ; Ellisman MH; Berggren P-O
- Conference publication: DIABETOLOGIA. 2019;62:S122Moruzzi N; Valladolid-Acebes I; Paschen M; Moede T; Leibiger B; Berggren P-O; Leibiger IB
- Conference publication: DIABETOLOGIA. 2019;62:S71-S72Valladolid-Acebes I; Recio-Lopez P; Berggren P-O; Juntti-Berggren L
- Conference publication: DIABETOLOGIA. 2019;62:S293-S294Recio-Lopez P; Berggren P-O; Juntti-Berggren L; Valladolid-Acebes I
- Article: NATURE COMMUNICATIONS. 2019;10(1):3700Arrojo e Drigo R; Jacob S; Garcia-Prieto CF; Zheng X; Fukuda M; Hoa TTN; Stelmashenko O; Martins Pecanha FL; Rodriguez-Diaz R; Bushong E; Deerinck T; Phan S; Ali Y; Leibiger I; Chua M; Boudier T; Song S-H; Graf M; Augustine GJ; Ellisman MH; Berggren P-O
- Article: CURRENT BIOLOGY. 2019;29(15):2477-2487.e6Cederroth CR; Park J-S; Basinou V; Weger BD; Tserga E; Sarlus H; Magnusson AK; Kadri N; Gachon F; Canton B
- Corrigendum: DIABETOLOGIA. 2019;62(8):1517Abdulreda MH; Molano RD; Faleo G; Lopez-Cabezas M; Shishido A; Ulissi U; Fotino C; Hernandez LF; Tschiggfrie A; Aldrich VR; Tamayo-Garcia A; Bayer AS; Ricordi C; Caicedo A; Buchwald P; Pileggi A; Berggren P-O
- Article: DIABETOLOGIA. 2019;62(7):1237-1250Abdulreda MH; Molano RD; Faleo G; Lopez-Cabezas M; Shishido A; Ulissi U; Fotino C; Hernandez LF; Tschiggfrie A; Aldrich VR; Tamayo-Garcia A; Bayer AS; Ricordi C; Caicedo A; Buchwald P; Pileggi A; Berggren P-O
- Article: JOURNAL OF ULTRASOUND IN MEDICINE. 2019;38(7):1723-1731Rohl S; Eriksson L; Saxelin R; Lengquist M; Ostenson C-G; Hedin U; Caidahl K; Razuvaev A
- Article: EBIOMEDICINE. 2019;45:529-541van Krieken PP; Voznesenskaya A; Dicker A; Xiong Y; Park JH; Lee JI; Ilegems E; Berggren P-O
- Article: NATURE COMMUNICATIONS. 2019;10(1):2692Mitrofanova A; Mallela SK; Ducasa GM; Yoo TH; Rosenfeld-Gur E; Zelnik ID; Molina J; Santos JV; Ge M; Sloan A; Kim JJ; Pedigo C; Bryn J; Volosenco I; Faul C; Zeidan YH; Hernandez CG; Mendez AJ; Leibiger I; Burke GW; Futerman AH; Barisoni L; Ishimoto Y; Inagi R; Merscher S; Fornoni A
- Article: CELLULAR SIGNALLING. 2019;58:131-136Kim J; Dare E; Rajasekaran SS; Ryu SH; Berggren P-O; Barker CJ
- Article: SCANDINAVIAN JOURNAL OF PUBLIC HEALTH. 2019;47(4):408-416Agardh EE; Lundin A; Lager A; Allebeck P; Koupil I; Andreasson S; Ostenson C-G; Danielsson A-K
- Article: LAKARTIDNINGEN. 2019;116:FL3MÖstenson C-G
- Article: DIABETOLOGIA. 2019;62(5):811-821Abdulreda MH; Berman DM; Shishido A; Martin C; Hossameldin M; Tschiggfrie A; Hernandez LF; Hernandez A; Ricordi C; Parel J-M; Jankowska-Gan E; Burlingham WJ; Arrieta-Quintero EA; Perez VL; Kenyon NS; Berggren P-O
- Review: PHARMACOLOGY & THERAPEUTICS. 2019;197:103-121Yang S-N; Berggren P-O
- Article: SCIENTIFIC REPORTS. 2019;9(1):6291Johansson U; Widhe M; Shalaly ND; Arregui IL; Nileback L; Tasiopoulos CP; Astrand C; Berggren P-O; Gasser C; Hedhammar M
- Article: ELIFE. 2019;8:e41608Pernemalm M; Sandberg A; Zhu Y; Boekel J; Tamburro D; Schwenk JM; Bjork A; Wahren-Herlenius M; Amark H; Ostenson C-G; Westgren M; Lehtio J
- Article: PLATELETS. 2019;30(3):348-355Spectre G; Mobarrez F; Stalesen R; Ostenson C-G; Varon D; Wallen H; Hjemdahl P
- Article: OCCUPATIONAL AND ENVIRONMENTAL MEDICINE. 2019;76(4):201-207Pyko A; Andersson N; Eriksson C; de Faire U; Lind T; Mitkovskaya N; Ogren M; Ostenson C-G; Pedersen NL; Rizzuto D; Wallas AK; Pershagen G
- Article: PLOS ONE. 2019;14(3):e0213530De Man J; Aweko J; Daivadanam M; Alvesson HM; Delobelle P; Mayega RW; Ostenson C-G; Kirunda B; Kasujja FX; Guwattude D; Puoane T; Sanders D; Peterson S; Tomson G; Sundberg CJ; Absetz P; Van Olmen J
- Article: SCIENTIFIC REPORTS. 2019;9(1):3918Fan Y; Zheng X; Ali Y; Berggrenvo P-O; Loo SCJ
- Study protocol: BMC PUBLIC HEALTH. 2019;19(1):273Bonn SE; Lof M; Ostenson C-G; Lagerros YT
- Article: EUROPEAN JOURNAL OF IMMUNOLOGY. 2019;49(3):443-453Tripathi P; Sedimbi SK; Singh AK; Lofboml L; Issazadeh-Navikas S; Weiss S; Foerster I; Karlsson MCI; Yrlid U; Kadri N; Cardell SL
- Article: EUROPEAN JOURNAL OF IMMUNOLOGY. 2019;49(2):313-322miR-31 regulates energy metabolism and is suppressed in Tcells from patients with Sjogren's syndromeJohansson A; Nyberg WA; Sjostrand M; Moruzzi N; Bergman P; Khademi M; Andersson M; Piehl F; Berggren P-O; Covacu R; Jagodic M; Espinosa A
- Article: SCIENTIFIC REPORTS. 2019;9(1):627Lundqvist LCE; Rattigan D; Ehtesham E; Demmou C; Ostenson C-G; Sandstrom C
- Corrigendum: CELLULAR SIGNALLING. 2019;53:415Rajasekaran SS; Kim J; Gaboardi G-C; Gromada J; Shears SB; dos Santos KT; Nolasco EL; Ferreira SDS; Illies C; Kohler M; Gu C; Ryu SH; Martins JO; Dare E; Barker CJ; Berggren P-O
- Book chapter: CO-IMMUNOPRECIPITATION METHODS FOR BRAIN TISSUE: NEUROMETHODS. 2019;p. 123-135Borroto-Escuela DO; Narvaez M; Zannoni M; Contri C; Crespo-Ramírez M; di Palma M; Ambrogini P; Borroto-Escuela DY; Brito I; Pita-Rodríguez M; Valladolid-Acebes I; de la Mora MP; Fuxe K
- Article: GLOBAL HEALTH ACTION. 2019;12(1):1609313Al-Murani F; Aweko J; Nordin I; Delobelle P; Kasujja F; Ostenson C-G; Peterson SS; Daivadanam M; Alvesson HM
- Article: FASEB JOURNAL. 2019;33(1):204-218Paschen M; Moede T; Valladolid-Acebes I; Leibiger B; Moruzzi N; Jacob S; Garcia-Prieto CF; Brismar K; Leibiger IB; Berggren P-O
- Article: ENDOKRYNOLOGIA POLSKA. 2019;70(2):172-178Korzeniowska KA; Brzezinski M; Szarejko K; Radziwill M; Anyszek T; Czupryniak L; Soszynski P; Berggren P-O; Mysliwiec M
- Article: DIABETOLOGIA. 2019;62(1):123-135Sevillano J; Gracia Sanchez-Alonso M; Zapateria B; Calderon M; Alcala M; Limones M; Pita J; Gramage E; Vicente-Rodriguez M; Horrillo D; Medina-Gomez G; Jesus Obregon M; Viana M; Valladolid-Acebes I; Herradon G; Pilar Ramos-Alvarez M
- Article: BMC PUBLIC HEALTH. 2018;18(1):1409Mayega RW; Ekirapa E; Kirunda B; Nalwadda C; Aweko J; Tomson G; Ostenson CG; Van Olmen J; Daivadanam M; Kiguli J
- Article: INTERNATIONAL JOURNAL OF MOLECULAR SCIENCES. 2018;19(12):E3942-3942Mahdi A; Jiao T; Tratsiakovich Y; Yang J; Ostenson C-G; Pernow J; Zhou Z
- Article: JOURNAL OF BIOPHOTONICS. 2018;11(12):e201800206Kohler M; Paulson B; Kim Y; Lee S; Dicker A; van Krieken P; Kim JY; Pack C-G; Joo J; Berggren P-O; Kim JK
- Article: ENVIRONMENT INTERNATIONAL. 2018;121(Pt 1):832-841Persson A; Pyko A; Lind T; Bellander T; Ostenson C-G; Pershagen G; Eriksson C; Lohmus M
- Article: ENVIRONMENT INTERNATIONAL. 2018;120:163-171Weinmayr G; Pedersen M; Stafoggia M; Andersen ZJ; Galassi C; Munkenast J; Jaensch A; Oftedal B; Krog NH; Aamodt G; Pyko A; Pershagen G; Korek M; De Faire U; Pedersen NL; Ostenson C-G; Rizzuto D; Sorensen M; Tjonneland A; Bueno-de-Mesquita B; Vermeulen R; Eeftens M; Concin H; Lang A; Wang M; Tsai M-Y; Ricceri F; Sacerdote C; Ranzi A; Cesaroni G; Forastiere F; de Hoogh K; Beelen R; Vineis P; Kooter I; Sokhi R; Brunekreef B; Hoek G; Raaschou-Nielsen O; Nagel G
- Article: INTERNATIONAL JOURNAL OF CANCER. 2018;143(7):1632-1643Nagel G; Stafoggia M; Pedersen M; Andersen ZJ; Galassi C; Munkenast J; Jaensch A; Sommar J; Forsberg B; Olsson D; Oftedal B; Krog NH; Aamodt G; Pyko A; Pershagen G; Korek M; De Faire U; Pedersen NL; Ostenson C-G; Fratiglioni L; Sorensen M; Tjonneland A; Peeters PH; Bueno-de-Mesquita B; Vermeulen R; Eeftens M; Plusquin M; Key TJ; Concin H; Lang A; Wang M; Tsai M-Y; Grioni S; Marcon A; Krogh V; Ricceri F; Sacerdote C; Ranzi A; Cesaroni G; Forastiere F; Tamayo-Uria I; Amiano P; Dorronsoro M; de Hoogh K; Beelen R; Vineis P; Brunekreef B; Hoek G; Raaschou-Nielsen O; Weinmayr G
- Article: LABORATORY ANIMAL RESEARCH. 2018;34(3):126-131Åvall K; Berggren P-O; Juntti-Berggren L
- Article: INTERNATIONAL JOURNAL OF HYGIENE AND ENVIRONMENTAL HEALTH. 2018;221(8):1133-1141Pyko A; Lind T; Mitkovskaya N; Ogren M; Ostenson C-G; Wallas A; Pershagen G; Eriksson C
- Article: IMMUNOLOGY AND CELL BIOLOGY. 2018;96(8):820-830Iacobaeus E; Douagi I; Jitschin R; Marcusson-Stahl M; Andren AT; Gavin C; Lefsihane K; Davies LC; Mougiakakos D; Kadri N; Le Blanc K
- Article: INTERNATIONAL JOURNAL OF ENVIRONMENTAL RESEARCH AND PUBLIC HEALTH. 2018;15(9):E1810-1810Aweko J; De Man J; Absetz P; Ostenson C-G; Peterson SS; Alvesson HM; Daivadanam M
- Article: JACC-JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY. 2018;72(7):769-780Zhou Z; Mahdi A; Tratsiakovich Y; Zahoran S; Kovamees O; Nordin F; Gonzalez AEU; Alvarsson M; Ostenson C-G; Andersson DC; Hedin U; Hermesz E; Lundberg JO; Yang J; Pernow J
- Article: JOVE-JOURNAL OF VISUALIZED EXPERIMENTS. 2018;(138)Zheng X; Ho CQW; Zheng X; Lee KL; Gradin K; Pereira TS; Berggren P-O; Ali Y
- Article: SCIENTIFIC REPORTS. 2018;8(1):11202Heshmati Y; Kharazi S; Turkoz G; Chang D; Dolatabadi EK; Bostrom J; Krstic A; Boukoura T; Wagner E; Kadri N; Mansson R; Altun M; Qian H; Walfridsson J
- Article: CELL REPORTS. 2018;24(4):922-934Lee K; Kim J; Kohler M; Yu J; Shi Y; Yang S-N; Ryu SH; Berggren P-O
- Article: NUTRIENTS. 2018;10(7):E933-933Zambrana S; Lundqvist LCE; Mamani O; Catrina S-B; Gonzales E; Ostenson C-G
- Review: FRONTIERS IN MEDICINE. 2018;5:192Oliva-Vilarnau N; Hankeova S; Vorrink SU; Mkrtchian S; Andersson ER; Lauschke VM
- Article: JOURNAL OF ETHNOPHARMACOLOGY. 2018;220:155-158Mohanty S; Zambrana S; Dieulouard S; Kamolvit W; Nilsen V; Gonzales E; Ostenson C-G; Brauner A
- Article: CELLULAR SIGNALLING. 2018;46:120-128Rajasekaran SS; Kim J; Gaboardi G-C; Gromada J; Shears SB; dos Santos KT; Nolasco EL; Ferreira SDS; Illies C; Kohler M; Gu C; Ryu SH; Martins JO; Dare E; Barker CJ; Berggren P-O
- Letter: DIABETES & METABOLISM. 2018;44(3):303-304Avall K; Berggren P-O; Juntti-Berggren L
- Article: NEUROSCIENCE LETTERS. 2018;674:75-80Daraio T; Valladolid-Acebes I; Brismar K; Bark C
- Review: PHARMACOLOGY & THERAPEUTICS. 2018;185:86-98Yunn N-O; Kim J; Kim Y; Leibiger I; Berggren P-O; Ryu SH
- Article: BMJ OPEN. 2018;8(3):e019981Guwatudde D; Absetz P; Delobelle P; Ostenson C-G; Van JO; Alvesson HM; Mayega RW; Kiracho EE; Kiguli J; Sundberg CJ; Sanders D; Tomson G; Puoane T; Peterson S; Daivadanam M
- Article: CELL METABOLISM. 2018;27(3):549-558.e4Rodriguez-Diaz R; Molano RD; Weitz JR; Abdulreda MH; Berman DM; Leibiger B; Leibiger IB; Kenyon NS; Ricordi C; Pileggi A; Caicedo A; Berggren P-O
- Article: ACTA NEUROPATHOLOGICA COMMUNICATIONS. 2018;6(1):14Lietzau G; Davidsson W; Ostenson C-G; Chiazza F; Nathanson D; Pintana H; Skogsberg J; Klein T; Nystrom T; Darsalia V; Patrone C
- Article: NEURO-ONCOLOGY. 2018;20(3):420-432Andersen ZJ; Pedersen M; Weinmayr G; Stafoggia M; Galassi C; Jorgensen JT; Sommar JN; Forsberg B; Olsson D; Oftedal B; Aasvang GM; Schwarze P; Pyko A; Pershagen G; Korek M; Faire UD; Ostenson C-G; Fratiglioni L; Eriksen KT; Poulsen AH; Tjonneland A; Brauner EV; Peeters PH; Bueno-de-Mesquita B; Jaensch A; Nagel G; Lang A; Wang M; Tsai M-Y; Grioni S; Marcon A; Krogh V; Ricceri F; Sacerdote C; Migliore E; Vermeulen R; Sokhi R; Keuken M; de Hoogh K; Beelen R; Vineis P; Cesaroni G; Brunekreef B; Hoek G; Raaschou-Nielsen O
- Article: CELL METABOLISM. 2018;27(2):378-392.e5Agudelo LZ; Ferreira DMS; Cervenka I; Bryzgalova G; Dadvar S; Jannig PR; Pettersson-Klein AT; Lakshmikanth T; Sustarsic EG; Porsmyr-Palmertz M; Correia JC; Izadi M; Martinez-Redondo V; Ueland PM; Midttun O; Gerhart-Hines Z; Brodin P; Pereira T; Berggren P-O; Ruas JL
- Article: NUTRIENTS. 2018;10(1):E94-94Zambrana S; Lundqvist LCE; Veliz V; Catrina S-B; Gonzales E; Ostenson C-G
- Article: BMC PUBLIC HEALTH. 2018;18(1):119Bonn SE; Alexandrou C; Steiner KH; Wiklander K; Ostenson C-G; Lof M; Lagerros YT
- Article: FRONTIERS IN MOLECULAR NEUROSCIENCE. 2018;11:19Tröster P; Haseleu J; Petersen J; Drees O; Schmidtko A; Schwaller F; Lewin GR; Ter-Avetisyan G; Winter Y; Peters S; Feil S; Feil R; Rathjen FG; Schmidt H
- Book chapter: RECEPTOR-RECEPTOR INTERACTIONS IN THE CENTRAL NERVOUS SYSTEM: NEUROMETHODS. 2018;p. 15-23Narvaez M; Corrales F; Brito I; Valladolid-Acebes I; Fuxe K; Borroto-Escuela DO
- Book chapter: RECEPTOR-RECEPTOR INTERACTIONS IN THE CENTRAL NERVOUS SYSTEM: NEUROMETHODS. 2018;p. 299-315Borroto-Escuela DO; Narvaez M; Valladolid-Acebes I; Shumilov K; Di Palma M; Wydra K; Schaefer T; Reyes-Resina I; Navarro G; Mudó G; Filip M; Sartini S; Friedland K; Schellekens H; Beggiato S; Ferraro L; Tanganelli S; Franco R; Belluardo N; Ambrogini P; Pérez de la Mora M; Fuxe K
- Book chapter: RECEPTOR-RECEPTOR INTERACTIONS IN THE CENTRAL NERVOUS SYSTEM: NEUROMETHODS. 2018;p. 283-298Brito I; Narvaez M; Savelli D; Shumilov K; Di Palma M; Sartini S; Skieterska K; Van Craenenbroeck K; Valladolid-Acebes I; Zaldivar-Oro R; Filip M; Cuppini R; Rivera A; Liu F; Ambrogini P; Pérez de la Mora M; Fuxe K; Borroto-Escuela DO
- Article: BMJ GLOBAL HEALTH. 2018;3(6):e001068van Olmen J; Delobelle P; Guwatudde D; Absetz P; Sanders D; Alvesson HM; Puoane T; Ostenson C-G; Tomson G; Mayega RW; Sundberg CJ; Peterson S; Daivadanam M
- Article: EVIDENCE-BASED COMPLEMENTARY AND ALTERNATIVE MEDICINE. 2018;2018:1098015Kamolvit W; Nilsen V; Zambrana S; Mohanty S; Gonzales E; Ostenson C-G; Brauner A
- Article: EUROPEAN UROLOGY FOCUS. 2018;4(1):113-120Pedersen M; Stafoggia M; Weinmayr G; Andersen ZJ; Galassi C; Sommar J; Forsberg B; Olsson D; Oftedal B; Krog NH; Aamodt G; Pyko A; Pershagen G; Korek M; De Faire U; Pedersen NL; Odiaeresi C-G; Fratiglioni L; Sorensen M; Eriksen KT; Tjonneland A; Peeters PH; Bueno-de-Mesquita B; Vermeulen R; Eeftens M; Plusquin M; Key TJ; Jaensch A; Nagel G; Concin H; Wang M; Tsai M-Y; Grioni S; Marcon A; Krogh V; Ricceri F; Sacerdote C; Ranzi A; Cesaroni G; Forastiere F; Tamayo I; Amiano P; Dorronsoro M; Stayner LT; Kogevinas M; Nieuwenhuijsen MJ; Sokhi R; de Hoogh K; Beelen R; Vineis P; Brunekreef B; Hoek G; Raaschou-Nielsen O
- Article: CELLULAR SIGNALLING. 2018;42:127-133Rajasekaran SS; Illies C; Shears SB; Wang H; Ayala TS; Martins JO; Dare E; Berggren P-O; Barker CJ
- Article: SCIENTIFIC REPORTS. 2017;7(1):17777Jeong H; Koh A; Lee J; Park D; Lee JO; Lee MN; Jo K-J; Huynh NKT; Kim E; Min B-S; Kim HS; Berggren P-O; Ryu SH
- Article: CELL REPORTS. 2017;21(10):2748-2759Park SJ; Lee SB; Suh Y; Kim S-J; Lee N; Hong J-H; Park C; Woo Y; Ishizuka K; Kim J-H; Berggren P-O; Sawa A; Park SK
- Article: PSYCHIATRIC GENETICS. 2017;27(6):210-218Hukic DS; Osby U; Olsson E; Hilding A; Ostenson C-G; Gu HF; Ehrenborg E; Edman G; Schalling M; Lavebratt C; Frisen L
- Article: JOURNAL OF DIABETES. 2017;9(12):1082-1090Elrayah-Eliadarous HA; Ostenson C-G; Eltom M; Johansson P; Sparring V; Wahlstrom R
- Article: FASEB JOURNAL. 2017;31(12):5296-5306Montane J; de Pablo S; Castaño C; Rodríguez-Comas J; Cadavez L; Obach M; Visa M; Alcarraz-Vizán G; Sanchez-Martinez M; Nonell-Canals A; Parrizas M; Servitja J-M; Novials A
- Article: ENVIRONMENTAL HEALTH PERSPECTIVES. 2017;125(11):117005Pyko A; Eriksson C; Lind T; Mitkovskaya N; Wallas A; Ogren M; Ostenson C-G; Pershagen G
- Article: ENVIRONMENTAL HEALTH PERSPECTIVES. 2017;125(10):107005Andersen ZJ; Stafoggia M; Weinmayr G; Pedersen M; Galassi C; Jorgensen JT; Oudin A; Forsberg B; Olsson D; Oftedal B; Aasvang GM; Aamodt G; Pyko A; Pershagen G; Korek M; De Faire U; Pedersen NL; Ostenson C-G; Fratiglioni L; Eriksen KT; Tjonneland A; Peeters PH; Bueno-de-Mesquita B; Plusquin M; Key TJ; Jaensch A; Nagel G; Lang A; Wang M; Tsai M-Y; Fournier A; Boutron-Ruault M-C; Baglietto L; Grioni S; Marcon A; Krogh V; Ricceri F; Sacerdote C; Migliore E; Tamayo-Uria I; Amiano P; Dorronsoro M; Vermeulen R; Sokhi R; Keuken M; de Hoogh K; Beelen R; Vineis P; Cesaroni G; Brunekreef B; Hoek G; Raaschou-Nielsen O
- Article: INFECTION AND IMMUNITY. 2017;85(10):e00069-e00017Sultana MA; Du A; Carow B; Angbjar CM; Weidner JM; Kanatani S; Fuks JM; Muliaditan T; James J; Mansfield IO; Campbell TM; Liu L; Kadri N; Lambert H; Barragan A; Chambers BJ
- Article: AAPS PHARMSCITECH. 2017;18(7):2648-2657Fan YL; Hou HW; Tay HM; Guo WM; Berggren P-O; Loo SCJ
- Article: SCIENTIFIC REPORTS. 2017;7(1):12440van Krieken PP; Dicker A; Eriksson M; Herrera PL; Ahlgren U; Berggren P-O; Ilegems E
- Review: MOLECULAR METABOLISM. 2017;6(9):1002-1009Leibiger IB; Berggren P-O
- Conference publication: ACTA PHYSIOLOGICA. 2017;221:82Dolensek J; Valladolid-Acebes I; Gosak M; Klemen MS; Bombek LK; Pohorec V; Berggren PO; Brismar K; Stozer A; Rupnik MS
- Conference publication: ACTA PHYSIOLOGICA. 2017;221:14Daraio T; Bombek LK; Gosak M; Valladolid-Acebes I; Klemen MS; Refai E; Berggren PO; Brismar K; Rupnik MS; Bark C
- Article: SCANDINAVIAN JOURNAL OF IMMUNOLOGY. 2017;86(3):135-142Ganesan S; Luu TT; Chambers BJ; Meinke S; Brodin P; Vivier E; Wetzel DM; Koleske AJ; Kadri N; Hoglund P
- Article: PLOS ONE. 2017;12(8):e0183569Lee B; Song T; Lee K; Kim J; Berggren P-O; Ryu SH; Jo J
- Article: SCIENTIFIC REPORTS. 2017;7(1):7906Kumar P; Millischer V; Villaescusa JC; Nilsson IAK; Ostenson C-G; Schalling M; Osby U; Lavebratt C
- Article: SCIENTIFIC REPORTS. 2017;7(1):7744Daraio T; Bombek LK; Gosak M; Valladolid-Acebes I; Klemen MS; Refai E; Berggren P-O; Brismar K; Rupnik MS; Bark C
- Article: CELL REPORTS. 2017;20(6):1490-1501Diez JA; Arrojo e Drigo R; Zheng X; Stelmashenko OV; Chua M; Rodriguez-Diaz R; Fukuda M; Kohler M; Leibiger I; Tun SBB; Ali Y; Augustine GJ; Barathi VA; Berggren P-O
- Article: PSYCHONEUROENDOCRINOLOGY. 2017;82:46-50Lietzau G; Darsalia V; Pintana H; Ostenson C-G; Nystrom T; Fisahn A; Patrone C
- Article: CELLULAR AND MOLECULAR LIFE SCIENCES. 2017;74(15):2827-2838Alcarraz-Vizán G; Castaño C; Visa M; Montane J; Servitja J-M; Novials A
- Article: SCIENTIFIC REPORTS. 2017;7(1):6646Nord C; Eriksson M; Dicker A; Eriksson A; Grong E; Ilegems E; Marvik R; Kulseng B; Berggren P-O; Gorzsas A; Ahlgren U
- Meeting abstract: JOURNAL OF VASCULAR SURGERY. 2017;65(6):193SEriksson L; Rohl S; Saxelin R; Caidahl K; Ostenson C-G; Razuvajev A
- Article: SCANDINAVIAN JOURNAL OF IMMUNOLOGY. 2017;85(6):417-424O'Flaherty SM; Sutummaporn K; Haggtoft WL; Worrall AP; Rizzo M; Braniste V; Hoglund P; Kadri N; Chambers BJ
- Article: NATURE COMMUNICATIONS. 2017;8:15627Wagner AK; Kadri N; Snall J; Brodin P; Gilfillan S; Colonna M; Bernhardt GN; Hoglund P; Karre K; Chambers BJ
- Article: INTERNATIONAL JOURNAL OF CANCER. 2017;140(7):1528-1537Raaschou-Nielsen O; Pedersen M; Stafoggia M; Weinmayr G; Andersen ZJ; Galassi C; Sommar J; Forsberg B; Olsson D; Oftedal B; Krog NH; Aasvang GM; Pyko A; Pershagen G; Korek M; De Faire U; Pedersen NL; OEstenson C-G; Fratiglioni L; Sorensen M; Eriksen KT; Tjonneland A; Peeters PH; Bueno-de-Mesquita HBA; Plusquin M; Key TJ; Jaensch A; Nagel G; Foeger B; Wang M; Tsai M-Y; Grioni S; Marcon A; Krogh V; Ricceri F; Sacerdote C; Migliore E; Tamayo I; Amiano P; Dorronsoro M; Sokhi R; Kooter I; de Hoogh K; Beelen R; Eeftens M; Vermeulen R; Vineis P; Brunekreef B; Hoek G
- Article: PSYCHIATRY RESEARCH. 2017;250:217-220Hukic DS; Lavebratt C; Olsson E; Ostenson C-G; Eriksson SV; Erlinge D; Schalling M; Osby U
- Article: EUROPEAN HEART JOURNAL. 2017;38(13):983-990Fuks KB; Weinmayr G; Basagana X; Gruzieva O; Hampel R; Oftedal B; Sorensen M; Wolf K; Aamodt G; Aasvang GM; Aguilera I; Becker T; Beelen R; Brunekreef B; Caracciolo B; Cyrys J; Elosua R; Eriksen KT; Foraster M; Fratiglioni L; Hilding A; Houthuijs D; Korek M; Kunzli N; Marrugat J; Nieuwenhuijsen M; Ostenson C-G; Penell J; Pershagen G; Raaschou-Nielsen O; Swart WJR; Peters A; Hoffmann B
- Article: SCIENTIFIC REPORTS. 2017;7:44261Hoa TTN; Drigo RAE; Berggren P-O; Boudier T
- Article: STEM CELLS. 2017;35(3):766-776Davies LC; Heldring N; Kadri N; Le Blanc K
- Article: PLOS ONE. 2017;12(2):e0172901Lee B; Song T; Lee K; Kim J; Han S; Berggren P-O; Ryu SH; Jo J
- Article: JOURNAL OF ETHNOPHARMACOLOGY. 2017;198:214-220Mohanty S; Kamolvit W; Zambrana S; Sandstroem C; Gonzales E; Oestenson C-G; Brauner A
- Article: EBIOMEDICINE. 2017;16:262-274Qin T; Liang T; Zhu D; Kang Y; Xie L; Dolai S; Sugita S; Takahashi N; Ostenson C-G; Banks K; Gaisano HY
- Article: JOURNAL OF CARDIOVASCULAR PHARMACOLOGY. 2017;69(2):101-109Eriksson L; Rohl S; Saxelin R; Lengquist M; Kronqvist M; Caidahl K; Ostenson C-G; Razuvaev A
- Review: CURRENT OPINION IN LIPIDOLOGY. 2017;28(1):27-31Juntti-Berggren L; Berggren P-O
- Article: PLOS ONE. 2017;12(1):e0170748Ahmed AS; Li J; Abdul AMD; Ahmed M; Ostenson C-G; Salo PT; Hewitt C; Hart DA; Ackermann PW
- Article: HORMONE MOLECULAR BIOLOGY AND CLINICAL INVESTIGATION. 2017;29(1):13-26Al-Qahtani SM; Bryzgalova G; Valladolid-Acebes I; Korach-Andre M; Dahlman-Wright K; Efendic S; Berggren P-O; Portwood N
- Article: CELL REPORTS. 2016;17(12):3281-3291Almaca J; Molina J; Menegaz D; Pronin AN; Tamayo A; Slepak V; Berggren P-O; Caicedo A
- Doctoral thesis: 2016Moruzzi N
- Article: BIOSCIENCE REPORTS. 2016;36(6):e00421Larsson M; Lietzau G; Nathanson D; Ostenson C-G; Mallard C; Johansson ME; Nystrom T; Patrone C; Darsalia V
- Article: SCIENTIFIC REPORTS. 2016;6:37996Thuy TL; Ganesan S; Wagner AK; Sarhan D; Meinke S; Garbi N; Haemmerling G; Alici E; Karre K; Chambers BJ; Hoglund P; Kadri N
- Article: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 2016;101(11):3952-3958Kovamees O; Shemyakin A; Checa A; Wheelock CE; Lundberg JO; Ostenson C-G; Pernow J
- Article: VIRAL IMMUNOLOGY. 2016;29(8):452-458Niklasson B; Arbrandt G; Kawecki A; Juntti-Berggren L; Berggren P-O; Al-Qahtani SM; Gustafsson A-L; Bryzgalova G; Klitz W
- Article: DIABETES. 2016;65(10):2888-2899Strawbridge RJ; Hilding A; Silveira A; Osterholm C; Sennblad B; McLeod O; Tsikrika P; Foroogh F; Tremoli E; Baldassarre D; Veglia F; Rauramaa R; Smit AJ; Giral P; Kurl S; Mannarino E; Grossi E; Syvanen A-C; Humphries SE; de Faire U; Ostenson C-G; Maegdefessel L; Hamsten A; Backlund A
- Article: EMBO JOURNAL. 2016;35(18):2026-2044Lu J; Liu K-C; Schulz N; Karampelias C; Charbord J; Hilding A; Rautio L; Bertolino P; Ostenson C-G; Brismar K; Andersson O
- Article: PLOS ONE. 2016;11(9):e0162733Lin Y; Krogh-Andersen K; Pelletier J; Marcotte H; Ostenson C-G; Hammarstrom L
- Article: OBESITY REVIEWS. 2016;17(9):907-914Wu L; Shen C; Ahmed MS; Ostenson C-G; Gu HF
- Article: INVESTIGATIONAL NEW DRUGS. 2016;34(4):522-529Zaitseva II; Zaitsev SV; Berggren P-O
- Article: JOURNAL OF BIOENERGETICS AND BIOMEMBRANES. 2016;48(4):413-423Bergamini C; Moruzzi N; Volta F; Faccioli L; Gerdes J; Mondardini MC; Fato R
- Article: NATURE. 2016;535(7612):430-434Bader E; Migliorini A; Gegg M; Moruzzi N; Gerdes J; Roscioni SS; Bakhti M; Brandl E; Irmler M; Beckers J; Aichler M; Feuchtinger A; Leitzinger C; Zischka H; Wang-Sattler R; Jastroch M; Tschoep M; Machicao F; Staiger H; Haering H-U; Chmelova H; Chouinard JA; Oskolkov N; Korsgren O; Speier S; Lickert H
- Editorial: ADIPOCYTE. 2016;5(3):318-325Valladolid-Acebes I; Daraio T; Brismar K; Hokfelt T; Bark C
- Article: EUROPEAN JOURNAL OF NUTRITION. 2016;55(4):1615-1622Ibias J; Miguens M; del Rio D; Valladolid-Acebes I; Stucchi P; Ambrosio E; Martin M; Morales L; Ruiz-Gayo M; Del Olmo N
- Article: PSYCHIATRIC GENETICS. 2016;26(3):136-139Hukic DS; Lavebratt C; Frisen L; Backlund L; Hilding A; Gu HF; Ostenson C-G; Erlinge D; Ehrenborg E; Schalling M; Osby U
- Article: BIOMATERIALS. 2016;90:50-61Shalaly ND; Ria M; Johansson U; Avall K; Berggren P-O; Hedhammar M
- Article: TOXICOLOGY IN VITRO. 2016;33:1-8Zaitseva II; Berggren P-O; Zaitsev SV
- Article: MOLECULAR MEDICINE. 2016;21(1):969-978Muller C; Yassin K; Li L-S; Palmblad M; Efendic S; Berggren P-O; Cerami A; Brines M; Ostenson C-G
- Article: THROMBOSIS RESEARCH. 2016;141:93-97Spectre G; Stalesen R; Ostenson C-G; Hjemdahl P
- Article: DIABETOLOGIA. 2016;59(5):1007-1011Ali Y; Diez J; Selander L; Zheng X; Edlund H; Berggren P-O
- Article: JOURNAL OF THE AMERICAN SOCIETY OF NEPHROLOGY. 2016;27(4):1029-1041Villarreal R; Mitrofanova A; Maiguel D; Morales X; Jeon J; Grahammer F; Leibiger IB; Guzman J; Fachado A; Yoo TH; Katin AB; Gellermann J; Merscher S; Burke GW; Berggren P-O; Oh J; Huber TB; Fornoni A
- Article: SCIENTIFIC REPORTS. 2016;6:23103Park KS; Kim SH; Das A; Yang S-N; Jung KH; Kim MK; Berggren P-O; Lee Y; Chai JC; Kim HJ; Chai YG
- Article: CELL METABOLISM. 2016;23(3):541-546Abdulreda MH; Rodriguez-Diaz R; Caicedo A; Berggren P-O
- Article: DIABETES CARE. 2016;39(3):345-352Wagner H; Alvarsson M; Mannheimer B; Degerblad M; Ostenson C-G
- Article: TRANSPLANTATION. 2016;100(3):554-562Watanabe M; Lundgren T; Saito Y; Cerami A; Brines M; Ostenson C-G; Kumagai-Braesch M
- Article: SCIENTIFIC REPORTS. 2016;6:21448Paschen M; Moede T; Leibiger B; Jacob S; Bryzgalova G; Leibiger IB; Berggren P-O
- Article: ONCOTARGET. 2016;7(5):5865-5876Lietzau G; Nystrom T; Ostenson C-G; Darsalia V; Patrone C
- Article: FASEB JOURNAL. 2016;30(2):884-894Sun J; Xu M; Ortsater H; Lundeberg E; Juntti-Berggren L; Chen YQ; Haeggstrom JZ; Gudmundsson GH; Diana J; Agerberth B
- Article: TRANSFUSION. 2016;56(2):370-382Meinke S; Sandgren P; Mortberg A; Karlstrom C; Kadri N; Wikman A; Hoglund P
- Article: ENVIRONMENT INTERNATIONAL. 2016;87:66-73Raaschou-Nielsen O; Beelen R; Wang M; Hoek G; Andersen ZJ; Hoffmann B; Stafoggia M; Samoli E; Weinmayr G; Dimakopoulou K; Nieuwenhuijsen M; Xun WW; Fischer P; Eriksen KT; Sorensen M; Tjonneland A; Ricceri F; De Hoogh K; Key T; Eeftens M; Peeters PH; Bueno-de-Mesquita HB; Meliefste K; Oftedal B; Schwarze PE; Nafstad P; Galassi C; Migliore E; Ranzi A; Cesaroni G; Badaloni C; Forastiere F; Penell J; De Faire U; Korek M; Pedersen N; Ostenson C-G; Pershagen G; Fratiglioni L; Concin H; Nagel G; Jaensch A; Ineichen A; Naccarati A; Katsoulis M; Trichpoulou A; Keuken M; Jedynska A; Kooter IM; Kukkonen J; Brunekreef B; Sokhi RS; Katsouyanni K; Vineis P
- Doctoral thesis: 2016Shi Y
- Article: MOLECULAR AND CELLULAR ENDOCRINOLOGY. 2016;420:57-65Montane J; de Pablo S; Obach M; Cadavez L; Castaño C; Alcarraz-Vizán G; Visa M; Rodríguez-Comas J; Parrizas M; Servitja JM; Novials A
- Article: CELLR4. 2016;4(4):e2120Shishido A; Caicedo A; Rodriguez-Diaz R; Pileggi A; Berggren P-O; Abdulreda MH
- Review: NANOPARTICLES FOR RATIONAL VACCINE DESIGN. 2016;395:95-114Kadri N; Wagner AK; Ganesan S; Karre K; Wickstrom S; Johansson MH; Hoglund P
- Article: JOURNAL OF INORGANIC BIOCHEMISTRY. 2016;154:29-34Pelletier J; Domingues N; Castro MMCA; Ostenson C-G
- Article: CELL BIOLOGY AND TRANSLATIONAL MEDICINE, VOL 14: STEM CELLS IN LINEAGE SPECIFIC DIFFERENTIATION AND DISEASE. 2016;938:11-24Abdulreda MH; Rodriguez-Diaz R; Cabrera O; Caicedo A; Berggren P-O
- Article: JOURNAL OF DIABETES RESEARCH. 2016;2016:6278709-6Danielsson AK; Lundin A; Yaregal A; Östenson CG; Allebeck P; Agardh EE
- Article: DIABETOLOGIA. 2015;58(12):2810-2818Almaca J; Liang T; Gaisano HY; Nam HG; Berggren P-O; Caicedo A
- Article: JOURNAL OF DIABETES AND ITS COMPLICATIONS. 2015;29(8):1234-1239Abu Seman N; Anderstam B; Mohamud WNW; Ostenson C-G; Brismar K; Gu HF
- Article: NUTRITION & METABOLISM. 2015;12:38Voznesenskaya A; Tordoff MG
- Article: SCIENTIFIC REPORTS. 2015;5:15599Jiang H; Tong Y; Yan D; Jia S; Ostenson C-G; Chen Z
- Article: CELL REPORTS. 2015;13(1):15-22Leibiger B; Moede T; Paschen M; Yunn N-O; Lim JH; Ryu SH; Pereira T; Berggren P-O; Leibiger IB
- Review: DIABETOLOGIA. 2015;58(10):2218-2228Arrojo e Drigo R; Ali Y; Diez J; Srinivasan DK; Berggren P-O; Boehm BO
- Article: PLOS ONE. 2015;10(9):e0135781Ahmed MS; Pelletier J; Leumann H; Gu HF; Ostenson C-G
- Article: NUCLEIC ACIDS RESEARCH. 2015;43(16):7688-7701Yunn N-O; Koh A; Han S; Lim JH; Park S; Lee J; Kim E; Jang SK; Berggren P-O; Ryu SH
- Editorial: CELL CYCLE. 2015;14(17):2715-2716Juntti-Berggren L; Ali Y; Berggren P-O
- Conference publication: DIABETOLOGIA. 2015;58:S293-S294Ansurudeen I; Daraio T; Kolonelou C; Bark C; Brismar K; Valladolid-Acebes I
- Article: PLOS GENETICS. 2015;11(9):e1005500Yamashita T; Hakizimana P; Wu S; Hassan A; Jacob S; Temirov J; Fang J; Mellado-Lagarde M; Gursky R; Horner L; Leibiger B; Leijon S; Centonze VE; Berggren P-O; Frase S; Auer M; Brownell WE; Fridberger A; Zuo J
- Article: JOURNAL OF EXPOSURE SCIENCE AND ENVIRONMENTAL EPIDEMIOLOGY. 2015;25(5):517-523Korek MJ; Bellander TD; Lind T; Bottai M; Eneroth KM; Caracciolos B; de Faire UH; Fratiglioni L; Hilding A; Leander K; Magnusson PKE; Pedersen NL; Ostenson C-G; Pershagen G; Pene JC
- Article: DIABETES & VASCULAR DISEASE RESEARCH. 2015;12(5):315-324Ring M; Eriksson MJ; Fritz T; Nyberg G; Ostenson CG; Krook A; Zierath JR; Caidahl K
- Article: CELLULAR SIGNALLING. 2015;27(9):1873-1881Song P; Kwon Y; Yea K; Moon H-Y; Yoon JH; Ghim J; Hyun H; Kim D; Koh A; Berggren P-O; Suh P-G; Ryu SH
- Review: IMMUNOLOGICAL REVIEWS. 2015;267(1):167-177Kadri N; Thuy LT; Hoeglund P
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2015;309(4):E418-E427Lindfors C; Katz A; Selander L; Johansen JE; Marconi G; Schalling M; Hokfelt T; Berggren P-O; Zaitsev S; Nilsson IAK
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2015;112(31):E4326-E4335Valladolid-Acebes I; Daraio T; Brismar K; Harkany T; Oegren SO; Hoekfelt TGM; Bark C
- Article: JOURNAL OF FUNCTIONAL FOODS. 2015;17:283-292Luthje P; Lokman EF; Sandstrom C; Ostenson C-G; Brauner A
- Article: OCCUPATIONAL AND ENVIRONMENTAL MEDICINE. 2015;72(8):594-601Pyko A; Eriksson C; Oftedal B; Hilding A; Ostenson C-G; Krog NH; Julin B; Aasvang GM; Pershagen G
- Article: EXPERIMENTAL CELL RESEARCH. 2015;336(1):158-170Berglund E; Dare E; Branca RMM; Akcakaya P; Frobom R; Berggren P-O; Lui W-O; Larsson C; Zedenius J; Orre L; Lehtio J; Kim J; Branstrom R
- Article: COMPLEX PSYCHIATRY. 2015;1(2):76-81Backlund L; Wei YB; Martinsson L; Melas PA; Liu JJ; Mu N; Östenson C-G; Ekström TJ; Schalling M; Lavebratt C
- Article: FASEB JOURNAL. 2015;29(7):2970-2979Visa M; Alcarraz-Vizán G; Montane J; Cadavez L; Castaño C; Villanueva-Peñacarrillo ML; Servitja J-M; Novials A
- Article: PLOS ONE. 2015;10(6):e0130169Johansson U; Ria M; Avall K; Shalaly ND; Zaitsev SV; Berggren P-O; Hedhammar M
- Article: SCIENTIFIC REPORTS. 2015;5:10740Ilegems E; van Krieken PP; Edlund PK; Dicker A; Alanentalo T; Eriksson M; Mandic S; Ahlgren U; Berggren P-O
- Review: ENVIRONMENTAL HEALTH PERSPECTIVES. 2015;123(6):525-533Beelen R; Hoek G; Raaschou-Nielsen O; Stafoggia M; Andersen ZJ; Weinmayr G; Hoffmann B; Wolf K; Samoli E; Fischer PH; Nieuwenhuijsen MJ; Xun WW; Katsouyanni K; Dimakopoulou K; Marcon A; Vartiainen E; Lanki T; Yli-Tuomi T; Oftedal B; Schwarze PE; Nafstad P; De Faire U; Pedersen NL; Otstenson C-G; Fratiglioni L; Penell J; Korek M; Pershagen G; Eriksen KT; Overvad K; Sorensen M; Eeftens M; Peeters PH; Meliefste K; Wang M; Bueno-de-Mesquita HB; Sugiri D; Kramer U; Heinrich J; de Hoogh K; Key T; Peters A; Hampel R; Concin H; Nagel G; Jaensch A; Ineichen A; Tsai M-Y; Schaffner E; Probst-Hensch NM; Schindler C; Ragettli MS; Vilier A; Clavel-Chapelon F; Declercq C; Ricceri F; Sacerdote C; Galassi C; Migliore E; Ranzi A; Cesaroni G; Badaloni C; Forastiere F; Katsoulis M; Trichopoulou A; Keuken M; Jedynska A; Kooter IM; Kukkonen J; Sokhi RS; Vineis P; Brunekreef B
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2015;112(20):E2611-E2619Avall K; Ali Y; Leibiger IB; Leibiger B; Moede T; Paschen M; Dicker A; Dare E; Kohler M; Ilegems E; Abdulreda MH; Graham M; Crooke RM; Tay VSY; Refai E; Nilsson SK; Jacob S; Selander L; Berggren P-O; Juntti-Berggren L
- Article: BIOSCIENCE REPORTS. 2015;35(3):e00195Yi J; Yang X; Zheng L; Yang G; Sun L; Bao Y; Wu Y; Huang Y; Yu C; Yang S-N; Li Y
- Article: PLOS ONE. 2015;10(4):e0121984Mannheimer B; Wagner H; Otstenson C-G; Diczfalusy U
- Article: NATURE MEDICINE. 2015;21(4):363-372Marquard J; Otter S; Welters A; Stirban A; Fischer A; Eglinger J; Herebian D; Kletke O; Klemen MS; Stozer A; Wnendt S; Piemonti L; Kohler M; Ferrer J; Thorens B; Schliess F; Rupnik MS; Heise T; Berggren P-O; Kloecker N; Meissner T; Mayatepek E; Eberhard D; Kragl M; Lammert E
- Article: MOLECULAR & CELLULAR PROTEOMICS. 2015;14(4):882-892Yoon JH; Kim D; Jang J-H; Ghim J; Park S; Song P; Kwon Y; Kim J; Hwang D; Bae Y-S; Suh P-G; Berggren P-O; Ryu SH
- Article: CLINICAL EPIGENETICS. 2015;7(1):30Abu Seman N; Mohamud WNW; Ostenson C-G; Brismar K; Gu HF
- Article: MOLECULAR MEDICINE. 2015;20(1):658-666Brines M; Dunne AN; van Velzen M; Proto PL; Ostenson C-G; Kirk RI; Petropoulos IN; Javed S; Malik RA; Cerami A; Dahan A
- Article: CELLULAR AND MOLECULAR LIFE SCIENCES. 2015;72(6):1197-1207Yang G; Shi Y; Yu J; Li Y; Yu L; Welling A; Hofmann F; Striessnig J; Juntti-Berggren L; Berggren P-O; Yang S-N
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2015;290(5):2812-2821Kara I; Poggi M; Bonardo B; Govers R; Landrier J-F; Tian S; Leibiger I; Day R; Creemers JWM; Peiretti F
- Corrigendum: PLOS ONE. 2015;10(10):e0141292Seed Ahmed M; Pelletier J; Leumann H; Gu HF; Östenson C-G
- Article: EVIDENCE-BASED COMPLEMENTARY AND ALTERNATIVE MEDICINE. 2015;2015:120572-7Lokman EF; Gu HF; Mohamud WNW; Ostenson C-G
- Editorial: NATURE MEDICINE. 2015;21(1):14-16Leibiger IB; Berggren P-O
- Review: ADVANCES IN BIOLOGICAL REGULATION. 2015;57:112-119Barker CJ; Li L; Köhler M; Berggren P-O
- Article: DIABETES RESEARCH AND CLINICAL PRACTICE. 2015;107(1):166-177Hilding A; Shen C; Ostenson C-G
- Article: FASEB JOURNAL. 2015;29(1):95-104Alcarraz-Vizán G; Casini P; Cadavez L; Visa M; Montane J; Servitja J-M; Novials A
- Article: BMJ OPEN DIABETES RESEARCH & CARE. 2015;3(1):e000120Olsson E; Westman J; Hukic DS; Eriksson SV; Edman G; Boden R; Jedenius E; Reutfors J; Berntsson A; Hilding A; Schalling M; Ostenson C-G; Osby U
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2014;111(49):17612-17617Almaca J; Molina J; Arrojo e Drigo R; Abdulreda MH; Jeon WB; Berggren P-O; Caicedo A; Nam HG
- Article: DIABETES. 2014;63(12):4218-4229Kibbey RG; Choi CS; Lee H-Y; Cabrera O; Pongratz RL; Zhao X; Birkenfeld AL; Li C; Berggren P-O; Stanley C; Shulman GI
- Article: DIABETES. 2014;63(12):4100-4114Li L; Trifunovic A; Kohler M; Wang Y; Berglund JP; Illies C; Juntti-Berggren L; Larsson N-G; Berggren P-O
- Article: NATURE COMMUNICATIONS. 2014;5:5308Gerdes JM; Christou-Savina S; Xiong Y; Moede T; Moruzzi N; Karlsson-Edlund P; Leibiger B; Leibiger IB; Ostenson C-G; Beales PL; Berggren P-O
- Review: CELLULAR AND MOLECULAR LIFE SCIENCES. 2014;71(21):4149-4177Yang S-N; Shi Y; Yang G; Li Y; Yu J; Berggren P-O
- Article: ANNALS OF MEDICINE. 2014;46(7):539-546Almgren M; Atkinson RL; Hilding A; He J; Brismar K; Schalling M; Ostenson C-G; Lavebratt C
- Article: CELL AND TISSUE RESEARCH. 2014;358(2):331-342Williams P; Tulke S; Ilegems E; Berggren P-O; Broberger C
- Editorial: SCIENCE TRANSLATIONAL MEDICINE. 2014;6(257):257fs39Ostenson C-G
- Article: ENVIRONMENTAL HEALTH PERSPECTIVES. 2014;122(9):919-925Stafoggia M; Cesaroni G; Peters A; Andersen ZJ; Badaloni C; Beelen R; Caracciolo B; Cyrys J; de Faire U; de Hoogh K; Eriksen KT; Fratiglioni L; Galassi C; Gigante B; Havulinna AS; Hennig F; Hilding A; Hoek G; Hoffmann B; Houthuijs D; Korek M; Lanki T; Leander K; Magnusson PK; Meisinger C; Migliore E; Overvad K; Ostenson C-G; Pedersen NL; Pekkanen J; Penell J; Pershagen G; Pundt N; Pyko A; Raaschou-Nielsen O; Ranzi A; Ricceri F; Sacerdote C; Swart WJR; Turunen AW; Vineis P; Weimar C; Weinmayr G; Wolf K; Brunekreef B; Forastiere F
- Article: JOURNAL OF NUTRITIONAL BIOCHEMISTRY. 2014;25(9):954-963Jiang H; Fen J; Du Z; Zhen H; Lin M; Jia S; Li T; Huan X; Ostenson C-G; Chen Z
- Review: ENVIRONMENTAL HEALTH PERSPECTIVES. 2014;122(9):896-905Fuks KB; Weinmayr G; Foraster M; Dratva J; Hampel R; Houthuijs D; Oftedal B; Oudin A; Panasevich S; Penell J; Sommar JN; Sorensen M; Tiittanen P; Wolf K; Xun WW; Aguilera I; Basagana X; Beelen R; Bots ML; Brunekreef B; Bueno-de-Mesquita HB; Caracciolo B; Cirach M; de Faire U; de Nazelle A; Eeftens M; Elosua R; Erbel R; Forsberg B; Fratiglioni L; Gaspoz J-M; Hilding A; Jula A; Korek M; Kraemer U; Kuenzli N; Lanki T; Leander K; Magnusson PKE; Marrugat J; Nieuwenhuijsen MJ; Oestenson C-G; Pedersen NL; Pershagen G; Phuleria HC; Probst-Hensch NM; Raaschou-Nielsen O; Schaffner E; Schikowski T; Schindler C; Schwarze PE; Sogaard AJ; Sugiri D; Swart WJR; Tsai M-Y; Turunen AW; Vineis P; Peters A; Hoffmann B
- Article: NEUROREPORT. 2014;25(11):819-822Cano V; Valladolid-Acebes I; Hernández-Nuño F; Merino B; Del Olmo N; Chowen JA; Ruiz-Gayo M
- Conference publication: ACTA PHYSIOLOGICA. 2014;211:69Valladolid-Acebes I; Daraio T; Brismar K; Ogren SO; Hokfelt T; Bark IC
- Article: PHYSIOLOGY & BEHAVIOR. 2014;135:49-54Tordoff MG; Downing A; Voznesenskaya A
- Article: INTERNATIONAL JOURNAL OF BIOCHEMISTRY & CELL BIOLOGY. 2014;53:66-76Moruzzi N; Del Sole M; Fato R; Gerdes JM; Berggren P-O; Bergamini C; Brismar K
- Article: DIABETES. 2014;63(8):2714-2726Molina J; Rodriguez-Diaz R; Fachado A; Jacques-Silva MC; Berggren P-O; Caicedo A
- Article: CELL. 2014;158(1):41-53Lo JC; Ljubicic S; Leibiger B; Kern M; Leibiger IB; Moede T; Kelly ME; Bhowmick DC; Murano I; Cohen P; Banks AS; Khandekar MJ; Dietrich A; Flier JS; Cinti S; Blueher M; Danial NN; Berggren P-O; Spiegelman BM
- Article: ENVIRONMENTAL HEALTH PERSPECTIVES. 2014;122(7):687-694Eriksson C; Hilding A; Pyko A; Bluhm G; Pershagen G; Ostenson C-G
- Article: CELL AND TISSUE RESEARCH. 2014;357(1):109-117Ahmed AS; Li J; Schizas N; Ahmed M; Ostenson C-G; Salo P; Hewitt C; Hart DA; Ackermann PW
- Article: DIABETOLOGIA. 2014;57(7):1456-1465Kim D; Kim J; Yoon JH; Ghim J; Yea K; Song P; Park S; Lee A; Hong C-P; Jang MS; Kwon Y; Park S; Jang MH; Berggren P-O; Suh P-G; Ryu SH
- Article: PLOS ONE. 2014;9(6):e98904Tseng C-N; Karlof E; Chang Y-T; Lengquist M; Rotzius P; Berggren P-O; Hedin U; Eriksson EE
- Article: DIABETOLOGIA. 2014;57(6):1159-1172Strawbridge RJ; Deleskog A; McLeod O; Folkersen L; Kavousi M; Gertow K; Baldassarre D; Veglia F; Leander K; Gigante B; Kauhanen J; Rauramaa R; Smit AJ; Mannarino E; Giral P; Dehghan A; Hofman A; Franco OH; Humphries SE; Tremoli E; de Faire U; Gustafsson S; Ostensson C-G; Eriksson P; Ohrvik J; Hamsten A
- Article: EPIDEMIOLOGY. 2014;25(3):368-378Beelen R; Stafoggia M; Raaschou-Nielsen O; Andersen ZJ; Xun WW; Katsouyanni K; Dimakopoulou K; Brunekreef B; Weinmayr G; Hoffmann B; Wolf K; Samoli E; Houthuijs D; Nieuwenhuijsen M; Oudin A; Forsberg B; Olsson D; Salomaa V; Lanki T; Yli-Tuomi T; Oftedal B; Aamodt G; Nafstad P; De Faire U; Pedersen NL; Ostenson C-G; Fratiglioni L; Penell J; Korek M; Pyko A; Eriksen KT; Tjonneland A; Becker T; Eeftens M; Bots M; Meliefste K; Wang M; Bueno-de-Mesquita B; Sugiri D; Kraemer U; Heinrich J; de Hoogh K; Key T; Peters A; Cyrys J; Concin H; Nagel G; Ineichen A; Schaffner E; Probst-Hensch N; Dratva J; Ducret-Stich R; Vilier A; Clavel-Chapelon F; Stempfelet M; Grioni S; Krogh V; Tsai M-Y; Marcon A; Ricceri F; Sacerdote C; Galassi C; Migliore E; Ranzi A; Cesaroni G; Badaloni C; Forastiere F; Tamayo I; Amiano P; Dorronsoro M; Katsoulis M; Trichopoulou A; Vineis P; Hoek G
- Article: ENVIRONMENT INTERNATIONAL. 2014;66:97-106Wang M; Beelen R; Stafoggia M; Raaschou-Nielsen O; Andersen ZJ; Hoffmann B; Fischer P; Houthuijs D; Nieuwenhuijsen M; Weinmayr G; Vineis P; Xun WW; Dimakopoulou K; Samoli E; Laatikainen T; Lanki T; Turunen AW; Oftedal B; Schwarze P; Aamodt G; Penell J; De Faire U; Korek M; Leander K; Pershagen G; Pedersen NL; Ostenson C-G; Fratiglioni L; Eriksen KT; Sorensen M; Tjonneland A; Bueno-de-Mesquita B; Eeftens M; Bots ML; Meliefste K; Kraemer U; Heinrich J; Sugiri D; Key T; de Hoogh K; Wolf K; Peters A; Cyrys J; Jaensch A; Concin H; Nagel G; Tsai M-Y; Phuleria H; Ineichen A; Kuenzli N; Probst-Hensch N; Schaffner E; Vilier A; Clavel-Chapelon F; Declerq C; Ricceri F; Sacerdote C; Marcon A; Galassi C; Migliore E; Ranzi A; Cesaroni G; Badaloni C; Forastiere F; Katsoulis M; Trichopoulou A; Keuken M; Jedynska A; Kooter IM; Kukkonen J; Sokhi RS; Brunekreef B; Katsouyanni K; Hoek G
- Article: BIOPOLYMERS. 2014;102(3):252-259Lindgren J; Refai E; Zaitsev SV; Abrahmsen L; Berggren P-O; Karlstrom AE
- Article: NORDIC JOURNAL OF PSYCHIATRY. 2014;68(4):251-258Osby U; Olsson E; Edman G; Hilding A; Eriksson SV; Ostenson CG
- Meeting abstract: JACC-JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY. 2014;63(12):a2144Lo JC; Ljubicic S; Leibiger B; Chatterjee-Bhowmick D; Kelly ME; Cohen P; Leibiger I; Banks A; Flier J; Bluher M; Danial N; Berggren P-O; Spiegelman BM
- Article: DIABETES RESEARCH AND CLINICAL PRACTICE. 2014;104(1):112-120Mayega RW; Guwatudde D; Makumbi FE; Nakwagala FN; Peterson S; Tomson G; Ostenson C-G
- Article: CELLULAR AND MOLECULAR LIFE SCIENCES. 2014;71(7):1289-1303Shi Y; Yang G; Yu J; Yu L; Westenbroek R; Catterall WA; Juntti-Berggren L; Berggren P-O; Yang S-N
- Article: JOURNAL OF EXPERIMENTAL MEDICINE. 2014;211(3):441-456Miska J; Abdulreda MH; Devarajan P; Lui JB; Suzuki J; Pileggi A; Berggren P-O; Chen Z
- Article: LANCET. 2014;383(9919):785-795Beelen R; Raaschou-Nielsen O; Stafoggia M; Andersen ZJ; Weinmayr G; Hoffmann B; Wolf K; Samoli E; Fischer P; Nieuwenhuijsen M; Vineis P; Xun WW; Katsouyanni K; Dimakopoulou K; Oudin A; Forsberg B; Modig L; Havulinna AS; Lanki T; Turunen A; Oftedal B; Nystad W; Nafstad P; De Faire U; Pedersen NL; Ostenson C-G; Fratiglioni L; Penell J; Korek M; Pershagen G; Eriksen KT; Overvad K; Ellermann T; Eeftens M; Peeters PH; Meliefste K; Wang M; Bueno-de-Mesquita B; Sugiri D; Kraemer U; Heinrich J; de Hoogh K; Key T; Peters A; Hampel R; Concin H; Nagel G; Ineichen A; Schaffner E; Probst-Hensch N; Kuenzli N; Schindler C; Schikowski T; Adam M; Phuleria H; Vilier A; Clavel-Chapelon F; Declercq C; Grioni S; Krogh V; Tsai M-Y; Ricceri F; Sacerdote C; Galassi C; Migliore E; Ranzi A; Cesaroni G; Badaloni C; Forastiere F; Tamayo I; Amiano P; Dorronsoro M; Katsoulis M; Trichopoulou A; Brunekreef B; Hoek G
- Article: ACTA PHYSIOLOGICA. 2014;210(3):590-599Yang J; Zheng X; Haugen F; Dare E; Lovdahl C; Schulte G; Fredholm BB; Valen G
- Article: JOURNAL OF INORGANIC BIOCHEMISTRY. 2014;131:115-122Domingues N; Pelletier J; Ostenson C-G; Castro MMCA
- Article: SCIENTIFIC REPORTS. 2014;4:3872Kistler AD; Caicedo A; Abdulreda MH; Faul C; Kerjaschki D; Berggren P-O; Reiser J; Fornoni A
- Article: BMJ-BRITISH MEDICAL JOURNAL. 2014;348:f7412Cesaroni G; Forastiere F; Stafoggia M; Andersen ZJ; Badaloni C; Beelen R; Caracciolo B; de Faire U; Erbel R; Eriksen KT; Fratiglioni L; Galassi C; Hampel R; Heier M; Hennig F; Hilding A; Hoffmann B; Houthuijs D; Joeckel K-H; Korek M; Lanki T; Leander K; Magnusson PKE; Migliore E; Ostenson C-G; Overvad K; Pedersen NL; Pekkanen JJ; Penell J; Pershagen G; Pyko A; Raaschou-Nielsen O; Ranzi A; Ricceri F; Sacerdote C; Salomaa V; Swart W; Turunen AW; Vineis P; Weinmayr G; Wolf K; de Hoogh K; Hoek G; Brunekreef B; Peters A
- Article: PLOS ONE. 2014;9(1):e85581Antonson P; Matic M; Portwood N; Kuiper RV; Bryzgalova G; Gao H; Windahl SH; Humire P; Ohlsson C; Berggren P-O; Gustafsson J-A; Dahlman-Wright K
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2014;443(1):223-228Ma Z; Moruzzi N; Catrina S-B; Grill V; Bjorklund A
- Article: PLOS ONE. 2014;9(7):e101797Cadavez L; Montane J; Alcarraz-Vizán G; Visa M; Vidal-Fàbrega L; Servitja J-M; Novials A
- Article: DIABETOLOGIA. 2014;57(1):148-156Wu H; Mezghenna K; Marmol P; Guo T; Moliner A; Yang S-N; Berggren P-O; Ibanez CF
- Article: AMERICAN JOURNAL OF NEPHROLOGY. 2014;40(5):408-416Abu Seman N; He B; Ojala JRM; Mohamud WNW; Ostenson C-G; Brismar K; Gu HF
- Article: DIABETIC MEDICINE. 2014;31(1):92-101Ostenson CG; Geelhoed-Duijvestijn P; Lahtela J; Weitgasser R; Jensen MM; Pedersen-Bjergaard U
- Article: HEALTH AND QUALITY OF LIFE OUTCOMES. 2013;11:217Reaney M; Mathieu C; Ostenson C-G; Matthaei S; Krarup T; Kiljanski J; Salaun-Martin C; Sapin H; Theodorakis M; Guerci B
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2013;110(51):20581-20586Ilegems E; Dicker A; Speier S; Sharma A; Bahow A; Edlund PK; Leibiger IB; Berggren P-O
- Article: DIABETES THERAPY. 2013;4(2):285-308Mathieu C; Ostenson C-G; Matthaei S; Reaney M; Krarup T; Guerci B; Kiljanski J; Salaun-Martin C; Sapin H; Theodorakis M
- Article: JOURNAL OF MEDICAL ECONOMICS. 2013;16(12):1453-1461Geelhoed-Duijvestijn PH; Pedersen-Bjergaard U; Weitgasser R; Lahtela J; Jensen MM; Östenson C-G
- Article: DIABETOLOGIA. 2013;56(12):2669-2678Schmidt-Christensen A; Hansen L; Ilegems E; Fransen-Pettersson N; Dahl U; Gupta S; Larefalk A; Hannibal TD; Schulz A; Berggren P-O; Holmberg D
- Article: CLINICAL EPIGENETICS. 2013;5(1):21Gu T; Gu HF; Hilding A; Sjoholm LK; Ostenson C-G; Ekstrom TJ; Brismar K
- Article: CLINICAL EPIGENETICS. 2013;5(1):20Gu HF; Gu T; Hilding A; Zhu Y; Karvestedt L; Ostenson C-G; Lai M; Kutsukake M; Frystyk J; Tamura K; Brismar K
- Article: ARTERIOSCLEROSIS THROMBOSIS AND VASCULAR BIOLOGY. 2013;33(11):2633-2638Deleskog A; Piksasova O; Silveira A; Gertow K; Baldassarre D; Veglia F; Sennblad B; Strawbridge RJ; Larsson M; Leander K; Gigante B; Kauhanen J; Rauramaa R; Smit AJ; Mannarino E; Giral P; Gustafsson S; Ostenson C-G; Humphries SE; Tremoli E; de Faire U; Ohrvik J; Hamsten A
- Article: NEUROBIOLOGY OF LEARNING AND MEMORY. 2013;106:18-25Valladolid-Acebes I; Fole A; Martín M; Morales L; Cano MV; Ruiz-Gayo M; Del Olmo N
- Article: PHYSIOLOGY & BEHAVIOR. 2013;122:159-162Voznesenskaya A; Tordoff MG
- Article: JOURNAL OF LEUKOCYTE BIOLOGY. 2013;95(2):265-274Jaczewska J; Abdulreda MH; Yau CY; Schmitt MM; Schubert I; Berggren P-O; Weber C; Koenen RR; Moy VT; Wojcikiewicz EP
- Article: DIABETES CARE. 2013;36(9):2683-2689Eriksson A-K; van den Donk M; Hilding A; Ostenson C-G
- Review: DIABETES OBESITY & METABOLISM. 2013;15(0 3):105-116Abdulreda MH; Berggren P-O
- Article: PLOS ONE. 2013;8(8):e71359Hu Z; Xiong Y; Han X; Geng C; Jiang B; Huo Y; Luo J
- Article: PLOS ONE. 2013;8(8):e72554Mayega RW; Guwatudde D; Makumbi F; Nakwagala FN; Peterson S; Tomson G; Ostenson C-G
- Article: OBESITY. 2013;21(8):1720-1725Langberg E-C; Ahmed MS; Efendic S; Gu HF; Ostenson C-G
- Article: LANCET ONCOLOGY. 2013;14(9):813-822Raaschou-Nielsen O; Andersen ZJ; Beelen R; Samoli E; Stafoggia M; Weinmayr G; Hoffmann B; Fischer P; Nieuwenhuijsen MJ; Brunekreef B; Xun WW; Katsouyanni K; Dimakopoulou K; Sommar J; Forsberg B; Modig L; Oudin A; Oftedal B; Schwarze PE; Nafstad P; De Faire U; Pedersen NL; Ostenson C-G; Fratiglioni L; Penell J; Korek M; Pershagen G; Eriksen KT; Sorensen M; Tjonneland A; Ellermann T; Eeftens M; Peeters PH; Meliefste K; Wang M; Bueno-de-Mesquita B; Key TJ; de Hoogh K; Concin H; Nagel G; Vilier A; Grioni S; Krogh V; Tsai M-Y; Ricceri F; Sacerdote C; Galassi C; Migliore E; Ranzi A; Cesaroni G; Badaloni C; Forastiere F; Tamayo I; Amiano P; Dorronsoro M; Trichopoulou A; Bamia C; Vineis P; Hoek G
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2013;110(31):12673-12678Park S-H; Ryu S-Y; Yu W-J; Han YE; Ji Y-S; Oh K; Sohn J-W; Lim A; Jeon J-P; Lee H; Lee K-H; Lee S-H; Berggren P-O; Jeon J-H; Ho W-K
- Article: PLOS ONE. 2013;8(7):e67498Ma Z; Moruzzi N; Catrina S-B; Hals I; Oberholzer J; Grill V; Bjorklund A
- Corrigendum: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 2013;98(7):3091MacDonald MJ; Langberg E-C; Tibell A; Sabat G; Kendrick MA; Szweda LI; Ostenson CG
- Article: AMERICAN JOURNAL OF TRANSPLANTATION. 2013;13(6):1461-1473Tan Y; Abdulreda MH; Cruz-Guilloty F; Cutrufello N; Shishido A; Martinez RE; Duffort S; Xia X; Echegaray-Mendez J; Levy RB; Berggren P-O; Perez VL
- Article: CELL RESEARCH. 2013;23(6):820-834Xiong Y; Hu Z; Han X; Jiang B; Zhang R; Zhang X; Lu Y; Geng C; Li W; He Y; Huo Y; Shibuya M; Luo J
- Article: DIABETES. 2013;62(6):2004-2014Steneberg P; Bernardo L; Edfalk S; Lundberg L; Backlund F; Ostenson C-G; Edlund H
- Meeting abstract: VALUE IN HEALTH. 2013;16(3):A167Weitgasser R; Lahtela J; Geelhoed-Duijvestijn P; Markert Jensen M; Ostenson CG
- Article: EXPERIMENTAL CELL RESEARCH. 2013;319(8):1229-1238Berglund E; Berglund D; Akcakaya P; Ghaderi M; Dare E; Berggren P-O; Kohler M; Aspinwall CA; Lui W-O; Zedenius J; Larsson C; Branstrom R
- Article: INTEGRATIVE BIOLOGY. 2013;5(4):712-719Christakou AE; Ohlin M; Vanherberghen B; Khorshidi MA; Kadri N; Frisk T; Wiklund M; Onfelt B
- Article: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 2013;98(4):E727-E731MacDonald MJ; Langberg E-C; Tibell A; Sabat G; Kendrick MA; Szweda LI; Ostenson CG
- Review: PHARMACOLOGICAL REVIEWS. 2013;65(2):641-669Barker CJ; Berggren P-O
- Article: MOLECULAR AND CELLULAR BIOLOGY. 2013;33(8):1608-1620Koh A; Lee MN; Yang YR; Jeong H; Ghim J; Noh J; Kim J; Ryu D; Park S; Song P; Koo S-H; Leslie NR; Berggren P-O; Choi JH; Suh P-G; Ryu SH
- Article: PHYSIOLOGY & BEHAVIOR. 2013;112-113:56-60Voznesenskaya A; Tordoff MG
- Article: JOVE-JOURNAL OF VISUALIZED EXPERIMENTS. 2013;(73):e50466Abdulreda MH; Caicedo A; Berggren P-O
- Article: PLOS ONE. 2013;8(2):e57458Estrogen Signalling and the Metabolic Syndrome: Targeting the Hepatic Estrogen Receptor Alpha ActionMatic M; Bryzgalova G; Gao H; Antonson P; Humire P; Omoto Y; Portwood N; Pramfalk C; Efendic S; Berggren P-O; Gustafsson J-A; Dahlman-Wright K
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2013;288(8):5732-5742Song P; Kim JH; Ghim J; Yoon JH; Lee A; Kwon Y; Hyun H; Moon H-Y; Choi H-S; Berggren P-O; Suh P-G; Ryu SH
- Article: DIABETES. 2013;62(2):531-542Korpos E; Kadri N; Kappelhoff R; Wegner J; Overall CM; Weber E; Holmberg D; Cardell S; Sorokin L
- Corrigendum: JOURNAL OF BIOLOGICAL CHEMISTRY. 2013;288(2):1277Jeon J; Leibiger I; Moede T; Walter B; Faul C; Maiguel D; Villarreal R; Guzman J; Berggren P-O; Mundel P; Ricordi C; Merscher-Gomez S; Fornoni A
- Article: EVIDENCE-BASED COMPLEMENTARY AND ALTERNATIVE MEDICINE. 2013;2013:727602-7Lokman FE; Gu HF; Mohamud WNW; Yusoff MM; Chia KL; Oestenson C-G
- Article: ADVANCES IN PHARMACOLOGICAL SCIENCES. 2013;2013:808914-7Mansor F; Gu HF; Ostenson C-G; Manneras-Holm L; Stener-Victorin E; Mohamud WNW
- Article: CLINICOECONOMICS AND OUTCOMES RESEARCH. 2013;5:355-367Kiiskinen U; Matthaei S; Reaney M; Mathieu C; Oestenson C-G; Krarup T; Theodorakis M; Kiljanski J; Salaun-Martin C; Sapin H; Guerci B
- Editorial: MOLECULAR METABOLISM. 2013;2(1):1-2Leibiger IB; Ilegems E; Berggren P-O
- Article: CELLR4. 2013;1(2):111-122Abdulreda MH; Caicedo A; Berggren P-O
- Article: DIABETES METABOLIC SYNDROME AND OBESITY. 2013;6:171-185Ostenson C-G; Matthaei S; Reaney M; Krarup T; Guerci B; Kiljanski J; Salaun-Martin C; Sapin H; Bruhn D; Mathieu C; Theodorakis M
- Article: JOURNAL OF NUTRITION AND METABOLISM. 2013;2013:765383-7Huyen VTT; Phan DV; Thang P; Hoa NK; Ostenson CG
- Article: NEUROPSYCHIATRIC DISEASE AND TREATMENT. 2013;9:371-377Boden R; Edman G; Reutfors J; Ostenson C-G; Osby U
- Review: ADVANCES IN BIOLOGICAL REGULATION. 2013;53(1):156-163Barker CJ; Leibiger IB; Berggren P-O
- Article: ACTA NEUROPATHOLOGICA. 2013;125(1):145-157Sole-Domenech S; Sjovall P; Vukojevic V; Fernando R; Codita A; Salve S; Bogdanovic N; Mohammed AH; Hammarstrom P; Nilsson KPR; LaFerla FM; Jacob S; Berggren P-O; Gimenez-Llort L; Schalling M; Terenius L; Johansson B
- Article: DIABETES-METABOLISM RESEARCH AND REVIEWS. 2013;29(1):25-32Fritz T; Caidahl K; Krook A; Lundstrom P; Mashili F; Osler M; Szekeres FLM; Ostenson CG; Wandell P; Zierath JR
- Article: AMERICAN JOURNAL OF CLINICAL NUTRITION. 2013;97(1):179-187Wirstrom T; Hilding A; Gu HF; Ostenson C-G; Bjorklund A
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2012;109(52):21456-21461Rodriguez-Diaz R; Speier S; Molano RD; Formoso A; Gans I; Abdulreda MH; Cabrera O; Molina J; Fachado A; Ricordi C; Leibiger I; Pileggi A; Berggren P-O; Caicedo A
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2012;109(51):20925-20930Leibiger B; Moede T; Muhandiramlage TP; Kaiser D; Sanchez PV; Leibiger IB; Berggren P-O
- Article: SCANDINAVIAN JOURNAL OF PUBLIC HEALTH. 2012;40(8):730-737Ostenson C-G; Hilding A; Grill V; Efendic S
- Article: JOURNAL OF APPLIED PHYSIOLOGY. 2012;113(11):1784-1791Ahmed AS; Schizas N; Li J; Ahmed M; Ostenson C-G; Salo P; Hewitt C; Hart DA; Ackermann PW
- Article: BMC PHYSIOLOGY. 2012;12:14Nyrén R; Chang CL; Lindström P; Barmina A; Vorrsjö E; Ali Y; Juntti-Berggren L; Bensadoun A; Young SG; Olivecrona T; Olivecrona G
- Article: DIABETES THERAPY. 2012;3(1):6Matthaei S; Reaney M; Mathieu C; Ostenson C-G; Krarup T; Guerci B; Kiljanski J; Petto H; Bruhn D; Theodorakis M
- Meeting abstract: VALUE IN HEALTH. 2012;15(7):A502Geelhoed-duijvestijn PHLM; Pedersen-bjergaard U; Jensen MM; Ostenson CG
- Article: PLOS ONE. 2012;7(10):e47632Mayega RW; Makumbi F; Rutebemberwa E; Peterson S; Ostenson C-G; Tomson G; Guwatudde D
- Article: CELLULAR AND MOLECULAR LIFE SCIENCES. 2012;69(17):2951-2966Howl J; Matou-Nasri S; West DC; Farquhar M; Slaninova J; Ostenson C-G; Zorko M; Ostlund P; Kumar S; Langel U; McKeating J; Jones S
- Article: ISLETS. 2012;4(5):343-348Ahmed MS; Kovoor A; Nordman S; Abu Seman N; Gu T; Efendic S; Brismar K; Ostenson C-G; Gu HF
- Review: ADVANCES IN BIOLOGICAL REGULATION. 2012;52(3):361-368Barker CJ; Berggren P-O
- Review: SCANDINAVIAN JOURNAL OF IMMUNOLOGY. 2012;76(3):246-255Rhost S; Sedimbi S; Kadri N; Cardell SL
- Article: DIABETES. 2012;61(9):2380-2384Spectre G; Ostenson C-G; Li N; Hjemdahl P
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2012;287(34):28932-28942Jeon J; Leibiger I; Moede T; Walter B; Faul C; Maiguel D; Villarreal R; Guzman J; Berggren P-O; Mundel P; Ricordi C; Merscher-Gomez S; Fornoni A
- Article: PLOS ONE. 2012;7(7):e41652Almgren M; Atkinson R; He J; Hilding A; Hagman E; Wolk A; Thorell A; Marcus C; Naslund E; Ostenson C-G; Schalling M; Lavebratt C
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2012;109(30):12183-12188Yang S-N; Shi Y; Yang G; Li Y; Yu L; Shin O-H; Bacaj T; Suedhof TC; Yu J; Berggren P-O
- Article: ATHEROSCLEROSIS. 2012;223(1):223-229Deleskog A; Piksasova O; Silveira A; Samnegard A; Tornvall P; Eriksson P; Gustafsson S; Ostenson C-G; Ohrvik J; Hamsten A
- Review: CARDIOVASCULAR RESEARCH. 2012;95(1):7-18Bir SC; Xiong Y; Kevil CG; Luo J
- Article: LAKARTIDNINGEN. 2012;109(25):1208-1209Agardh C-D; Ahrén B; Hanås R; Jansson S; Smith U; Toft E; Ostenson C-G
- Article: DIABETOLOGIA. 2012;55(6):1668-1678Deleskog A; Hilding A; Brismar K; Hamsten A; Efendic S; Ostenson C-G
- Article: NATURE PROTOCOLS. 2012;7(6):1015-1023Rodriguez-Diaz R; Dando R; Huang YA; Berggren P-O; Roper SD; Caicedo A
- Article: CELLULAR AND MOLECULAR LIFE SCIENCES. 2012;69(10):1733-1743Dekki N; Refai E; Holmberg R; Kohler M; Jornvall H; Berggren P-O; Juntti-Berggren L
- Article: JOURNAL OF IMMUNOLOGY. 2012;188(7):3138-3149Kadri N; Korpos E; Gupta S; Briet C; Löfbom L; Yagita H; Lehuen A; Boitard C; Holmberg D; Sorokin L; Cardell SL
- Article: DIABETIC MEDICINE. 2012;29(4):441-452Alcohol consumption and risk of pre-diabetes and type 2 diabetes development in a Swedish populationCullmann M; Hilding A; Ostenson C-G
- Article: JOURNAL OF NATURAL MEDICINES. 2012;66(2):257-264Fazliana M; Gu HF; Ostenson C-G; Yusoff MM; Nazaimoon WMW
- Article: PHYSIOLOGY & BEHAVIOR. 2012;105(5):1214-1218Golden GJ; Voznesenskaya A; Tordoff MG
- Article: DIABETES. 2012;61(3):665-673Henriksnas J; Lau J; Zang G; Berggren P-O; Kohler M; Carlsson P-O
- Article: EUROPEAN JOURNAL OF EPIDEMIOLOGY. 2012;27(3):153-162Eriksson A-K; Ekbom A; Hilding A; Ostenson C-G
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2012;302(4):E396-E402Valladolid-Acebes I; Merino B; Principato A; Fole A; Barbas C; Lorenzo MP; García A; Del Olmo N; Ruiz-Gayo M; Cano V
- Review: ACTA PHYSIOLOGICA. 2012;204(2):178-185Leibiger IB; Caicedo A; Berggren P-O
- Article: INTEGRATIVE BIOLOGY. 2012;4(2):209-219Koehler M; Dare E; Ali MY; Rajasekaran SS; Moede T; Leibiger B; Leibiger IB; Tibell A; Juntti-Berggren L; Berggren P-O
- Article: ENDOCRINOLOGY. 2012;153(2):690-699Stucchi P; Gil-Ortega M; Merino B; Guzmán-Ruiz R; Cano V; Valladolid-Acebes I; Somoza B; Le Gonidec S; Argente J; Valet P; Chowen JA; Fernández-Alfonso M; Ruiz-Gayo M
- Article: EVIDENCE-BASED COMPLEMENTARY AND ALTERNATIVE MEDICINE. 2012;2012:452313-7Huyen VTT; Phan DV; Thang P; Ky PT; Hoa NK; Ostenson CG
- Article: PLOS ONE. 2012;7(5):e36139Morales L; Del Olmo N; Valladolid-Acebes I; Fole A; Cano V; Merino B; Stucchi P; Ruggieri D; López L; Alguacil LF; Ruiz-Gayo M
- Article: PLOS ONE. 2012;7(3):e33712Bergamini C; Moruzzi N; Sblendido A; Lenaz G; Fato R
- Article: PANCREAS. 2012;41(1):15-21Cui X; Yang G; Pan M; Zhang X-N; Yang S-N
- Article: CELL TRANSPLANTATION. 2012;21(4):633-648Cechin SR; Perez-Alvarez I; Fenjves E; Molano RD; Pileggi A; Berggren P-O; Ricordi C; Pastori RL
- Article: DIABETES RESEARCH AND CLINICAL PRACTICE. 2012;95(1):145-152Eriksson A-K; Gustavsson JP; Hilding A; Granath F; Ekbom A; Ostenson C-G
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2011;286(47):40857-40866Soty M; Visa M; Soriano S; Carmona MDC; Nadal Á; Novials A
- Meeting abstract: VALUE IN HEALTH. 2011;14(7):A480Theodorakis M; Reaney MD; Bruhn D; Matthaei S; Mathieu C; Kiljanski J; Guerci B; Sapin H; Ostenson CG; Salaun-Martin C; Krarup T
- Article: DIABETIC MEDICINE. 2011;28(11):1362-1372Fritz T; Caidahl K; Osler M; Oestenson CG; Zierath JR; Waendell P
- Article: GENE. 2011;486(1-2):94-96Gu HF; Gu T; Ostenson C-G; Karvestedt L; Brismar K
- Article: PEPTIDES. 2011;32(10):2077-2085Moreno P; Acitores A; Gutiérrez-Rojas I; Nuche-Berenguer B; El Assar M; Rodriguez-Mañas L; Gomis R; Valverde I; Visa M; Malaisse WJ; Novials A; González N; Villanueva-Peñacarrillo ML
- Article: DIABETES. 2011;60(10):2571-2577Nyqvist D; Speier S; Rodriguez-Diaz R; Molano RD; Lipovsek S; Rupnik M; Dicker A; Ilegems E; Zahr-Akrawi E; Molina J; Lopez-Cabeza M; Villate S; Abdulreda MH; Ricordi C; Caicedo A; Pileggi A; Berggren P-O
- Article: DIABETES. 2011;60(10):2624-2634Strawbridge RJ; Dupuis J; Prokopenko I; Barker A; Ahlqvist E; Rybin D; Petrie JR; Travers ME; Bouatia-Naji N; Dimas AS; Nica A; Wheeler E; Chen H; Voight BF; Taneera J; Kanoni S; Peden JF; Turrini F; Gustafsson S; Zabena C; Almgren P; Barker DJP; Barnes D; Dennison EM; Eriksson JG; Eriksson P; Eury E; Folkersen L; Fox CS; Frayling TM; Goel A; Gu HF; Horikoshi M; Isomaa B; Jackson AU; Jameson KA; Kajantie E; Kerr-Conte J; Kuulasmaa T; Kuusisto J; Loos RJF; Luan J; Makrilakis K; Manning AK; Teresa Martinez-Larrad M; Narisu N; Mannila MN; Ohrvik J; Osmond C; Pascoe L; Payne F; Sayer AA; Sennblad B; Silveira A; Stancakova A; Stirrups K; Swift AJ; Syvanen A-C; Tuomi T; van 't Hooft FM; Walker M; Weedon MN; Xie W; Zethelius B; Ongen H; Malarstig A; Hopewell JC; Saleheen D; Chambers J; Parish S; Danesh J; Kooner J; Ostenson C-G; Lind L; Cooper CC; Serrano-Rios M; Ferrannini E; Forsen TJ; Clarke R; Franzosi MG; Seedorf U; Watkins H; Froguel P; Johnson P; Deloukas P; Collins FS; Laakso M; Dermitzakis ET; Boehnke M; McCarthy MI; Wareham NJ; Groop L; Pattou F; Gloyn AL; Dedoussis GV; Lyssenko V; Meigs JB; Barroso I; Watanabe RM; Ingelsson E; Langenberg C; Hamsten A; Florez JC
- Article: THROMBOSIS AND HAEMOSTASIS. 2011;106(3):491-499Spectre G; Arnetz L; Ostenson C-G; Brismar K; Li N; Hjemdahl P
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2011;286(32):28026-28040Mandic SA; Skelin M; Johansson JU; Rupnik MS; Berggren P-O; Bark C
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2011;108(31):12863-12868Abdulreda MH; Faleo G; Molano RD; Lopez-Cabezas M; Molina J; Tan Y; Echeverria OAR; Zahr-Akrawi E; Rodriguez-Diaz R; Edlund PK; Leibiger I; Bayer AL; Perez V; Ricordi C; Caicedo A; Pileggi A; Berggren P-O
- Article: ENDOCRINOLOGY. 2011;152(8):3040-3048Storling J; Juntti-Berggren L; Olivecrona G; Prause MC; Berggren P-O; Mandrup-Poulsen T
- Article: PLOS ONE. 2011;6(7):e22510Oberg AI; Yassin K; Csikasz RI; Dehvari N; Shabalina IG; Hutchinson DS; Wilcke M; Ostenson C-G; Bengtsson T
- Article: CELL METABOLISM. 2011;14(1):45-54Rodriguez-Diaz R; Abdulreda MH; Formoso AL; Gans I; Ricordi C; Berggren P-O; Caicedo A
- Article: BLOOD CANCER JOURNAL. 2011;1(7):e31Buentke E; Nordstrom A; Lin H; Bjorklund A-C; Laane E; Harada M; Lu L; Tegnebratt T; Stone-Elander S; Heyman M; Soderhall S; Porwit A; Ostenson C-G; Shoshan M; Tamm KP; Grander D
- Review: JOURNAL OF MOLECULAR MEDICINE-JMM. 2011;89(7):635-645Luo J; Xiong Y; Han X; Lu Y
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2011;108(26):10685-10689Holmberg R; Refai E; Hoog A; Crooke RM; Graham M; Olivecrona G; Berggren P-O; Juntti-Berggren L
- Article: NATURE MEDICINE. 2011;17(7):888-892Rodriguez-Diaz R; Dando R; Jacques-Silva MC; Fachado A; Molina J; Abdulreda MH; Ricordi C; Roper SD; Berggren P-O; Caicedo A
- Article: INTERNATIONAL JOURNAL OF BIOMEDICAL SCIENCE. 2011;7(2):131-136Yassin K; Huyen VTT; Hoa KN; Ostenson CG
- Article: DIABETES RESEARCH AND CLINICAL PRACTICE. 2011;92(2):288-292Deleskog A; Hilding A; Ostenson C-G
- Article: DIABETOLOGIA. 2011;54(5):1121-1126Perez VL; Caicedo A; Berman DM; Arrieta E; Abdulreda MH; Rodriguez-Diaz R; Pileggi A; Hernandez E; Dubovy SR; Parel JM; Ricordi C; Kenyon NM; Kenyon NS; Berggren PO
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-HEART AND CIRCULATORY PHYSIOLOGY. 2011;300(4):H1174-H1181Gronros J; Jung C; Lundberg JO; Cerrato R; Ostenson C-G; Pernow J
- Article: PLOS ONE. 2011;6(3):e17858Guo N; Parry EM; Li L-S; Kembou F; Lauder N; Hussain MA; Berggren P-O; Armanios M
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2011;108(7):2957-2962Lu Y; Xiong Y; Huo Y; Han J; Yang X; Zhang R; Zhu D-S; Klein-Hessling S; Li J; Zhang X; Han X; Li Y; Sheng B; He Y; Shibuya M; Feng G-S; Luo J
- Corrigendum: EXPERIMENTAL DIABETES RESEARCH. 2011;:319202-1Pan M; Yang G; Cui X; Yang S-N
- Review: SHENG LI KE XUE JIN ZHAN [PROGRESS IN PHYSIOLOGY]. 2011;42(1):6-10Xiong Y; Han X-F; Luo J-C
- Article: NEUROBIOLOGY OF LEARNING AND MEMORY. 2011;95(1):80-85Valladolid-Acebes I; Stucchi P; Cano V; Fernández-Alfonso MS; Merino B; Gil-Ortega M; Fole A; Morales L; Ruiz-Gayo M; Del Olmo N
- Article: EXPERIMENTAL DIABETES RESEARCH. 2011;2011:604989-7Pan M; Yang G; Cui X; Yang S-N
- Review: LAKARTIDNINGEN. 2010;107(45):2792-2795Ostenson C-G
- Article: LAKARTIDNINGEN. 2010;107(45):2791Ostenson C-G
- Review: CHEMICO-BIOLOGICAL INTERACTIONS. 2010;188(2):301-308Ceccatelli S; Dare E; Moors M
- Article: ENVIRONMENTAL RESEARCH. 2010;110(8):764-772Eriksson C; Bluhm G; Hilding A; Ostenson C-G; Pershagen G
- Article: DOKLADY BIOLOGICAL SCIENCES. 2010;435:418-420Voznesenskaya AE; Ambaryan AV; Klyuchnikova MA; Kotenkova EV; Voznesenskaya VV
- Article: JOURNAL OF MEDICAL CASE REPORTS. 2010;4:315Bränström R; Berglund E; Curman P; Forsberg L; Höög A; Grimelius L; Berggren P-O; Mattsson P; Hellman P; Juntti-Berggren L
- Article: BMC BIOCHEMISTRY. 2010;11:38Gustavsson C; Yassin K; Wahlstrom E; Cheung L; Lindberg J; Brismar K; Ostenson C-G; Norstedt G; Tollet-Egnell P
- Article: CELL METABOLISM. 2010;12(3):237-249Wunderlich FT; Stroehle P; Koenner AC; Gruber S; Tovar S; Broenneke HS; Juntti-Berggren L; Li L-S; van Rooijen N; Libert C; Berggren P-O; Bruening JC
- Article: EUROPEAN JOURNAL OF ENDOCRINOLOGY. 2010;163(2):233-242Lewitt MS; Hilding A; Brismar K; Efendic S; Ostenson C-G; Hall K
- Article: ARCHIVES OF BIOCHEMISTRY AND BIOPHYSICS. 2010;499(1-2):62-68Hasan NM; Longacre MJ; Ahmed MS; Kendrick MA; Gu H; Ostenson C-G; Fukao T; MacDonald MJ
- Article: ORGANIC & BIOMOLECULAR CHEMISTRY. 2010;8(12):2850-2858Mansell D; Rattray N; Etchells LL; Schwalbe CH; Blake AJ; Torres J; Kremer C; Bichenkova EV; Barker CJ; Freeman S
- Article: FASEB JOURNAL. 2010;24(6):1824-1837Leibiger B; Moede T; Uhles S; Barker CJ; Creveaux M; Domin J; Berggren P-O; Leibiger IB
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2010;396(1):111-115Leibiger IB; Brismar K; Berggren P-O
- Article: HORMONE AND METABOLIC RESEARCH. 2010;42(5):353-357Huyen VTT; Phan DV; Thang P; Hoa NK; Ostenson CG
- Article: LAKARTIDNINGEN. 2010;107(15):976-980Osterlind J; Eriksson MK; Ostenson C-G; Eliasson M
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2010;107(14):6465-6470Jacques-Silva MC; Correa-Medina M; Cabrera O; Rodriguez-Diaz R; Makeeva N; Fachado A; Diez J; Berman DM; Kenyon NS; Ricordi C; Pileggi A; Molano RD; Berggren P-O; Caicedo A
- Article: EXPERIMENTAL AND CLINICAL ENDOCRINOLOGY & DIABETES. 2010;118(4):220-225Elrayah-Eliadarous H; Yassin K; Eltom M; Abdelrahman S; Wahlstrom R; Ostenson C-G
- Article: SCIENCE SIGNALING. 2010;3(113):ra19Healy JA; Nilsson KR; Hohmeier HE; Berglund J; Davis J; Hoffman J; Kohler M; Li L-S; Berggren P-O; Newgard CB; Bennett V
- Article: JOURNAL OF ENDOCRINOLOGY. 2010;204(3):255-263Foo KS; Brauner H; Ostenson C-G; Broberger C
- Article: DIABETES CARE. 2010;33(3):658-660Leitao CB; Bernetti K; Tharavanij T; Cure P; Lauriola V; Berggren P-O; Ricordi C; Alejandro R
- Article: JOURNAL OF ETHNOPHARMACOLOGY. 2010;127(2):346-351Manneras L; Fazliana M; Nazaimoon WMW; Lonn M; Gu HF; Ostenson CG; Stener-Victorin E
- Article: METHODS IN MOLECULAR BIOLOGY. 2010;645:21-46Barker CJ; Illies C; Berggren P-O
- Article: METHODS IN MOLECULAR BIOLOGY. 2010;645:123-131Nagel A; Barker CJ; Berggren P-O; Illies C
- Review: METHODS IN MOLECULAR BIOLOGY. 2010;645:1-19Barker CJ; Berggren P-O
- Article: DIABETES. 2010;59(1):190-199Fornoni A; Jeon J; Santos JV; Cobianchi L; Jauregui A; Inverardi L; Mandic SA; Bark C; Johnson K; McNamara G; Pileggi A; Molano RD; Reiser J; Tryggvason K; Kerjaschki D; Berggren P-O; Mundel P; Ricordi C
- Review: CELLULAR AND MOLECULAR LIFE SCIENCES. 2009;66(24):3851-3871Barker CJ; Illies C; Gaboardi GC; Berggren P-O
- Article: JOURNAL OF PEPTIDE SCIENCE. 2009;15(12):842-848Catrina S-B; Refai E; Andersson M
- Article: CELLULAR AND MOLECULAR LIFE SCIENCES. 2009;66(23):3787-3795Zaitseva II; Hultcrantz M; Sharoyko V; Flodstrom-Tullberg M; Zaitsev SV; Berggren P-O
- Meeting abstract: EPIDEMIOLOGY. 2009;20(6):S140Eriksson C; Bluhm G; Hilding A; Ostenson C-G; Pershagen G
- Article: MICROBIOLOGY AND IMMUNOLOGY. 2009;53(10):567-572Holmberg R; Klitz W; Blixt M; Berggren P-O; Juntti-Berggren L; Niklasson B
- Article: BRITISH JOURNAL OF PHARMACOLOGY. 2009;158(2):486-493Chen Z; Leibiger I; Katz AI; Bertorello AM
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2009;284(35):23217-23224Xiong Y; Huo Y; Chen C; Zeng H; Lu X; Wei C; Ruan C; Zhang X; Hu Z; Shibuya M; Luo J
- Article: INTERNATIONAL JOURNAL OF TECHNOLOGY ASSESSMENT IN HEALTH CARE. 2009;25(3):350-358Johansson P; Ostenson C-G; Hilding A; Andersson C; Rehnberg C; Tillgren P
- Article: DIABETOLOGIA. 2009;52(6):1087-1091MacDonald MJ; Longacre MJ; Langberg E-C; Tibell A; Kendrick MA; Fukao T; Ostenson C-G
- Article: EXPERIMENTAL AND CLINICAL ENDOCRINOLOGY & DIABETES. 2009;117(4):186-190Nordman S; Ostenson C-G; Efendic S; Gu HF
- Article: INTEGRATIVE BIOLOGY. 2009;1(4):301-310Abdulreda MH; Bhalla A; Rico F; Berggren P-O; Chapman ER; Moy VT
- Article: METHODS AND FINDINGS IN EXPERIMENTAL AND CLINICAL PHARMACOLOGY. 2009;31(3):165-169Hoa NK; Phan DV; Thuan ND; Ostenson C-G
- Article: DIABETIC MEDICINE. 2009;26(4):345-353Alvarsson M; Hilding A; Ostenson C-G
- Article: METABOLISM-CLINICAL AND EXPERIMENTAL. 2009;58(4):449-455Hager P; Permert J; Wikstrom A-C; Herrington MK; Ostenson C-G; Strommer L
- Article: BIOCHEMICAL PHARMACOLOGY. 2009;77(7):1236-1245Margolies-Clark E; Jacques-Silva MC; Ganesan L; Umland O; Kenyon NS; Ricordi C; Berggren P-O; Buchwald P
- Article: BMC MEDICAL GENETICS. 2009;10:27Dahlman I; Nilsson M; Gu HF; Lecoeur C; Efendic S; Ostenson CG; Brismar K; Gustafsson J-A; Froguel P; Vaxillaire M; Dahlman-Wright K; Steffensen KR
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-HEART AND CIRCULATORY PHYSIOLOGY. 2009;296(3):H719-H727Bulhak AA; Jung C; Ostenson C-G; Lundberg JO; Sjoquist P-O; Pernow J
- Article: MATURITAS. 2009;62(1):91-97Fazliana M; Nazaimoon WMW; Gu HF; Ostenson C-G
- Article: CELL TRANSPLANTATION. 2009;18(1):23-30Lau J; Kampf C; Mattsson G; Nyqvist D; Kohler M; Berggren P-O; Carlsson P-O
- Article: JOURNAL OF BIOLOGICAL INORGANIC CHEMISTRY. 2009;14(1):51-59Veiga N; Torres J; Mansell D; Freeman S; Dominguez S; Barker CJ; Diaz A; Kremer C
- Review: ADVANCES IN ENZYME REGULATION. 2009;49(1):168-173Barker CJ; Illies C; Fiume R; Gaboardi GC; Yu J; Berggren P-O
- Article: PHYSIOLOGY & BEHAVIOR. 2008;95(5):668-676Bjorklund O; Halldner-Henriksson L; Yang J; Eriksson TM; Jacobson MA; Dare E; Fredholm BB
- Article: JOURNAL OF THROMBOSIS AND HAEMOSTASIS. 2008;6(12):2186-2192Razmara M; Hjemdahl P; Ostenson C-G; Li N
- Article: REGULATORY PEPTIDES. 2008;151(1-3):139-146Mosen H; Ostenson C-G; Lundquist I; Alm P; Henningsson R; Jimenez-Feltstrom J; Guenifi A; Efendic S; Salehi A
- Article: CHEMICAL COMMUNICATIONS. 2008;(41):5161-5163Mansell D; Rattray N; Etchells LL; Schwalbe CH; Blake AJ; Bichenkova EV; Bryce RA; Barker CJ; Diaz A; Kremer C; Freeman S
- Article: JOURNAL OF ENDOCRINOLOGY. 2008;199(2):275-286Lundholm L; Bryzgalova G; Gao H; Portwood N; Falt S; Berndt KD; Dicker A; Galuska D; Zierath IR; Gustafsson JA; Efendic S; Dahlman-Wright K; Khan A
- Article: PLOS GENETICS. 2008;4(11):e1000278Johansson JU; Ericsson J; Janson J; Beraki S; Stanic D; Mandic SA; Wikstrom MA; Hokfelt T; Ogren SO; Rozell B; Berggren P-O; Bark C
- Letter: EUROPEAN RESPIRATORY JOURNAL. 2008;32(5):1420-1421Catrina S-B; Juntti-Berggren L
- Article: PHARMACOGENETICS AND GENOMICS. 2008;18(11):967-975Dahlman I; Vaxillaire M; Nilsson M; Lecoeur C; Gu HF; Cavalcanti-Proenca C; Efendic S; Ostenson CG; Brismar K; Charpentler G; Gustafsson J-A; Froguel P; Dahlman-Wright K; Steffensen KR
- Article: EUROPEAN JOURNAL OF PHARMACOLOGY. 2008;596(1-3):6-13Bjorklund O; Shang M; Tonazzini I; Dare E; Fredholm BB
- Article: SCANDINAVIAN JOURNAL OF PSYCHOLOGY. 2008;49(5):459-467Gustavsson JP; Eriksson A-K; Hilding A; Gunnarsson M; Ostensson C-G
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2008;295(4):E904-E912Bryzgalova G; Lundholm L; Portwood N; Gustafsson J-A; Khan A; Efendic S; Dahlman-Wright K
- Review: EXPERT REVIEW OF CLINICAL IMMUNOLOGY. 2008;4(5):615-627Kadri N; Blomqvist M; Cardell SL
- Conference publication: DIABETOLOGIA. 2008;51:S202-S203Leibiger B; Moede T; Barker CJ; Uhles S; Domin J; Berggren P-O; Leibiger IB
- Article: CANCER RESEARCH. 2008;68(17):7156-7164Lui W-O; Zeng L; Rehmann V; Deshpande S; Tretiakova M; Kaplan EL; Leibiger I; Leibiger B; Enberg U; Hoog A; Larsson C; Kroll TG
- Article: JOURNAL OF STEROID BIOCHEMISTRY AND MOLECULAR BIOLOGY. 2008;111(3-5):262-267Bryzgalova G; Effendic S; Khan A; Rehnmark S; Barbounis P; Boulet J; Dong G; Singh R; Shapses S; Malm J; Webb P; Baxter JD; Grover GJ
- Corrigendum: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2008;373(1):178Quynh NTT; Islam MS; Floren A; Bartfai T; Langel U; Ostenson C-G
- Review: ANNUAL REVIEW OF NUTRITION. 2008;28:233-251Leibiger IB; Leibiger B; Berggren P-O
- Article: DIABETOLOGIA. 2008;51(7):1135-1145Lewitt MS; Hilding A; Ostenson C-G; Efendic S; Brismar K; Hall K
- Article: DIABETIC MEDICINE. 2008;25(7):834-842Eriksson A-K; Ekbom A; Granath F; Hilding A; Efendic S; Ostenson C-G
- Article: CELL METABOLISM. 2008;7(6):545-554Cabrera O; Jacques-Silva MC; Speier S; Yang S-N; Kohler M; Fachado A; Vieira E; Zierath JR; Kibbey R; Berman DM; Kenyon NS; Ricordi C; Caicedo A; Berggren P-O
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2008;105(20):7246-7251Bertolino P; Holmberg R; Reissmann E; Andersson O; Berggren P-O; Ibanez CF
- Article: PLOS ONE. 2008;3(5):e2165Salehi A; Abaraviciene SM; Jimenez-Feltstrom J; Ostenson C-G; Efendic S; Lundquist I
- Article: FEBS LETTERS. 2008;582(11):1613-1617Shafqat J; Ishrat M; Jagerbrink T; Sillard R; Maeorg U; Efendic S; Berggren P-O; Zaitsev SV; Jornvall H
- Article: NATURE MEDICINE. 2008;14(5):574-578Speier S; Nyqvist D; Cabrera O; Yu J; Molano RD; Pileggi A; Moede T; Kohler M; Wilbertz J; Leibiger B; Ricordi C; Leibiger IB; Caicedo A; Berggren P-O
- Article: CELLULAR AND MOLECULAR LIFE SCIENCES. 2008;65(7-8):1248-1255Zaitseva II; Storling J; Mandrup-Poulsen T; Berggren P-O; Zaitsev SV
- Article: INTERNATIONAL JOURNAL OF OBESITY. 2008;32(3):407-412Nordman S; Abulaiti A; Hilding A; Langberg E-C; Humphreys K; Ostenson C-G; Efendic S; Gu HF
- Article: DIABETES-METABOLISM RESEARCH AND REVIEWS. 2008;24(2):148-154Sundsten T; Ostenson CG; Bergsten P
- Article: NATURE PROTOCOLS. 2008;3(8):1278-1286Noninvasive high-resolution in vivo imaging of cell biology in the anterior chamber of the mouse eyeSpeier S; Nyqvist D; Koehler M; Caicedo A; Leibiger IB; Berggren P-O
- Article: EXPERIMENTAL DIABETES RESEARCH. 2008;2008:865850Paulsson JF; Schultz S; Kohler M; Leibiger I; Berggren P-O; Westermark GT
- Review: ADVANCES IN ENZYME REGULATION. 2008;48:276-294Berggren P-O; Barker CJ
- Article: TOXICOLOGY. 2007;241(3):119-133Bjoerklund O; Kahlstroem J; Salmi P; Oegren SO; Vahter M; Chen J-F; Fredholm BB; Dare E
- Article: SCIENCE. 2007;318(5854):1299-1302Illies C; Gromada J; Fiume R; Leibiger B; Yu J; Juhl K; Yang S-N; Barma DK; Falck JR; Saiardi A; Barker CJ; Berggren P-O
- Article: BIOSCIENCE REPORTS. 2007;27(6):321-326Dekki N; Nilsson R; Norgren S; Roessner SM; Appelskog I; Marcus C; Simell O; Pugliese A; Alejandro R; Ricordi C; Berggren PO; Juntti-Berggren L
- Article: CELL TRANSPLANTATION. 2007;16(10):1039-1048Cabrera O; Jacques-Silva MC; Berman DM; Fachado A; Echeverri F; Poo R; Khan A; Kenyon NS; Ricordi C; Berggren P-O; Caicedo A
- Published conference paper: DIABETES OBESITY & METABOLISM. 2007;9:180-186Ostenson C-G; Efendic S
- Article: MOLECULAR ENDOCRINOLOGY. 2007;21(11):2775-2784Yu J; Berggren P-O; Barker CJ
- Article: CELLULAR AND MOLECULAR LIFE SCIENCES. 2007;64(22):2985-2993Sharoyko VV; Zaitseva II; Leibiger B; Efendic S; Berggren P-O; Zaitsev SV
- Article: EPIDEMIOLOGY. 2007;18(6):716-721Eriksson C; Rosenlund M; Pershagen G; Hilding A; Ostenson C-G; Bluhm G
- Article: BMC HEALTH SERVICES RESEARCH. 2007;7:162Abdulhadi N; Al Shafaee M; Freudenthal S; Ostenson C-G; Wahlstrom R
- Article: EUROPEAN JOURNAL OF ENDOCRINOLOGY. 2007;157(4):459-464Langberg E-C; Gu HF; Nordman S; Efendic S; Oestenson C-G
- Article: GROWTH HORMONE & IGF RESEARCH. 2007;17(5):392-398Strawbridge RJ; Karvestedt L; Li C; Efendic S; Ostenson CG; Gu HF; Brismar K
- Article: JOURNAL OF CEREBRAL BLOOD FLOW AND METABOLISM. 2007;27(10):1710-1723Moreira TJTP; Cebere A; Cebers G; Ostenson C-G; Efendic S; Liljequist S
- Article: PHYSIOLOGY & BEHAVIOR. 2007;92(1-2):15-20Dare E; Schulte G; Karovic O; Hammarberg C; Fredholm BB
- Corrigendum: DIABETOLOGIA. 2007;50(10):2227Niklasson B; Hultman T; Kallies R; Niedrig M; Nilsson R; Berggren P-O; Juntti-Berggren L; Efendic S; Lernmark A; Klitz W
- Article: PSYCHONEUROENDOCRINOLOGY. 2007;32(8-10):1041-1051Brismar T; Maurex L; Cooray G; Juntti-Berggren L; Lindstrom P; Ekberg K; Adner N; Andersson S
- Article: CELL METABOLISM. 2007;6(3):217-228Yang S-N; Wenna ND; Yu J; Yang G; Qiu H; Yu L; Juntti-Berggren L; Koher M; Berggren P-O
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2007;293(3):E794-E801Richard A-MT; Webb D-L; Goodman JM; Schultz V; Flanagan JN; Getty-Kaushik L; Deeney JT; Yaney GC; Dunaway GA; Berggren P-O; Tornheim K
- Article: DIABETES CARE. 2007;30(9):2271-2276Jonasson JM; Brismar K; Sparen P; Lambe M; Nyren O; Ostenson C-G; Ye W
- Article: FEBS LETTERS. 2007;581(21):4080-4084Deeney JT; Branstrom R; Corkey BE; Larsson O; Berggren P-O
- Article: TRANSPLANTATION. 2007;84(3):308-315Berman DM; Cabrera O; Kenyon NM; Miller J; Tam SH; Khandekar VS; Picha KM; Soderman AR; Jordan RE; Bugelski PJ; Horninger D; Lark M; Davis JE; Alejandro R; Berggren P-O; Zimmerman M; O'Neil JJ; Ricordi C; Kenyon NS
- Article: DIABETOLOGIA. 2007;50(8):1670-1677Braenstroem R; Leibiger IB; Leibiger B; Klement G; Nilsson J; Arhem P; Aspinwall CA; Corkey BE; Larsson O; Berggren R-O
- Letter: DIABETOLOGIA. 2007;50(7):1559-1560Niklasson B; Hultman T; Kallies R; Niedrig M; Nilsson R; Berggren P-O; Juntti-Berggren L; Efendic S; Lernmark A; Klitz W
- Article: BEHAVIOURAL BRAIN RESEARCH. 2007;180(1):28-41Moreira T; Malec E; Ostenson C-G; Efendic S; Liljequist S
- Article: DIABETES. 2007;56(6):1544-1550Lau J; Mattsson G; Carlsson C; Nyqvist D; Kohler M; Berggren P-O; Jansson L; Carlsson P-O
- Article: JOURNAL OF HEALTH COMMUNICATION. 2007;12(3):269-283Andersson CM; Bjaras G; Tillgren P; Ostenson C-G
- Article: CELLULAR AND MOLECULAR LIFE SCIENCES. 2007;64(10):1310-1316Jagerbrink T; Lexander H; Palmberg C; Shafqat J; Sharoyko V; Berggren P-O; Efendic S; Zaitsev S; Jornvall H
- Published conference paper: ACTA PHYSIOLOGICA. 2007;190(1):63-75Yang J-N; Tiselius C; Dare E; Johansson B; Valen G; Fredholm BB
- Article: FASEB JOURNAL. 2007;21(7):1609-1621Uhles S; Moede T; Leibiger B; Berggren P-O; Leibiger IB
- Article: PANCREAS. 2007;34(3):359-363Ostenson C-G; Chen J; Sheu L; Gaisano HY
- Article: NATURE CELL BIOLOGY. 2007;9(4):453-460Zhang Q; Bengtsson M; Partridge C; Salehi A; Braun M; Cox R; Eliasson L; Johnson PRV; Renstrom E; Schneider T; Berggren P-O; Gopel S; Ashcroft FM; Rorsman P
- Article: BIOCHEMICAL PHARMACOLOGY. 2007;73(5):694-708Karovic O; Tonazzini I; Rebola N; Edstrom E; Lovdahl C; Fredholm BB; Dare E
- Article: CLINICAL SCIENCE. 2007;112(5-6):345-351Svensson AM; Ostenson C-G; Efendic S; Jansson L
- Review: NEUROSCIENCE. 2007;144(4):1169-1185Moreira T; Cebers G; Pickering C; Ostenson C-G; Efendic S; Liljequist S
- Article: IMMUNOLOGY LETTERS. 2007;108(2):129-136Kadri N; Potiron N; Ouary M; Jegou D; Gouin E; Bach JM; Lieubeau B
- Article: INTERNATIONAL JOURNAL OF EPIDEMIOLOGY. 2007;36(1):84-92Agardh EE; Ahlbom A; Andersson T; Efendic S; Grill V; Hallqvist J; Ostenson CG
- Article: FEBS JOURNAL. 2007;274(3):751-759Dun X-P; Wang J-H; Chen L; Lu J; Li F-F; Zhao Y-Y; Cederlund E; Bryzgalova G; Efendic S; Jornvall H; Chen Z-W; Bergman T
- Article: JOURNAL OF ENDOCRINOLOGY. 2007;192(2):389-394Hoa NK; Norberg A; Sillard R; Van Phan D; Thuan ND; Dzung DTN; Jornvall H; Ostenson C-G
- Published conference paper: CHEMOTHERAPY. 2007;53(3):169-172Abo EL-Sooud K; Khammas W; Kadri MN
- Article: DIABETES CARE. 2007;30(1):138-140Razmara M; Hjemdahl P; Yngen M; Ostenson C-G; Wallen NH; Li N
- Article: DIABETES CARE. 2007;30(1):71-76Ekberg K; Brismar T; Johansson B-L; Lindstrom P; Juntti-Berggren L; Norbhy A; Berne C; Arnqvist HJ; Bolinder J; Wahren J
- Article: CELL TRANSPLANTATION. 2007;16(6):621-627von Seth E; Nyqvist D; Andersson A; Carlsson P-O; Koehler M; Mattsson G; Nordin A; Berggrenj P-O; Jansson L
- Article: BMC FAMILY PRACTICE. 2006;7:72Abdulhadi N; Al-Shafaee MA; Ostenson C-G; Vernby A; Wahlström R
- Article: DIABETES. 2006;55(12):3299-3308Miettinen PJ; Ustinov J; Ormio P; Gao R; Palgi J; Hakonen E; Juntti-Berggren L; Berggren P-O; Otonkoski T
- Article: CELLULAR AND MOLECULAR LIFE SCIENCES. 2006;63(24):3055-3060Henriksson M; Johansson J; Moede T; Leibiger I; Shafqat J; Berggren P-O; Jornvall H
- Article: DIABETOLOGIA. 2006;49(11):2589-2598Hilding A; Eriksson A-K; Agardh EE; Grill V; Ahlbom A; Efendic S; Ostenson C-G
- Article: HORMONE AND METABOLIC RESEARCH. 2006;38(10):683-687Meyer C; Carlqvist H; Vorgerd M; Schoels L; Ostenson C-G; Ristow M
- Review: ENDOCRINE REVIEWS. 2006;27(6):621-676Yang S-N; Berggren P-O
- Article: FEBS LETTERS. 2006;580(21):5067-5070Efendiev R; Cinelli AR; Leibiger IB; Bertorello AM; Pedemonte CH
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2006;347(4):1121-1128Zaitseva II; Sharoyko V; Storling J; Efendic S; Guerin C; Mandrup-Poulsen T; Nicotera P; Berggren P-O; Zaitsev SV
- Article: BRAIN RESEARCH. 2006;1098(1):1-8Tofighi R; Tillmark N; Dare E; Aberg AM; Larsson JE; Ceccatelli S
- Article: AMERICAN JOURNAL OF RESPIRATORY CELL AND MOLECULAR BIOLOGY. 2006;35(1):127-132Chen Z; Krmar RT; Dada L; Efendiev R; Leibiger IB; Pedemonte CH; Katz AI; Sznajder JI; Bertorello AM
- Article: MOLECULAR ENDOCRINOLOGY. 2006;20(6):1287-1299Gao H; Bryzgalova G; Hedman E; Khan A; Efendic S; Gustafsson JA; Dahlman-Wright K
- Article: ENDOCRINE-RELATED CANCER. 2006;13(2):509-523Juhlin C; Larsson C; Yakoleva T; Leibiger I; Leibiger B; Alimov A; Weber G; Hoog A; Villablanca A
- Article: CELL CALCIUM. 2006;39(6):529-537Woolcott OO; Gustafsson AJ; Dzabic M; Pierro C; Tedeschi P; Sandgren J; Bari MR; Hoa NK; Bianchi M; Rakonjac M; Radmark O; Ostenson C-G; Islam MS
- Published conference paper: NUTRITION METABOLISM AND CARDIOVASCULAR DISEASES. 2006;16:S7-10Berggren PO; Leibiger IB
- Article: DIABETES. 2006;55(3):590-599Warwar N; Efendic S; Östenson CG; Haber EP; Cerasi E; Nesher R
- Article: DIABETES. 2006;55(3):574-581Zhang W; Lilja L; Mandic SA; Gromada J; Smidt K; Janson J; Takai Y; Bark C; Berggren PO; Meister B
- Article: DIABETOLOGIA. 2006;49(3):588-597Bryzgalova G; Gao H; Ahren B; Zierath JR; Galuska D; Steiler TL; Dahlman-Wright K; Nilsson S; Gustafsson J; Efendic S; Khan A
- Article: LAKARTIDNINGEN. 2006;103(6):375-377Sydhoff J; Josefsson L; Cronlund I; Hollenberg S; Kanström L; Ostenson C-G
- Article: DIABETES. 2006;55(2):435-440Ostenson CG; Gaisano H; Sheu L; Tibell A; Bartfai T
- Published conference paper: DIABETIC MEDICINE. 2006;23(2):134-140Yngen M; Östenson CG; Hjemdahl P; Wallén NH
- Editorial: NATURE MEDICINE. 2006;12(1):34-36Leibiger IB; Berggren PO
- Article: SCANDINAVIAN JOURNAL OF PUBLIC HEALTH. 2006;34(6):665-668The magnitude of bias in a cross-sectional study on lifestyle factors in relation to type 2 diabetesAgardh EE; Ahlbom A; Andersson T; Ostenson C-G
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2005;338(3):1455-1459Sharoyko VV; Zaitseva II; Varsanyi M; Portwood N; Leibiger B; Leibiger I; Berggren PO; Efendic S; Zaitsev SV
- Article: INTERNATIONAL JOURNAL OF PEPTIDE RESEARCH AND THERAPEUTICS. 2005;11(4):237-247Östlund P; Kilk K; Lindgren M; Hällbrink M; Jiang Y; Budihna M; Cerne K; Bavec A; Östenson CG; Zorko M; Langel Ü
- Article: SOCIAL SCIENCE & MEDICINE. 2005;61(11):2407-2422Andersson CM; Bjärås G; Tillgren P; Östenson CG
- Article: INTERNATIONAL JOURNAL OF OBESITY. 2005;29(12):1422-1428Lavebratt C; Sengul S; Gu HF; Persson B; Nordfors L; Östenson CG; Efendic S; Arner P; Hoffstedt J; Schalling M
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2005;102(47):17020-17025Refai E; Dekki N; Yang SN; Imreh G; Cabrera O; Yu LN; Yang G; Norgren S; Rössner SM; Inverardi L; Ricordi C; Olivecrona G; Andersson M; Jörnvall H; Berggren PO; Juntti-Berggren L
- Article: DIABETES RESEARCH AND CLINICAL PRACTICE. 2005;70(2):159-165Elrayah H; Eltom M; Bedri A; Belal A; Rosling H; Östenson CG
- Article: INFECTION AND IMMUNITY. 2005;73(10):6332-6339Linde CMA; Grundström S; Nordling E; Refai E; Brennan PJ; Andersson M
- Article: BRAIN RESEARCH. 2005;138(2):215-227Johansson JU; Lilja L; Chen XL; Higashida H; Meister B; Noda M; Zhong ZG; Yokoyama S; Berggren PO; Bark C
- Editorial: CELL METABOLISM. 2005;2(2):80-82Leibiger IB; Berggren PO
- Article: JOURNAL OF ENDOCRINOLOGY. 2005;186(2):333-341Nyqvist D; Mattsson G; Köhler M; Lev-Ram V; Andersson A; Carlsson PO; Nordin A; Berggren PO; Jansson L
- Article: DIABETES. 2005;54(8):2287-2293Nyqvist D; Köhler M; Wahlstedt H; Berggren PO
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2005;102(29):10303-10308Gromada J; Bark C; Smidt K; Efanov AM; Janson J; Mandic SA; Webb DL; Zhang W; Meister B; Jeromin A; Berggren PO
- Article: AMERICAN JOURNAL OF TRANSPLANTATION. 2005;5(7):1635-1645Ichii H; Inverardi L; Pileggi A; Molano RD; Cabrera O; Caicedo A; Messinger S; Kuroda Y; Berggren PO; Ricordi C
- Article: ENDOCRINOLOGY. 2005;146(7):3026-3036Storling J; Zaitsev SV; Kapelioukh IL; Karlsen AE; Billestrup N; Berggren PO; Mandrup-Poulsen T
- Article: DIABETES CARE. 2005;28(6):1495-1497Jörneskog G; Kalani M; Kuhl J; Båvenholm P; Katz A; Allerstrand G; Alvarsson M; Efendic S; Östenson CG; Pernow J; Wahren J; Brismar K
- Article: EXPERIMENTAL AND CLINICAL ENDOCRINOLOGY & DIABETES. 2005;113(5):282-287Nordman S; Ding B; Östenson CG; Kärvestedt L; Brismar K; Efendic S; Gu HF
- Letter: DIABETES CARE. 2005;28(5):1261Öberg D; Östenson CG
- Article: PANCREAS. 2005;30(4):314-317Chen J; Östenson CG
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-GASTROINTESTINAL AND LIVER PHYSIOLOGY. 2005;288(5):G972-G977Ghanem E; Lövdahl C; Daré E; Ledent C; Fredholm BB; Boeynaems JM; Van Driessche W; Beauwens R
- Article: REGULATORY PEPTIDES. 2005;127(1-3):45-53Ding B; Kull B; Liu ZR; Mottagui-Tabar S; Thonberg H; Gu HF; Brookes AJ; Grundemar L; Karlsson C; Hamsten A; Arner P; Östenson CG; Efendic S; Monné M; von Heijne G; Eriksson P; Wahlestedt C
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2005;329(2):673-677Lilja L; Meister B; Berggren PO; Bark C
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2005;328(1):213-220Quynh NTT; Islam MS; Florén A; Bartfai T; Langel Ü; Östenson CG
- Article: ENDOCRINOLOGY. 2005;146(3):1553-1558Mosén H; Salehi A; Alm P; Henningsson R; Jimenez-Feltström J; Ostenson C-G; Efendic S; Lundquist I
- Article: JOURNAL OF ENDOCRINOLOGY. 2005;184(2):319-327Svensson AM; Östenson CG; Bodin B; Jansson L
- Article: TOXICOLOGY IN VITRO. 2005;19(1):11-20Vettori MV; Caglieri A; Goldoni M; Castoldi AF; Darè E; Alinovi R; Ceccatelli S; Mutti A
- Article: FASEB JOURNAL. 2005;18(14):301-303Gustafsson AJ; Ingelman-Sundberg H; Dzabic M; Awasum J; Hoa NK; Östenson CG; Pierro C; Tedeschi P; Woolcott O; Chiounan S; Lund PE; Larsson O; Islam MS
- Editorial: JOURNAL OF CLINICAL INVESTIGATION. 2005;115(1):16-20Yang SN; Berggren PO
- Review: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2005;288(1):E16-E28Yang SN; Berggren PO
- Article: DIABETOLOGIA. 2005;48(1):35-40Kuhl J; Hilding A; Östenson CG; Grill V; Efendic S; Båvenholm P
- Article: MOLECULAR ENDOCRINOLOGY. 2005;19(1):198-212Ma XS; Zhang Y; Gromada J; Sewing S; Berggren PO; Buschard K; Salehi A; Vikman J; Rorsman P; Eliasson L
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2004;325(2):555-560Chen J; Östenson CG
- Review: LAKARTIDNINGEN. 2004;101(49):4011-4015Ostenson C-G; Båvenholm P; Efendic S
- Published conference paper: DIABETES. 2004;53:S181-S189Gromada J; Ma XS; Hoy M; Bokvist K; Salehi A; Berggren PO; Rorsman P
- Article: CELL. 2004;119(2):273-284Berggren PO; Yang SN; Murakami M; Efanov AM; Uhles S; Köhler M; Moede T; Fernström A; Appelskog LB; Aspinwall CA; Zaitsev SV; Larsson O; de Vargas LM; Fecher-Trost C; Weissgerber P; Ludwig A; Leibiger B; Juntti-Berggren L; Barker CJ; Gromada J; Freichel M; Leibiger IB; Flockerzi V
- Article: EXPERIMENTAL AND CLINICAL ENDOCRINOLOGY & DIABETES. 2004;112(9):520-525Hoa NK; Phan DV; Thuan ND; Östenson CG
- Article: NAUNYN-SCHMIEDEBERGS ARCHIVES OF PHARMACOLOGY. 2004;370(4):270-278Halldner L; Lopes LV; Daré E; Lindström K; Johansson B; Ledent C; Cunha RA; Fredholm BB
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2004;279(40):41361-41367Norberg Å; Hoa NK; Liepinsh E; Van Phan D; Thuan ND; Jörnvall H; Sillard R; Östenson CG
- Article: REGULATORY PEPTIDES. 2004;121(1-3):107-112Xie L; Lu J; Östenson CG; Xu T; Chen ZW
- Article: TOXICOLOGY. 2004;201(1-3):125-132Daré E; Tofighi R; Nutt L; Vettori MV; Emgård A; Mutti A; Ceccatelli S
- Article: DIABETOLOGIA. 2004;47(Suppl 1):a1-a464Veitenhansl M; Stegner K; Hierl FX; Dieterle C; Feldmeier H; Gutt B; Landgraf R
- Article: DIABETES. 2004;53(8):2137-2142Gu HF; Efendic S; Nordman S; Östenson CG; Brismar K; Brookes AJ; Prince JA
- Article: NATURE IMMUNOLOGY. 2004;5(8):836-843Hornef MW; Pütsep K; Karlsson J; Refai E; Andersson M
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2004;320(3):717-721Zhang W; Lilja L; Bark C; Berggren PO; Meister B
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2004;279(28):29534-29541Lilja L; Johansson JU; Gromada J; Mandic SA; Fried G; Berggren PO; Bark C
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2004;101(27):10090-10094Juntti-Berggren L; Refai E; Appelskog I; Andersson M; Imreh G; Dekki N; Uhles S; Yu L; Griffiths WJ; Zaitsev S; Leibiger I; Yang SN; Olivecrona G; Jörnvall H; Berggren PO
- Article: MOLECULAR ENDOCRINOLOGY. 2004;18(7):1658-1669Zhang QM; Köhler M; Yang SN; Zhang F; Larsson O; Berggren PO
- Article: REGULATORY PEPTIDES. 2004;119(1-2):61-67Ahmad T; Ugarph-Morawski A; Li H; Bileviciute-Ljungar I; Finn A; Östenson CG; Kreicbergs A
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2004;279(23):24152-24162Thyrell L; Hjortsberg L; Arulampalam V; Panaretakis T; Uhles S; Dagnell M; Zhivotovsky B; Leibiger I; Grandér D; Pokrovskaja K
- Article: JOURNAL OF INTERNAL MEDICINE. 2004;255(6):645-652Agardh EE; Carlsson S; Ahlbom A; Efendic S; Grill V; Hammar N; Hilding A; Östenson CG
- Article: BIOCHEMICAL JOURNAL. 2004;380(Pt 2):465-473Barker CJ; Wright J; Hughes PJ; Kirk CJ; Michell RH
- Article: LAKARTIDNINGEN. 2004;101(20):1776Sundberg CJ; Ostenson C-G; Ståhle A
- Article: DIABETES CARE. 2004;27(3):716-721Explanations of socioeconomic differences in excess risk of type 2 diabetes in Swedish men and womenAgardh EE; Ahlbom A; Andersson T; Efendic S; Grill V; Hallqvist J; Östenson CG
- Article: DIABETOLOGIA. 2004;47(3):537-540Yngen M; Östenson CG; Hu H; Li N; Hjemdahl P; Wallén NH
- Article: DIABETOLOGIA. 2004;47(2):277-283Bränström R; Aspinwall CA; Välimäki S; Östensson CG; Tibell A; Eckhard M; Brandhorst H; Corkey BE; Berggren PO; Larsson O
- Published conference paper: DIABETES. 2004;53:S51-S59Davani B; Portwood N; Bryzgalova G; Reimer MK; Heiden T; Östenson CG; Okret S; Ahren B; Efendic S; Khan A
- Published conference paper: DIABETES. 2004;53:S31-S35Gu HF; Abulaiti A; Östenson CG; Humphreys K; Wahlestedt C; Brookes AJ; Efendic S
- Article: SCANDINAVIAN JOURNAL OF SURGERY. 2004;93(1):34-36Wennström I; Berggren P; Akerud L; Järhult J
- Article: JOURNAL OF THROMBOSIS AND HAEMOSTASIS. 2004;2(1):58-64Hu H; Li N; Yngen M; Östenson CG; Wallén NH; Hjemdahl P
- Article: JOURNAL OF CELL BIOLOGY. 2003;163(6):1327-1337Uhles S; Moede T; Leibiger B; Berggren PO; Leibiger IB
- Article: JNCI-JOURNAL OF THE NATIONAL CANCER INSTITUTE. 2003;95(23):1797-1800Zendehdel K; Nyrén O; Östenson CG; Adami HO; Ekbom A; Ye WM
- Article: DIABETES. 2003;52(12):2943-2950Köhler M; Zaitsev SV; Zaitseva II; Leibiger B; Leibiger IB; Turunen M; Kapelioukh IL; Bakkman L; Appelskog IB; de Monvel JB; Imreh G; Berggren PO
- Article: DIABETES. 2003;52(12):2896-2904Kawamori D; Kajimoto Y; Kaneto H; Umayahara Y; Fujitani Y; Miyatsuka T; Watada H; Leibiger IB; Yamasaki Y; Hori M
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2003;278(47):46210-46218Yu J; Leibiger B; Yang SN; Caffery JJ; Shears SB; Leibiger IB; Barker CJ; Berggren PO
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2003;278(45):44753-44757Mendez CF; Leibiger IB; Leibiger B; Hoy M; Gromada J; Berggren PO; Bertorello AM
- Article: DIABETES CARE. 2003;26(11):2994-2998Terry PD; Weiderpass E; Östenson CG; Cnattingius S
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2003;278(42):40710-40716Juntti-Berggren L; Webb DL; Arkhammar POG; Schultz V; Schweda EKH; Tornheim K; Berggren PO
- Article: DIABETES. 2003;52(10):2526-2531Kamp F; Kizilbash N; Corkey BE; Berggren PO; Hamilton JA
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2003;278(37):35168-35171Hoy M; Berggren PO; Gromada J
- Meeting abstract: EPIDEMIOLOGY. 2003;14(5):S60Bluhm GL; Östenson CG
- Article: EUROPEAN JOURNAL OF PUBLIC HEALTH. 2003;13(3):235-239Andersson CM; Bjärås GEM; Tillgren P; Östenson CG
- Article: BLOOD COAGULATION & FIBRINOLYSIS. 2003;14(6):551-556Antovic JP; Yngen M; Östenson CG; Antovic A; Wallen HN; Jorneskög G; Blombäck M
- Article: DIABETOLOGIA. 2003;46(8):1124-1130Carlsson PO; Berne C; Östenson CG; Andersson A; Jansson L
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2003;285(2):E438-E446Lehtihet M; Welsh N; Berggren PO; Cook GA; Sjöholm Å
- Article: DEVELOPMENTAL CELL. 2003;5(1):73-83Yoon JC; Xu G; Deeney JT; Yang SN; Rhee J; Puigserver P; Levens AR; Yang RJ; Zhang CY; Lowell BB; Berggren PO; Newgard CB; Bonner-Weir S; Weir G; Spiegelman BM
- Article: JOURNAL OF ENDOCRINOLOGY. 2003;178(1):111-116Ahmad T; Ohlsson C; Sääf M; Östenson CG; Kreicbergs A
- Article: NAUNYN-SCHMIEDEBERGS ARCHIVES OF PHARMACOLOGY. 2003;367(5):500-508Daré E; Fetissov S; Hökfelt T; Hall H; Ögren SO; Ceccatelli S
- Article: AMERICAN JOURNAL OF NEPHROLOGY. 2003;23(3):140-151Chen M; Jahnukainen T; Bao WJ; Daré E; Ceccatelli S; Celsi G
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2003;100(9):5187-5192Olsen HL; Hoy M; Zhang W; Bertorello AM; Bokvist K; Capito K; Efanov AM; Meister B; Thams P; Yang SN; Rorsman P; Berggren PO; Gromada J
- Article: DIABETES CARE. 2003;26(3):719-724Agardh EE; Ahlbom A; Andersson T; Efendic S; Grill V; Hallqvist J; Norman A; Östenson CG
- Article: MOLECULAR BIOLOGY OF THE CELL. 2003;14(3):1149-1157Bertorello AM; Komarova Y; Smith K; Leibiger IB; Efendiev R; Pedemonte CH; Borisy G; Sznajder JI
- Article: JOURNAL OF GENERAL PHYSIOLOGY. 2003;121(3):181-197Eliasson L; Ma XS; Renström E; Barg S; Berggren PO; Galvanovskis J; Gromada J; Jing XJ; Lundquist I; Salehi A; Sewing S; Rorsman P
- Article: INVESTIGATIVE RADIOLOGY. 2003;38(3):171-176Ahmad T; Ohlsson C; Östenson CG; Kreicbergs A
- Article: CELLULAR AND MOLECULAR LIFE SCIENCES. 2003;60(2):378-381Norberg Å; Gruber S; Angelucci F; Renlund S; Wadensten H; Efendic S; Östenson CG; Jörnvall H; Sillard R; Mathé AA
- Article: PERITONEAL DIALYSIS INTERNATIONAL. 2003;23(1):5-13CAPD peritonitis induces the production of a novel peptide, daintain/allograft inflammatory factor-1Brauner A; Hertting O; Alkstrand E; Sandberg E; Chromek M; Chen ZW; Östenson CG
- Review: FEBS LETTERS. 2002;532(1-2):1-6Leibiger IB; Leibiger B; Berggren PO
- Published conference paper: DIABETES. 2002;51:S448-S454Efendic S; Efanov AM; Berggren PO; Zaitsev SV
- Article: JOURNAL OF ENDOCRINOLOGY. 2002;175(3):637-647Guest PC; Abdel-Halim SM; Gross DI; Clark A; Poitout V; Amaria R; Ostenson CG; Hutton JC
- Article: EUROPEAN JOURNAL OF ENDOCRINOLOGY. 2002;147(6):825-833Strömmer L; Wickbom M; Wang F; Herrington MK; Östenson CG; Arnelo U; Permert J
- Article: ENDOCRINOLOGY. 2002;143(12):4592-4598Sjöholm Å; Lehtihet M; Efanov AM; Zaitsev SV; Berggren PO; Honkanen RE
- Review: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2002;283(6):E1113-E1122Barker CJ; Leibiger IB; Leibiger B; Berggren PO
- Article: HEALTH PROMOTION INTERNATIONAL. 2002;17(4):317-327Andersson CM; Bjärås GEM; Östenson CG
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2002;277(46):44108-44114Efendiev R; Yudowski GA; Zwiller J; Leibiger B; Katz AI; Berggren PO; Pedemonte CH; Leibiger IB; Bertorello AM
- Article: FEBS LETTERS. 2002;531(2):199-203Hoy M; Maechler P; Efanov AM; Wollheim CB; Berggren PO; Gromada J
- Article: DIABETES-METABOLISM RESEARCH AND REVIEWS. 2002;18(6):473-483Murakawa Y; Zhang WX; Pierson CR; Brismar T; Östenson CG; Efendic S; Sima AAF
- Article: EUROPEAN JOURNAL OF NEUROSCIENCE. 2002;16(10):2013-2016Fonfría E; Daré E; Benelli M; Suñol C; Ceccatelli S
- Article: ENDOCRINOLOGY. 2002;143(11):4218-4226Gaisano HY; Ostenson CG; Sheu L; Wheeler MB; Efendic S
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2002;277(40):37176-37183Shiota C; Larsson O; Shelton KD; Shiota M; Efanov AM; Hoy M; Lindner J; Kooptiwut S; Juntti-Berggren L; Gromada J; Berggren PO; Magnuson MA
- Article: CELLULAR AND MOLECULAR LIFE SCIENCES. 2002;59(7):1185-1189Shafqat J; Juntti-Berggren L; Zhong Z; Ekberg K; Köhler M; Berggren PO; Johansson J; Wahren J; Jörnvall H
- Article: BRAIN PATHOLOGY. 2002;12(3):287-299Storch MK; Weissert R; Stefferl A; Birnbacher R; Wallström E; Dahlman I; Ostensson CG; Linington C; Olsson T; Lassmann H
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2002;99(10):6773-6777Hoy M; Efanov AM; Bertorello AM; Zaitsev SV; Olsen HL; Bokvist K; Leibiger B; Leibiger IB; Zwiller J; Berggren PO; Gromada J
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2002;277(19):17108-17111Doné SC; Leibiger IB; Efendiev R; Katz AI; Leibiger B; Berggren PO; Pedemonte CH; Bertorello AM
- Article: JOURNAL OF NEURAL TRANSMISSION. 2002;109(5-6):597-605Götz ME; Koutsilieri E; Riederer P; Ceccatelli S; Daré E
- Article: DIABETES. 2002;51(5):1425-1436Ji JZ; Yang SN; Huang XH; Li XD; Shen L; Diamant N; Berggren PO; Gaisano HY
- Article: AMERICAN JOURNAL OF EPIDEMIOLOGY. 2002;155(9):834-841Grill V; Dinesen B; Carlsson S; Efendic S; Pedersen O; Östenson CG
- Article: BRAIN RESEARCH. 2002;933(1):12-22Daré E; Tofighi R; Vettori MV; Momoi T; Poli D; Saido TC; Mutti A; Ceccatelli S
- Doctoral thesis: 2002Daré E
- Published conference paper: BIOCHEMICAL SOCIETY TRANSACTIONS. 2002;30(2):312-317Leibiger B; Moede T; Uhles S; Berggren PO; Leibiger IB
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2002;291(4):1038-1044Zhang W; Khan A; Östenson CG; Berggren PO; Efendic S; Meister B
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2002;291(4):945-950Östensen CG; Sandberg-Nordqvist AC; Chen J; Hällbrink M; Rotin D; Langel Ü; Efendic S
- Article: MAMMALIAN GENOME. 2002;13(1):20-23Li FY; Leibiger B; Leibiger I; Larsson C
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2002;282(1):E117-E124Efanov AM; Appelskog IB; Abdel-Halim SM; Khan A; Bränström R; Larsson O; Östenson CG; Mest HJ; Berggren PO; Efendic S; Zaitsev SV
- Article: HUMAN MOLECULAR GENETICS. 2001;10(23):2637-2644Liu L; Doné SC; Khoshnoodi J; Bertorello A; Wartiovaara J; Berggren PO; Tryggvason K
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2001;276(40):36946-36950Deeney JT; Köhler M; Kubik K; Brown G; Schultz V; Tornheim K; Corkey BE; Berggren PO
- Article: NEUROTOXICITY RESEARCH. 2001;3(5):501-514Dare E; Gorman AM; Ahlbom E; Gotz M; Momoi T; Ceccatelli S
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2001;276(37):34530-34536Kindmark H; Köhler M; Brown G; Bränström R; Larsson O; Berggren PO
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2001;276(36):34199-34205Lilja L; Yang SN; Webb DL; Juntti-Berggren L; Berggren PO; Bark C
- Article: NEUROREPORT. 2001;12(12):2737-2741Johansen JE; Teixeira VL; Johansson C; Serrao P; Berggren PO; Soares-Da-Silva P; Schalling M; Bertorello AM
- Article: FASEB JOURNAL. 2001;15(10):1753-1763Yang SN; Yu J; Mayr GW; Hofmann F; Larsson O; Berggren PO
- Article: HEARING RESEARCH. 2001;157(1-2):43-51Ågrup C; Berggren PO; Bagger-Sjöbäck D
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2001;284(4):918-922Efanov AM; Hoy M; Bränström R; Zaitsev SV; Magnuson MA; Efendic S; Gromada J; Berggren PO
- Article: FREE RADICAL BIOLOGY AND MEDICINE. 2001;30(12):1347-1356Daré E; Li W; Zhivotovsky B; Yuan XM; Ceccatelli S
- Article: DIABETES. 2001;50(6):1449-1454Båvenholm PN; Pigon J; Östenson CG; Efendic S
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2001;98(11):6319-6324Glavas NA; Ostenson C; Schaefer JB; Vasta V; Beavo JA
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2001;283(2):364-368Sjöholm Å; Berggren PO; Honkanen RE
- Article: JOURNAL OF ANTIMICROBIAL CHEMOTHERAPY. 2001;47(5):575-580Linde CMA; Hoffner SE; Refai E; Andersson M
- Article: JOURNAL OF PHYSIOLOGY-LONDON. 2001;532(3):649-659Chen XL; Zhong ZG; Yokoyama S; Bark C; Meister B; Berggren PO; Roder J; Higashida H; Jeromin A
- Article: FEBS LETTERS. 2001;494(1-2):79-84Li FY; Nikali K; Gregan J; Leibiger I; Leibiger B; Schweyen R; Larsson C; Suomalainen A
- Article: NUTRITION. 2001;17(4):332-336Effect of carbohydrate feeding on insulin action in skeletal muscle after surgical trauma in the ratStrömmer L; Isaksson B; Wickbom M; Arnelo U; Östenson CG; Herrington M; Permert J
- Article: DIABETES. 2001;50(4):797-802Efanov AM; Zaitsev SV; Mest HJ; Raap A; Appelskog IB; Larsson O; Berggren PO; Efendic S
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2001;276(13):9971-9977Lemmens R; Larsson O; Berggren PO; Islam MS
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2001;281(5):1070-1073Efanov AM; Zaitsev SV; Berggren PO; Mest HJ; Efendic S
- Article: BIOCHIMICA ET BIOPHYSICA ACTA: INTERNATIONAL JOURNAL OF BIOCHEMISTRY AND BIOPHYSICS. 2001;1546(1):79-86Hällbrink M; Holmqvist T; Olsson M; Östenson CG; Efendic S; Langel Ü
- Published conference paper: ACTA PHYSIOLOGICA. 2001;171(3):241-247Östenson CG
- Article: MOLECULAR CELL. 2001;7(3):559-570Leibiger B; Leibiger IB; Moede T; Kemper S; Kulkarni RN; Kahn CR; de Vargas LM; Berggren PO
- Article: BLOOD COAGULATION & FIBRINOLYSIS. 2001;12(2):109-116Yngen M; Östenson CG; Li N; Hjemdahl P; Wallén NH
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2001;280(4):1161-1168Gogg S; Chen J; Efendic S; Smith U; Östenson CG
- Published conference paper: DIABETES. 2001;50:S15-S19Moede T; Leibiger B; Berggren PO; Leibiger IB
- Published conference paper: DIABETES. 2001;50:S77-S81Chandra J; Yang SN; Köhler M; Zaitsev S; Juntti-Berggren L; Berggren PO; Zhivotovsky B; Orrenius S
- Published conference paper: DIABETES. 2001;50:S70-S76Zaitsev SV; Appelskog IB; Kapelioukh IL; Yang SN; Köhler M; Efendic S; Berggren PO
- Published conference paper: DIABETES. 2001;50:S44-S47Chandra J; Zhivotovsky B; Zaitsev S; Juntti-Berggren L; Berggren PO; Orrenius S
- Published conference paper: DIABETES. 2001;50:S108-S112Ling ZC; Hong-Lie C; Östenson CG; Efendic S; Khan A
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-CELL PHYSIOLOGY. 2001;280(2):C317-C323Sjöholm Å; Arkhammar P; Berggren PO; Andersson A
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2001;276(2):924-930Hoy M; Bokvist K; Weng XG; Hansen J; Juhl K; Berggren PO; Buschard K; Gromada J
- Article: ENVIRONMENTAL TOXICOLOGY AND PHARMACOLOGY. 2001;9(3):61-70Giménez-Llort L; Ahlbom E; Daré E; Vahter M; Ögren SO; Ceccatelli S
- Article: PATIENT EDUCATION AND COUNSELING. 2001;42(1):9-14Bjärås G; Härberg LK; Sydhoff J; Östenson CG
- Article: INTERNATIONAL JOURNAL OF INFECTIOUS DISEASES. 2001;5(1):9-18Gan X; Jarstrand C; Herting E; Berggren P; Robertson B
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2000;275(52):41521-41527Zhang W; Efanov A; Yang SN; Fried G; Kölare S; Brown H; Zaitsev S; Berggren PO; Meister B
- Article: JOURNAL OF NEUROSCIENCE RESEARCH. 2000;62(4):557-565Daré E; Götz ME; Zhivotovsky B; Manzo L; Ceccatelli S
- Article: DIABETIC MEDICINE. 2000;17(11):776-781Carlsson S; Hammar N; Efendic S; Persson PG; Östenson CG; Grill V
- Article: JOURNAL OF INTERNAL MEDICINE. 2000;248(5):387-396Henriksson F; Agardh CD; Berne C; Bolinder J; Lönnqvist F; Stenström P; Östenson CG; Jönsson B
- Article: NATURE GENETICS. 2000;26(3):336-340Silva JP; Köhler M; Graff C; Oldfors A; Magnuson MA; Berggren PO; Larsson NG
- Article: ATHEROSCLEROSIS. 2000;153(1):23-35Ares MPS; Pörn-Ares MI; Moses S; Thyberg J; Juntti-Berggren L; Berggren PO; Hultgårdh-Nilsson A; Kallin B; Nilsson J
- Article: AMERICAN JOURNAL OF PHYSIOLOGY-ENDOCRINOLOGY AND METABOLISM. 2000;279(5):E1139-E1144Svensson AM; Östenson CG; Jansson L
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 2000;277(2):334-340Sjöholm Å; Berggren PO; Cooney RV
- Journal article: BIOCHEMICAL SOCIETY TRANSACTIONS. 2000;28(5):a273Barker CJ; Yu J; Leibiger B; Leibiger I; Caffrey JJ; Shears SB; Berggren PO
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2000;275(39):30153-30156Leibiger B; Wåhlander K; Berggren PO; Leibiger IB
- Article: DIABETES. 2000;49(9):1409-1412Larsson O; Barker CJ; Berggren PO
- Article: JOURNAL OF INTERNAL MEDICINE. 2000;248(2):103-110Persson PG; Carlsson S; Svanström L; Östenson CG; Efendic S; Grill V
- Article: SCANDINAVIAN JOURNAL OF IMMUNOLOGY. 2000;52(2):117-122Pashenkov M; Efendic S; Zhu J; Zou LP; Östenson CG; Mustafa M
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2000;275(28):21033-21040Sjöholm Å; Zhang QM; Welsh N; Hansson A; Larsson O; Tally M; Berggren PO
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2000;97(12):6556-6561Yudowski GA; Efendiev R; Pedemonte CH; Katz AI; Berggren PO; Bertorello AM
- Article: ENDOCRINE. 2000;12(3):273-278Sjöholm Å; Sandberg E; Östenson CG; Efendic S
- Article: METABOLISM-CLINICAL AND EXPERIMENTAL. 2000;49(4):486-491Preoperative feeding does not reverse postoperative insulin resistance in skeletal muscle in the ratStrömmer L; Isaksson B; Arnelo U; Lundkvist I; Östenson CG; Wickbom M; Herrington M; Permert J
- Article: PANCREAS. 2000;20(3):282-289Sjöholm A; Sandberg E; Östenson CG; Efendic S
- Article: PANCREAS. 2000;20(3):264-269Wang F; Permert J; Östenson CG
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 2000;275(13):9363-9368Deeney JT; Gromada J; Hoy M; Olsen HL; Rhodes CJ; Prentki M; Berggren PO; Corkey BE
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 2000;97(7):3242-3247Ogimoto G; Yudowski GA; Barker CJ; Köhler M; Katz AI; Féraille E; Pedemonte CH; Berggren PO; Bertorello AM
- Article: DIABETES. 2000;49(3):383-391Brown H; Meister B; Deeney J; Corkey BE; Yang SN; Larsson O; Rhodes CJ; Seino S; Berggren PO; Fried G
- Article: LAKARTIDNINGEN. 2000;97(9):981-982Hjalmarson A; Rössner S; Ostenson CG
- Article: PATIENT EDUCATION AND COUNSELING. 2000;39(2-3):281-284Hjalmarson A; Rössner S; Östenson CG
- Article: DIABETES, NUTRITION AND METABOLISM - CLINICAL AND EXPERIMENTAL. 2000;13(1):7-12Juntti-Berggren L; Pigon J; Hellström P; Holst JJ; Efendic S
- Article: DIABETES. 2000;49(1):59-64Zhuang H; Bhattacharjee A; Hu FQ; Zhang M; Goswami T; Wang L; Wu SW; Berggren PO; Li M
- Article: FEBS LETTERS. 1999;461(3):229-234Moede T; Leibiger B; Pour HG; Berggren PO; Leibiger IB
- Article: NUCLEAR MEDICINE AND BIOLOGY. 1999;26(8):931-936Refai E; Jonsson C; Andersson M; Jacobsson H; Larsson S; Kogner P; Hassan M
- Published conference paper: PEDIATRIC RESEARCH. 1999;46(3):328-332Borgström P; Hassan M; Wassberg E; Refai E; Jonsson C; Larsson SA; Jacobsson H; Kogner P
- Published conference paper: ANTICANCER RESEARCH. 1999;19(5A):3737-3741Barker CJ; Berggren PO
- Letter: SCANDINAVIAN JOURNAL OF PUBLIC HEALTH. 1999;27(3):237-238Bjärås G; Härberg LK; Östenson CG
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 1999;96(18):10164-10169Yang SN; Larsson O; Bränström R; Bertorello AM; Leibiger B; Leibiger IB; Moede T; Köhler M; Meister B; Berggren PO
- Article: DIABETOLOGIA. 1999;42(Suppl 1):a1-a330Melander A; Olsson J; Lindberg G
- Article: DIABETES. 1999;48(8):1557-1561Ristow M; Carlqvist H; Hebinck J; Vorgerd M; Krone W; Pfeiffer A; Müller-Wieland D; Östenson CG
- Article: DIABETES CARE. 1999;22(7):1043-1047Carlsson S; Persson PG; Alvarsson M; Efendic S; Norman A; Svanström L; Östenson CG; Grill V
- Article: ENDOCRINOLOGY. 1999;140(7):3045-3053Salehi A; Henningsson R; Mosén H; Östenson CG; Efendic S; Lundquist I
- Article: REGULATORY PEPTIDES. 1999;82(1-3):45-51Hällbrink M; Saar K; Östenson CG; Soomets U; Efendic S; Howl J; Wheatley M; Zorko M; Langel Ü
- Published conference paper: ANNALS OF THE NEW YORK ACADEMY OF SCIENCES. 1999;881:241-252Zaitsev SV; Efanov AM; Raap A; Efanova IB; Schloos J; Steckel-Hamann B; Larsson O; Östenson CG; Berggren PO; Mest HJ; Efendic S
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 1999;96(10):5539-5544Barg S; Renström E; Berggren PO; Bertorello A; Bokvist K; Braun M; Eliasson L; Holmes WE; Köhler M; Rorsman P; Thévenod F
- Article: NUCLEAR MEDICINE AND BIOLOGY. 1999;26(4):413-420Hassan M; Eskilsson A; Nilsson C; Jonsson C; Jacobsson H; Refai E; Larsson S; Efendic S
- Article: ENDOCRINOLOGY. 1999;140(3):1200-1204Wang L; Bhattacharjee A; Zuo Z; Hu FQ; Honkanen RE; Berggren PO; Li M
- Review: LAKARTIDNINGEN. 1999;96(8):888-892Efendic S; Bystedt T; Ostenson CG
- Article: JOURNAL OF NEUROPHYSIOLOGY. 1999;81(2):781-787Yang SN; Tang YG; Zucker RS
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 1999;274(4):2000-2008Owada S; Larsson O; Arkhammar P; Katz AI; Chibalin AV; Berggren PO; Bertorello AM
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 1999;274(4):1920-1927Chibalin AV; Ogimoto G; Pedemonte CH; Pressley TA; Katz AI; Féraille E; Berggren PO; Bertorello AM
- Article: DIABETOLOGIA. 1999;42(1):15-23Grill V; Persson G; Carlsson S; Norman A; Alvarsson M; Östensson CG; Svanström L; Efendic S
- Article: WOUND REPAIR AND REGENERATION. 1999;7(1):65-71Hehenberger K; Hansson A; Heilborn JD; Abdel-Halim SM; Östensson CG; Brismar K
- Article: FEBS LETTERS. 1998;441(1):97-102Köhler M; Norgren S; Berggren PO; Fredholm BB; Larsson O; Rhodes CJ; Herbert TP; Luthman H
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 1998;273(50):33501-33507Efanova IB; Zaitsev SV; Zhivotovsky B; Köhler M; Efendic S; Orrenius S; Berggren PO
- Article: FEBS LETTERS. 1998;439(3):267-270Zaitsev SV; Andersson M; Efanov AM; Efanova IB; Östenson CG; Juntti-Berggren L; Berggren PO; Mutt V; Efendic S
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 1998;273(47):31395-31400Bränström R; Leibiger IB; Leibiger B; Corkey BE; Berggren PO; Larsson O
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 1998;252(1):162-165Efanova IB; Zaitsev SV; Efanov AM; Östenson CG; Raap A; Mest HJ; Berggren PO; Efendic S
- Article: DIABETOLOGIA. 1998;41(11):1368-1373Guenifi A; Abdel-Halim SM; Efendic S; Östenson CG
- Article: AMERICAN JOURNAL OF EPIDEMIOLOGY. 1998;148(6):539-545Carlsson S; Persson PG; Alvarsson M; Efendic S; Norman A; Svanström L; Östenson CG; Grill V
- Article: EMBO JOURNAL. 1998;17(17):5048-5058Brown H; Larsson O; Bränström R; Yang SN; Leibiger B; Leibiger I; Fried G; Moede T; Deeney JT; Brown GR; Jacobsson G; Rhodes CJ; Braun JEA; Scheller RH; Corkey BE; Berggren PO; Meister B
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 1998;273(34):21777-21782Källström H; Islam MS; Berggren PO; Jonsson AB
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 1998;95(16):9307-9312Leibiger B; Moede T; Schwarz T; Brown GR; Köhler M; Leibiger IB; Berggren PO
- Article: JOURNAL OF MOLECULAR ENDOCRINOLOGY. 1998;21(1):7-17Darè E; Kifor O; Brown EM; Weber G
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 1998;273(23):14113-14118Bränström R; Efendic S; Berggren PO; Larsson O
- Article: ENDOCRINOLOGY. 1998;139(6):2670-2675Ling ZC; Efendic S; Wibom R; Abdel-Halim SM; Östenson CG; Landau BR; Khan A
- Article: DIABETOLOGIA. 1998;41(6):634-639Ling ZC; Khan A; Delauny F; Davani B; Östenson CG; Gustafsson J; Okret S; Landau BR; Efendic S
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 1998;95(11):6145-6150Islam MS; Leibiger I; Leibiger B; Rossi D; Sorrentino V; Ekström TJ; Westerblad H; Andrade FH; Berggren PO
- Article: MOLECULAR CELL. 1998;1(6):933-938Leibiger IB; Leibiger B; Moede T; Berggren PO
- Article: MOLECULAR BIOLOGY OF THE CELL. 1998;9(5):1209-1220Chibalin AV; Zierath JR; Katz AI; Berggren PO; Bertorello AM
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 1998;273(15):8814-8819Chibalin AV; Pedemonte CH; Katz AI; Féraille E; Berggren PO; Bertorello AM
- Article: AMERICAN JOURNAL OF PHYSIOLOGY. 1998;274(4):e751-e757Efanov AM; Zaitsev SV; Efanova IB; Zhu SS; Östenson CG; Berggren PO; Efendic S
- Article: MAMMALIAN GENOME. 1998;9(4):310-311Betz R; Leibiger B; Farnebo F; Lagercrantz S; Piehl F; Leibiger I; Larsson C
- Article: JOURNAL OF ENDOCRINOLOGY. 1998;156(3):573-581Zhang Q; Berggren PO; Hansson A; Tally M
- Article: CURRENT EYE RESEARCH. 1998;17(3):251-256Agardh CD; Agardh E; Hultberg B; Qian YN; Östenson CG
- Article: SCANDINAVIAN JOURNAL OF PUBLIC HEALTH. 1998;26(1):75-80Persson PG; Carlsson S; Grill V; Hagman U; Lundgren AC; Ostenson CG; Perers M; Wallen A
- Article: PANCREAS. 1998;16(2):134-140Wróblewski R; Pålsgård E; Juntti-Berggren L; Berggren PO; Roomans GM
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 1998;243(1):117-121Zhang QM; Berggren PO; Larsson O; Hall K; Tally M
- Article: DIABETES. 1998;47(2):211-218Efanova IB; Zaitsev SV; Brown G; Berggren PO; Efendic S
- Article: DIABETES. 1998;47(1):1-4Zierath JR; Frevert EU; Ryder JW; Berggren PO; Kahn BB
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 1997;94(25):13879-13884Chen ZW; Ahren B; Ostenson CG; Cintra A; Bergman T; Moller C; Fuxe K; Mutt V; Jornvall H; Efendic S
- Journal article: LAKARTIDNINGEN. 1997;94(48):4473-4477Larsson O; Kindmark H; Bränström R; Berggren PO
- Article: DIABETOLOGIA. 1997;40(12):1480-1484Islam MS; Berggren PO
- Article: AMERICAN JOURNAL OF PHYSIOLOGY. 1997;273(5):c1458-c1465Chibalin AV; Katz AI; Berggren PO; Bertorello AM
- Article: JOURNAL OF DIABETES AND ITS COMPLICATIONS. 1997;11(6):319-322Ostenson CG; Fiere V; Ahmed M; Lindstrom P; Brismar K; Brismar T; Kreicbergs A
- Article: NEUROSCIENCE. 1997;81(2):503-515Yang SN; Lippoldt A; Jansson A; Phillips MI; Ganten D; Fuxe K
- Article: SCIENCE. 1997;278(5337):471-474Larsson O; Barker CJ; Sjoholm A; Carlqvist H; Michell RH; Bertorello A; Nilsson T; Honkanen RE; Mayr GW; Zwiller J; Berggren PO
- Article: FEBS JOURNAL. 1997;249(2):518-522Chen ZW; Bergman T; Ostenson CG; Efendic S; Mutt V; Jornvall H
- Article: JOURNAL OF CLINICAL INVESTIGATION. 1997;100(8):2094-2098Delaunay F; Khan A; Cintra A; Davani B; Ling ZC; Andersson A; Ostenson CG; Gustafsson JA; Efendic S; Okret S
- Article: BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS. 1997;239(1):129-133Zaitsev S; Efanova I; Ostenson CG; Efendic S; Berggren PO
- Published conference paper: EUROPEAN JOURNAL OF CANCER. 1997;33(12):2084-2089Kogner P; Borgström P; Bjellerup P; Schilling FH; Refai E; Jonsson C; Dominici C; Wassberg E; Bihl H; Jacobsson H; Theodorsson E; Hassan M
- Article: JOURNAL OF LIPID RESEARCH. 1997;38(10):2049-2061Ares MPS; PornAres MI; Thyberg J; JunttiBerggren L; Berggren PO; Diczfalusy U; Kallin B; Bjorkhem I; Orrenius S; Nilsson J
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 1997;272(38):23703-23706Zhang QM; Berggren PO; Tally M
- Article: BIOCHEMICAL JOURNAL. 1997;325(Pt 3):771-778Brown GR; Kohler M; Berggren PO
- Article: ENDOCRINOLOGY. 1997;138(8):3308-3313Ostenson CG; Zaitsev S; Berggren PO; Efendic S; Langel U; Bartfai T
- Article: FEBS LETTERS. 1997;411(2-3):301-307Branstrom R; Hoog A; Wahl MA; Berggren PO; Larsson O
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 1997;272(28):17390-17394Branstrom R; Corkey BE; Berggren PO; Larsson O
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 1997;94(12):6232-6237Zhang QM; Tally M; Larsson O; Kennedy RT; Huang L; Hall K; Berggren PO
- Article: HEALTH PROMOTION INTERNATIONAL. 1997;12(2):151-160Bjaras G; Ahlbom A; Alvarsson M; Burstrom B; Diderichsen F; Efendic S; Grill V; Haglund BJ; Norman A; Persson PG; Svanstrom L; Tillgren P; Ullen H; Ostenson CG
- Article: DIABETES. 1997;46(6):947-952Carlsson PO; Jansson L; Ostenson CG; Kallskog O
- Article: NEUROSCIENCE. 1997;78(3):803-813Yang SN; Fior DR; Hansson AC; Cintra A; Castellano M; Ganten U; Ganten D; Agnati LF; Fuxe K
- Article: JOURNAL OF DIABETES AND ITS COMPLICATIONS. 1997;11(3):158-162Agardh CD; Agardh E; Zhang H; Ostenson CG
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 1997;94(9):4435-4439Efanov AM; Zaitsev SV; Berggren PO
- Article: REGULATORY PEPTIDES. 1997;69(2):63-68Chen ZW; Bergman T; Jornvall H; Bonetto V; Norberg A; Mutt V; Longone P; Costa E; Efendic S; Ostenson CG
- Letter: PANCREAS. 1997;14(2):212-214Bjorklund A; Ostenson CG; Grill VE
- Article: BIOCHEMICAL JOURNAL. 1997;321(Pt 2):347-354Islam MS; Kindmark H; Larsson O; Berggren PO
- Article: PEPTIDES. 1997;18(7):1055-1059Svensson AM; Efendic S; Ostenson CG; Jansson L
- Article: HEARING RESEARCH. 1996;102(1-2):155-166Agrup C; Berggren PO; Kohler M; Spangberg ML; BaggerSjoback D
- Article: NEUROREPORT. 1996;7(15-17):2701-2705Yang SN; Finnman UB; Fuxe K
- Article: DIABETES CARE. 1996;19(11):1200-1206JunttiBerggren L; Pigon J; Karpe F; Hamsten A; Gutniak M; Vignati L; Efendic S
- Article: DIABETES. 1996;45(11):1610-1618Zaitsev SV; Efanov AM; Efanova IB; Larsson O; Ostenson CG; Gold G; Berggren PO; Efendic S
- Article: BIOCHEMICAL JOURNAL. 1996;319(Pt 2):521-527Varadi A; Molnar E; Ostenson CG; Ashcroft SJH
- Article: PANCREAS. 1996;13(3):259-267Leckstrom A; Ostenson CG; Efendic S; Arnelo U; Permert J; Lundquist I; Westermark P
- Article: DIABETOLOGIA. 1996;39(10):1165-1172Damert A; Leibiger B; Leibiger IB
- Article: JOURNAL OF CLINICAL ENDOCRINOLOGY & METABOLISM. 1996;81(10):3702-3708Pigon J; Giacca A; Ostenson CG; Lam L; Vranic M; Efendic S
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 1996;271(32):19074-19079Webb DL; Islam MS; Efanov AM; Brown G; Kohler M; Larsson O; Berggren PO
- Article: DIABETES CARE. 1996;19(8):857-863Gutniak MK; Juntti-Berggren L; Hellström PM; Guenifi A; Holst JJ; Efendic S
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 1996;271(30):18154-18160Deeney JT; Cunningham BA; Chheda S; Bokvist K; JunttiBerggren L; Lam K; Korchak HM; Corkey BE; Berggren PO
- Article: REGULATORY PEPTIDES. 1996;63(2-3):123-128Carlsson PO; Ostenson CG; Efendic S; Langel U; Jansson L
- Article: NEUROSCIENCE. 1996;73(2):519-530Yang SN; Bunnemann B; Cintra A; Fuxe K
- Article: DIABETES. 1996;45(7):934-940AbdelHalim SM; Guenifi A; Khan A; Larsson O; Berggren PO; Ostenson CG; Efendic S
- Article: DIABETES. 1996;45(7):886-890MacDonald MJ; Efendic S; Ostenson CG
- Article: HEALTH PROMOTION INTERNATIONAL. 1996;11(2):143-156Sanderson C; Haglund BJA; Tillgren P; Svanstrom L; Ostenson CG; Holm LE; Ullen H; Smajkic A
- Article: ENDOCRINOLOGY. 1996;137(6):2415-2423Hoog A; SandbergNordqvist AC; AbdelHalim SM; CarlssonSkwirut C; Guenifi A; Tally M; Ostenson CG; Falkmer S; Sara VR; Efendic S; Schalling M; Grimelius L
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 1996;93(10):5161-5165Larsson O; Kindmark H; Branstrom R; Fredholm B; Berggren PO
- Article: JOURNAL OF BIOLOGICAL CHEMISTRY. 1996;271(18):10623-10626Larsson O; Deeney JT; Branstrom R; Berggren PO; Corkey BE
- Article: SOMATIC CELL AND MOLECULAR GENETICS. 1996;22(3):201-210Dare E; Zhang LH; Jenssen D
- Article: DIABETES. 1996;45(5):580-586Zhou YP; Berggren PO; Grill V
- Article: JOURNAL OF ENDOCRINOLOGY. 1996;149(1):145-154Josefsen K; Stenvang JP; Kindmark H; Berggren PO; Horn T; Kjaer T; Buschard K
- Article: CARBOHYDRATE RESEARCH. 1996;282(1):81-99Spiers ID; Barker CJ; Chung SK; Chang YT; Freeman S; Gardiner JM; Hirst PH; Lambert PA; Michell RH; Poyner DR; Schwalbe CH; Smith AW; Solomons KR
- Article: BIOCHEMICAL JOURNAL. 1996;314(Pt 1):91-94Nilsson T; Schultz V; Berggren PO; Corkey BE; Tornheim K
- Article: SCIENCE. 1996;271(5250):813-815Eliasson L; Renstrom E; Ammala C; Berggren PO; Bertorello AM; Bokvist K; Chibalin A; Deeney JT; Flatt PR; Gabel J; Gromada J; Larsson O; Lindstrom P; Rhodes CJ; Rorsman P
- Article: DIABETES & METABOLISM. 1996;22(1):31-36Moberg E; Kollind M; Ostenson CG; Lins PE; Adamson U
- Article: NATURE GENETICS. 1996;12(1):31-37Galli J; Li LS; Glaser A; Ostenson CG; Jiao H; FakhraiRad H; Jacob HJ; Lander ES; Luthman H
- Article: DIABETES. 1995;44(11):1280-1284ABDELHALIM SM; OSTENSON CG; ANDERSSON A; JANSSON L; EFENDIC S
- Article: PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES OF THE UNITED STATES OF AMERICA. 1995;92(21):9712-9716ZAITSEV SV; EFENDIC S; ARKHAMMAR P; BERTORELLO AM; BERGGREN PO
- Other: JOURNAL OF MOLECULAR BIOLOGY. 1995;252(5):514-521Darè E; Zhang LH; Jenssen D; Bianchi V
- Article: NEUROSCIENCE. 1995;68(3):729-736YANG SN; DASGUPTA S; LLEDO PM; VINCENT JD; FUXE K
- Article: FREE RADICAL BIOLOGY AND MEDICINE. 1995;19(3):259-269COONEY RV; HARWOOD PJ; FRANKE AA; NARALA I; SUNDSTROM AK; BERGGREN PO; MORDAN LJ
- Article: FEBS LETTERS. 1995;369(2-3):315-320KINDMARK H; KOHLER M; LARSSON O; KHAN A; BERGGREN PO
- Article: ENDOCRINOLOGY. 1995;136(8):3546-3551ZHOU YP; OSTENSON CG; LING ZC; GRILL V
- Article: ENDOCRINOLOGY. 1995;136(8):3391-3397SJOHOLM A; HONKANEN RE; BERGGREN PO
- Article: JOURNAL OF INFECTION. 1995;31(1):27-31BRAUNER A; KATOULI M; OSTENSON CG
- Article: AMERICAN JOURNAL OF PHYSIOLOGY. 1995;268(6):E1046-E1050PIGON J; EFENDIC S; OSTENSON CG; LAM L; VRANIC M; GIACCA A
- Article: MOLECULAR AND CELLULAR ENDOCRINOLOGY. 1995;111(2):159-165EIZIRIK DL; SANDLER S; WELSH N; JUNTTIBERGGREN L; BERGGREN PO
- Article: BRAIN RESEARCH. 1995;679(1):168-177FIOR DR; YANG SN; GANTEN U; GANTEN D; FUXE K
- Article: BIOCHEMICAL SOCIETY TRANSACTIONS. 1995;23(2):169SBarker CJ; Wright J; Kirk CJ; Michell RH
- Article: GENOMICS. 1995;26(3):467-472LAGERCRANTZ J; CARSON E; PHELAN C; GRIMMOND S; ROSEN A; DARE E; NORDENSKJOLD M; HAYWARD NK; LARSSON C; WEBER G
- Published conference paper: ZOOLOGICAL STUDIES. 1995;34:191-194KOHLER M; NORGREN S; FREDHOLM BB; LARSSON O; LUTHMAN H; BERGGREN PO
- Article: BIOCHEMICAL JOURNAL. 1995;306(Pt 3):679-686ISLAM MS; LARSSON O; NILSSON T; BERGGREN PO
- Article: BIOCHEMICAL JOURNAL. 1995;306(Pt 2):557-564BARKER CJ; FRENCH PJ; MOORE AJ; NILSSON T; BERGGREN PO; BUNCE CM; KIRK CJ; MICHELL RH
- Article: CLINICAL SCIENCE. 1995;88(3):301-306NOLTE LA; ABDELHALIM SM; MARTIN IK; GUENIFI A; ZIERATH JR; OSTENSON CG; WALLBERGHENRIKSSON H
- Article: PANCREAS. 1995;10(2):148-153GUENIFI A; ABDELHALIM SM; HOOG A; FALKMER S; OSTENSON CG
- Article: NEUROREPORT. 1995;6(2):384-388YANG SN; BJELKE B; NARVAEZ JA; CINTRA A; AGNATI LF; FUXE K
- Article: CELL BIOLOGY INTERNATIONAL. 1995;19(1):25-34PALSGARD E; LINDH U; JUNTTIBERGGREN L; BERGGREN PO; ROOMANS GM
- Journal article: EUROPEAN JOURNAL OF PHARMACOLOGY. 1994;271(1):201-212YANG SN; FIOR DR; HEDLUND PB; AGNATI LF; FUXE K
- Journal article: EUROPEAN JOURNAL OF PHARMACOLOGY. 1994;262(3):271-282FIOR DR; YANG SN; HEDLUND PB; NARVAEZ JA; AGNATI LF; FUXE K
- Journal article: BRAIN RESEARCH. 1994;654(1):137-144YANG SN; FIOR DR; HEDLUND PB; AGNATI LF; FUXE K
- Journal article: NEUROSCIENCE LETTERS. 1994;171(1-2):27-31YANG SN; FIOR DR; HEDLUND PB; NARVAEZ JA; AGNATI LF; FUXE K
- Journal article: BIOCHEMICAL JOURNAL. 1994;297(Pt 2):265-268BARKER CJ; NILSSON T; KIRK CJ; MICHELL RH; BERGGREN PO
- Journal article: BRAIN RESEARCH. 1993;621(1):126-132YANG SN; NARVAEZ JA; BJELKE B; AGNATI LF; FUXE K
- Show more
Staff and contact
Group leader
- Ismael Valladolid AcebesResearch Specialist
All members of the group
- Christopher BarkerSenior Research Specialist | Docent
- Per Olof BerggrenProfessor, Senior
- Lisa BerggrenProfessor, Senior
- Galyna BryzgalovaResearch Specialist
- Elisabetta DaréSenior Research Specialist | Docent
- Nadir KadriSenior Research Specialist
- Elisabeth KrogBiomedical Scientist
- Martin KöhlerResearch Specialist
- Ingo LeibigerSenior Research Specialist | Docent
- Barbara LeibigerSenior Research Specialist | Docent
- Riccardo LizzaniPhd Student
- Noah MoruzziResearch Specialist
- Nuria Oliva VilarnauPostdoctoral Researcher
- Pere Rehues MasipPostdoctoral Researcher
- Yue ShiResearch Specialist
- Heidi StensmyrenAffiliated to Research
- Yvonne Linnea StrömbergLaboratory Assistent
- Philip Jonathan TrösterResearch Specialist
- Montserrat Visa MajoralResearch Specialist
- Zhenghao WangPostdoctoral Researcher
- Yan XiongResearch Specialist
- Guang YangResearch Assistant
- Shao-Nian YangSenior Research Specialist | Docent
- Lina YuResearch Assistant
- Kaixuan ZhaoPostdoctoral Researcher
- Valéry ZwillerVisiting Professor
- Dimitri van SimaeysResearch Specialist
- Christel Maria ÅgrenAdministrator
- Claes-Göran ÖstensonProfessor Emeritus
Publication list P-O Berggren
Publication List, pdf
News Signal Transduction
New method provides unique insight into the body's insulin production
11 November, 2025
Researchers at Karolinska Institutet have developed a new method for studying the body's insulin-producing cells in mice. By placing small parts of the pancreas in an unexpected place in the body, the researchers can follow the work of the cells in detail. The study, which has been published in Nature Communications, may eventually contribute to better treatments for diabetes.
Researchers use the eye as a window to study liver health
1 February, 2024
Researchers at Karolinska Institutet have developed a method to study liver function and disease without requiring invasive procedures. After transplanting liver cells into the eye of mice, the cornea can be used as a window into the body to monitor liver health over time. The study is published in the journal Nature Communications.
Eye implant may be used to treat diabetes
18 October, 2023
Researchers at Karolinska Institutet and KTH Royal Institute of Technology have developed a microscale device for implantation in the eye, which presents new opportunities for cell-based treatment of diabetes and other diseases. The study is published in the journal Advanced Materials.
New strategy to preserve insulin-producing cells in diabetes
30 March, 2022
High blood glucose is responsible for several complications in type 1 and type 2 diabetes. Researchers at Karolinska Institutet have identified a new antidiabetic substance that preserves the activity of insulin-producing beta cells and prevents high blood glucose in mice. The study is published in the journal Science Translational Medicine.
The Crown Princess visits Rolf Luft Research Center at MMK
22 November, 2021
On November 9, 2021, the Rolf Luft Research Center for Diabetes and Endocrinology, Karolinska Institutet, was visited by the Crown Princess Victoria. Ongoing research on diabetes was presented to her and a visit to the laboratory was also included.
Could islet transplantation to the eye help stabilise the glucose levels of people with type 1 diabetes?
28 September, 2021
Led by Professor Per-Olof Berggren at Karolinska Institutet in Sweden, a Phase 1 clinical trial is intended to reveal whether islet transplantation to the anterior eye chamber of people with type 1 diabetes could help restore insulin secretion to maintain glucose levels. The Novo Nordisk Foundation has awarded Karolinska Institutet up to DKK 10 million for the project.
Metabolic derangements caused by a high-fat diet may be possible to eliminate
12 March, 2021
Intake of a high-fat diet leads to an increased risk for obesity, type 2 diabetes, cardiovascular diseases and fatty liver. A study in mice from Karolinska Institutet shows that it is possible to eliminate the deleterious effects of a high-fat diet by lowering the levels of apolipoprotein CIII (apoCIII), a key regulator of lipid metabolism. The study is published in the journal Science Advances.
Findings about hair-like structures on cells inside vessels may be relevant for diabetes treatment
4 December, 2020
A new study from Karolinska Institutet and the Helmholtz Diabetes Research Center shows that primary cilia, hair-like protrusions on endothelial cells inside vessels, play an important role in the blood supply and delivery of glucose to the insulin-producing beta cells in the pancreatic islets. The findings are published in eLife and may be relevant for transplantation therapies in diabetes, as formation of functional blood vessels is important for the treatment to be successful.
KI receives over SEK 440 million from Swedish Research Council for medical research
9 November, 2020
The Swedish Research Council has awarded five-year grants totalling SEK 442,680,000 to 110 researchers at Karolinska Institutet. All in all, the SRC is awarding almost SEK 1 billion to 247 researchers in the fields of medicine and health.
Calcium channels play a key role in the development of diabetes
23 December, 2019
Researchers at Karolinska Institutet have deciphered the diabetogenic role of a certain type of calcium channel in insulin-secreting beta cells. The researchers believe that blockade of these channels could be a potential new treatment strategy for diabetes. The study is published in the scientific journal PNAS.
New clues to the link between ALS and type 2 diabetes
9 December, 2019
Patients with ALS (amyotrophic lateral sclerosis) often suffer from type 2 diabetes. This phenomenon has since long remained mechanistically enigmatic. Now, researchers at Karolinska Institutet in Sweden have identified a molecular mechanism linking these two diseases. The study is published in the scientific journal PNAS.
New eye monitoring system developed to monitor islet transplantation
26 November, 2019
A collaboration between Professor Per-Olof Berggren from the group Signal Transduction and researchers in the Republic of Korea has led to the development of a soft, smart contact lens to monitor the intraocular pressure and applied it for noninvasive monitoring in association with the intraocular islet transplantation in diabetes.
Multimillion grant from SSF
5 September, 2019
Two researchers at Karolinska Institutet, Per-Olof Berggren and Birgitta Henriques-Normark, receive a total of SEK 68 million in grants from the Swedish Foundation for Strategic Research, SSF. The Med-X research program is an interdisciplinary initiative in medicine and technical science. The purpose is to provide new solutions for clinical needs.
Novel findings explain indirect regulation of glucose homeostasis
16 August, 2019
The hormone secreting part of the pancreas, the islets of Langerhans, has a unique cyto-architecture that allows functional interrelationships between the different cell types. Somatostatin is secreted by the delta cell and is an effective inhibitor of the insulin secreting beta cell and the glucagon secreting alpha cell. According to a novel study from Karolinska Institutet, published in the journal Nature Communications, the delta cell can thereby indirectly affect glucose homeostasis.
Transplanted cells reveal early signs of type 1 diabetes
15 May, 2019
By the time type 1 diabetes is diagnosed, most of the insulin-producing beta cells have already been destroyed. Now, using an innovative transplantation technique, researchers at Karolinska Institutet and University of Miami Miller School of Medicine have been able to intervene to save the beta cells in mice by discovering early signs of the disease. The study is published in Diabetologia, the scientific journal of the European Association for the Study of Diabetes (EASD).
ERC grant for research on insulin-producing cells
28 March, 2019
The ERC Advanced Grant is one of the most prestigious funding programmes for research in Europe. Per-Olof Berggren, professor of experimental endocrinology at Karolinska Institutet, is now awarded this grant for the second time.
Per-Olof Berggren elected member of the Korean Academy of Science and Technology
31 May, 2018
Per-Olof Berggren, Professor of Experimental Endocrinology and Research Group Leader of the group Signal Transduction at the Department of Molecular Medicine and Surgery has been elected member of the Korean Academy of Science and Technology, KAST.
The body’s “glucostat” identified
06 Mar 2018
It is the pancreatic islets that have the overall responsibility for maintaining normal blood glucose levels in our bodies, according to a new study by researchers at Karolinska Institutet in Sweden and the University of Miami Miller School of Medicine, USA. The findings, published in the...
Method developed that gives researchers better conditions for studying insulin-producing cells
09 Aug 2017
Researchers have established a unique method enabling them to study the function of insulin-producing cells under conditions that are similar to those in humans. This can pave the way to development of new medicines for the treatment of diabetes. Beta cells in the pancreas produce the hormone...
KI professor shares award with historical celebrities
18 Apr 2017
Per-Olof Berggren, professor of experimental endocrinology at KI, has been elected member of the American Academy of Arts & Science. He share this distinguished award with historical celebrities, such as Albert Einstein, Winston Churchill and Nelson Mandela. Professor Per-Olof Berggren is a...
Per-Olof Berggren appointed Distinguished Professor in Italy
24 Oct 2016
Professor Per-Olof Berggren has been appointed Distinguished Professor at the University of Rome Tor Vergata, Italy. Per-Olof Berggren , Professor of Experimental Endocrinology and Research Group Leader of the group Signal Transduction at the Department of Molecular Medicine and Surgery. Research...
Professor Per-Olof Berggren elected member of the National Academy of Medicine
18 Oct 2016
Per-Olof Berggren , Professor of Experimental Endocrinology at the Department of Molecular Medicine and Surgery, Karolinska Institutet, has been elected as member of the National Academy of Medicine, NAM. The National Academy of Medicine announced the election of 70 U.S. members and 9 international...
New non-invasive method for studying the development of insulin-producing cells
04 Apr 2016
Researchers at Karolinska Institutet and Nanyang Technological University in Singapore have produced a unique method for monitoring the development of insulin-producing cells, which play a key part in regulating blood glucose concentration. The method, which is described in the journal Diabetologia...
Insulin-secreting cells report on insulin resistance
22 Feb 2016
Diabetes researchers at Karolinska Institutet have developed a novel technique that makes it possible to monitor insulin resistance in a non-invasive manner over time in mice. The new method, presented in the journal Scientific Reports , can be used to assess insulin resistance during progression...
Type 2 diabetes drug can exhaust insulin-producing cells
12 Feb 2016
Long-term use of liraglutide, a substance that helps to lower blood sugar levels in patients with type 2 diabetes, can have a deteriorating effect on insulin-producing beta cells, leading to an increase in blood sugar levels. This according to a study on mice implanted with human insulin-producing...
Novel mechanism of insulin resistance in type 2 diabetes
17 Sep 2015
Insensitivity to insulin, also called insulin resistance, is associated with type 2 diabetes and affects several cell types and organs in the body. Now, scientists from Sweden’s Karolinska Institutet have discovered a mechanism that explains how insulin-producing cells can be insulin resistant and...
New knowledge about how type 2 diabetes develops
19 May 2015
An international research team, led from Sweden’s Karolinska Institutet, have presented new knowledge about what happens when type 2 diabetes develops. By studying the insulin-producing beta cells in mice in real time, they have managed to identify a key part of the process that leads to the death...
Young vessels rejuvenate aged insulin-producing beta cells
18 Nov 2014
A recent study published in the journal PNAS shows that young capillary vessels rejuvenate aged pancreatic islets. The finding challenges prevailing views on the causes of age-dependent impaired glucose balance regulation, a condition that often develops into diabetes type 2. The international...
Study explains the link between cilia and diabetes
06 Nov 2014
Tiny extensions on cells, cilia, play an important role in insulin release, according to a new study, which is published in Nature Communications . The researchers report that the cilia of beta cells in the pancreas are covered with insulin receptors and that changed ciliary function can be...
Mechanism behind age-dependent diabetes discovered
17 Sep 2014
Ageing of insulin-secreting cells is coupled to a progressive decline in signal transduction and insulin release, according to a recent study by researchers at Karolinska Institutet in Sweden. The finding, which is published in the journal Diabetes , provides a new molecular mechanism underlying...
KI researchers win Wallenberg Scholarship
12 Dec 2012
The Wallenberg Scholars programme supports and stimulates some of the most successful researchers at Swedish universities. Two researchers at Karolinska Institutet have now been awarded SEK 15 million each to be divided over a period of five years. Per-Olof Berggren Per-Olof Berggren, professor of...
Prizes awarded for diabetes and metabolism research
19 Sep 2012
Two researchers at Karolinska Institutet are to be rewarded for their scientific achievements, which are potentially of major significance to the treatment of diseases such as type II diabetes. Professor Per-Olof Berggren receives the Axel Hirsch Prize for his discoveries on insulin release in the...
Associated International Labs
Diabetes Research Institute
University of Miami Miller School of Medicine
Department of Surgery
1450 NW 10th Avenue
Miami, FL 33136
USA
Since 2002
Pohang University of Science and Technology
Department of Life Sciences
Gyeongbuk 37673
Pohang
Republic of Korea
Since 2010
Singapore Eye Research Institute SERI
The Academia, 20 College Rd
Level 6 Discovery Tower
Singapore 169856
Since 2013
West China Hospital
Sichuan University
No.37 Guoxue Alley
Wuhou District, Chengdu City
Sichuan Province
PR China
Since 2019
Ulster University
School of Biomedical Sciences Research
Coleraine campus
Cromore Road
Coleraine
Co. Londonderry
BT52 1SA
United Kingdom
Since 2020
Tecnológico de Monterrey
Hospital Zambrano Hellion 2° piso
Av. Batallón de San Patricio 112, Real San Agustín, 66278
San Pedro Garza García, N.L.
Mexico
Since 2021