Centre for Pharmacoepidemiology
The Centre for Pharmacoepidemiology (CPE) works in a dynamic environment at the intersection of academia, healthcare, authorities and the pharmaceutical industry.
We conduct academic research on drug safety, drug effectiveness and drug utilization and perform regulator-mandated Post-Authorisation Safety Studies (PASS) and Drug Utilization Studies (DUS) in the area of pharmacoepidemiology and real-world evidence (RWE).
 Photo: Annika Evolahti
Photo: Annika EvolahtiMenu for this area
News
 Photo: NA
Photo: NASIGMA Consortium Board meeting
At the end of May, CPE hosted the SIGMA Consortium Board meeting with its 15 members across Europe. Discussions focused on the fast-changing environment as well as SIGMA’s continued long-term commitment to high-quality scientific research.
 Photo: N/A
Photo: N/AEmerging Topics in Perinatal Pharmacoepidemiology
Welcome to an afternoon symposium on emerging topics in perinatal pharmacoepidemiology.
This event is open to students, postdocs, researchers, and clinicians within the KI community and surrounding institutions with an interest in medication use and vaccination during pregnancy, perinatal drug safety, and collaborative pharmacoepidemiological research.
Events Archive

Master’s thesis projects at CPE
There are open opportunities for master’s thesis projects at CPE within the areas of dermatology and perinatal pharmacoepidemiology.
Please contact CPE@MedS.ki.se.

Welcome Fredh to CPE!
Fredh Netterström Wedin has joined CPE as a PhD student in perinatal pharmacoepidemiology under the primary supervision of Cathrine Axfors. Fredh holds a Master’s degree in Public Health and has previously worked at a research consultancy focusing on register based pharmacoepidemiology. In addition to register based drug research, he has a strong interest in meta-research and evidence synthesis, areas in which he has published studies with both methodological and clinical focus. At CPE, Fredh will conduct research on medication use and drug safety during pregnancy, combining methodological studies with applied register based pharmacoepidemiological research.

Congratulations Adam to his dissertation!
We are happy to announce that Adam Nygren successfully defended her thesis ”Pharmacoepidemiological studies on depression – treatments, treatment-resistance and consequences” on 30th of January 2026.

Congratulations to the CPE team members for their presentations at NorPEN!
At this year´s NorPEN conference, CPE was well represented with several oral presentations:
Julia Eriksson presented “ Modeling Clinical Indications for GLP-1 receptor agonists in Sweden using machine learning classification”
Silvia Segovia Chacón shared her work on “First trimester melatonin exposure and risk of major congenital malformations: a population-based cohort study”
In addition, Felix Wittström received the award for Best PharmacoEPISLAM presentation for his study “Prevalence and characteristics of woman with bipolar disorder discontinuing lithium treatment in pregnancy: leveraging prescription free-text information in Sweden”.

Welcome Stamatia to CPE!
Stamatìa Tsampà has joined CPE as a doctoral student in perinatal pharmacoepidemiology under the main supervision of Cathrine Axfors. Stamatìa is a medical doctor and holds a master’s degree in Public Health Sciences, KI. During her thesis, she was working on discovering trends and patterns in medication use during pregnancy based on the Swedish registers and is joining CPE to contribute on systematic exploration of pregnancy drug safety using tree-based scan statistics.

Therapy more effective than pills for sleep problems
Read the interview with Johan Reutfors, psychiatrist and associate professor at CPE, in the article Therapy more effective than pills for sleep problems

Welcome Cathrine to CPE!
Cathrine Axfors recently joined CPE as an Assistant Professor. Cathrine is a medical doctor with a PhD in perinatal psychiatry at Uppsala University, and she just came back to Sweden after two international postdocs in meta-research/evidence synthesis. Her previous research covers perinatal epidemiology, clinical trial transparency, and evidence synthesis for COVID-19. At CPE, Cathrine will start up a new project on systematic exploration of pregnancy drug safety in Swedish registers using tree-based scan statistics.

Patterns of Sexual Dysfunction in Depression: New Findings from a Swedish Population-Based Study
At this year’s conference of the European Psychiatric Association in Madrid, Josef Isung presented findings from our ongoing study on sexual dysfunction in depression. The results show that the incidence of sexual dysfunction, including cases treated pharmacologically, is highest in the first year following a depression diagnosis and increases with age.

Top cited article
Congratulations to Silvia, Pär and Carolyn for their paper “Detection of major congenital malformations depends on length of follow-up in Swedish National Health Register Data: Implications for pharmacoepidemiological research on medication safety in pregnancy”, that is among the top 10 most-cited papers published in Paediatric and Perinatal Epidemiology in 2023.

Welcome Essi to CPE!
Essi Whaites Heinonen is new postdoc at CPE who will be working with Carolyn Cesta at 50% combined with clinical work as a neonatologist at Karolinska University Hospital. With her planned studies at CPE, Essi aims to create a neonatology-arm within the group, focusing on the neonatal effects after maternal use of medications during pregnancy.

Welcome Awad to CPE!
Awad Smew is new postdoc at CPE who will be working with Carolyn Cesta. His main interest lies in optimising safety of maternal care through pharmacoepidemiologic studies of drug use during pregnancy as well as understanding effects of the pregnant women exposed to surgery, anaesthesia, or intensive care.
This relates to Awad´s clinical work as a physician in anaesthesia and intensive care medicine at Karolinska University Hospital Solna.

Welcome to CPE's master student Asina!
Asina is a public health master's student from Lunds University. This last semester of her master studies, Asina wants to specialize in the field of epidemiology as this is the area of public health she is most passionate about. She will be supervised by Marie Linder and David Hägg during her thesis work, were she will investigate the association between malignant melanoma and atopic dermatitis. Asina completed her undergraduate degree in the field of biological sciences, with a major in genetics.

Welcome to CPE's master student Ingunn!
Ingunn is a master’s student in Mathematical Statistics at Stockholm University. She will be writing her master’s thesis under the supervision of Pär Karlsson and Marie Linder, focusing on causal survival analysis. She completed her BSc in Applied Mathematics, majoring in statistics and data science in Reykjavík, Iceland.

In Memoriam Helle Kieler 1956-2024
In memory of Helle Kieler, former CPE director who passed away last fall, an In Memoriam has been written and published in the journal “Pharmacoepidemiology and Drug Safety”.
The memorial article can be found via the following link; https://doi.org/10.1002/pds.70073

Congratulations Julia on beginning your doctoral studies!
Julia Eriksson joined CPE as a statistician in June 2023 and will start her next chapter as a PhD student in January under the supervision of Marie Linder and other CPE/KEP researchers. Her project will investigate the exposure of glucagon-like peptide-1 (GLP-1) analogues in Sweden, focusing on use, indication, safety and efficacy. Julia has a Master's degree in Mathematical Statistics from Stockholm University and has worked as a statistician at Karolinska Institutet since 2021.

Grant funding received for studies on medication use in pregnancy
CPE’s assistant professor Carolyn Cesta and affiliated researcher Cathrine Axfors have received a total of 17 million Swedish kronor in funding from individual starting grants from the Swedish Research Council (VR) and a project grant from the Swedish Research Council for Health, Working Life and Welfare (FORTE). The funding will be used for projects investigating the safety of medication use in pregnancy using Swedish national register data in collaboration with Nordic and international collaborators.

Course in pharmacoepidemiology
We are happy to inform that CPE will arrange a course in pharmacoepidemiology 17-21 February 2025, “Drug exposure Definitions and Study Design Approaches in Register-based data”.
The application period for the course is from 15 October to 5 November. https://doctoralcourses.application.ki.se/fubasextern/info?kurs=K2F6033

Professor emeritus and former centre director Helle Kieler has passed away
Helle Kieler, professor emeritus and former centre director Helle Kieler , passed away 1 September at the age of 68.
From 2006 to 2023, Helle led the work at CPE and under her leadership CPE developed into one of Europe´s largest pharmacoepidemiologic centre.
Helle has made a lasting mark on the field of pharmacoepidemiology with her research into the safety, effectiveness and utilization of medication, Nordic/international collaboration and the establishment of collaborations between academia, health care, industry and authorities.
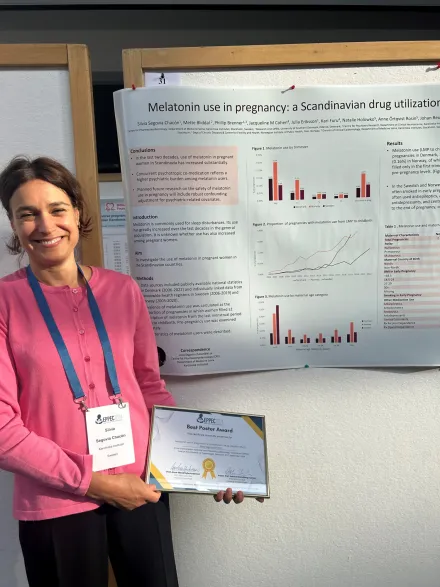
Prize for best poster at EPPEC
At the European Perinatal and Paediatric Epidemiology Conference (EPPEC), Copenhagen, 16-17 September, Silvia Segovia Chacón was awarded the prize for the best poster.
Her poster was titled: Melatonin use in pregnancy: a Scandinavian drug utilization study.

Congratulations Aya to her dissertation!
We are happy to announce that Aya Nakitanda successfully defended her thesis “ Antibiotics in pregnancy: utilization, determinants and safety” on 13th of June 2024.
Aya Nakitanda, Antibiotics in pregnancy:utilization, determinants and safety (2024-06-13)

2024 ISPE Annual Meeting
CPE will be attending, moderating and presenting at ICPE 2024, Berlin. For poster presentations by CPE employees Diego H. Giunta, David Hägg, Marie Linder and Felix Wittström , see ICPE web page https://2024ispe.eventscribe.net/posterspeakers.asp?pfp=BrowsebySpeaker

CPE joins The SIGMA consortium
CPE, Karolinska Institutet has recently joined The SIGMA consortium as full members. The SIGMA consortium is a alliance of ENCePP research centres with the aims to integrate expertise and data from multiple European Research Centres. The vision is to provide best evidence on the use, benefits and harms of medications, vaccines and devices for the betterment of public health and patient care.

InPreSS consortium study, lead by CPE’s Carolyn Cesta, cited in The New York Times
A recent study by the International Pregnancy Safety Study (InPreSS) consortium, was cited in The New York Times in a piece on the use of weight-loss drugs before pregnancy. Ozempic and other GLP-1 receptor agonists, recently gained regulatory approval for treatment of obesity, in addition to their original indication, Type 2 diabetes.

Newer diabetes drugs do not increase fetal risk
Congratulations to Carolyn Cesta and her collaborators on the new publication in JAMA Internal Medicine. The study showed that newer diabetes medicines do not appear to increase the risk of birth defects. The largest comparative study to date found no increased risk compared to treatment with insulin, which is considered safe during pregnancy.

Congratulations Silvia on beginning your doctoral studies!
Silvia Segovia Chacón joined CPE in 2019 as a research assistant and then project manager. This September she starts her next chapter with CPE as a PhD student under the supervision of Carolyn Cesta and other CPE/KEP researchers. Her project will focus on medication utilization and safety in reproductive aged women and during pregnancy, which complements her background as a midwife and interest in women’s health.

Welcome Julia Eriksson to CPE!
Julia is a statistician with a master’s degree in mathematical statistics from Stockholm University. In the last two years, she has worked at the Division of Biostatistics at the Institutet of Environmental Medicine, Karolinska Instiutute. She has experience of using different types of regression models, meta analysis and some machine learning models. Julia has been involved in several types of research groups on sleep, domestic violence, primary care studies, stroke, cancer etc. She will join the team of statisticians at CPE to contribute to pharmacoepidemiologic research to improve public health.

CPE researcher elected as co-chair to the Nordic Pharmacoepidemiological Network (NorPEN)
Carolyn Cesta, assistant professor at CPE, has been elected as the co-chair of the Nordic Pharmacoepidemioloigcal Network (NorPEN) https://www.norpen.org/. NorPEN was created in 2008 and is a network academic research groups, public authorities, and national register holders with the purpose of facilitating research within the field of pharmacoepidemiology in the Nordic countries. NorPEN hosts an annual conference and in recent years has focused on providing educational opportunities (e.g., courses, seminars) for trainees in the Nordic countries.

Newly published study in antipsychotic-exposed pregnancies
A new study published in JAMA Psychiatry by the International Pregnancy Safety Study (InPreSS) Consortium, including researchers at CPE and colleagues in Denmark, Finland, Iceland, Norway, and the US, showed that in a cohort of more than 26,000 antipsychotic-exposed pregnancies, prenatal exposure to antipsychotics was generally not associated with an increase in risk of major congenital malformations. This study was a multi-year undertaking initiated by CPE director Helle Kieler, focusing on how best to apply a uniform study design and analytic approach across many countries to be able to study uncommon exposures (e.g., individual drugs) and rare outcomes (e.g., specific malformations subtypes) in detail.

Together with Scandinavian collaborators, Johan Reutfors, psychiatrist and researcher at the CPE, have found a considerably higher prescription of antidepressants for children and adolescents in Sweden than in Denmark and Norway.
The pattern was similar for sleeping medication. The researchers are now moving on to try to find factors behind the differences. More about this has been reported by the Swedish Radio and the Aftonbladet Daily news podcast.

Welcome to Felix Wittström to CPE!
Felix has joined CPE as a doctoral student in reproductive and psychiatric pharmacoepidemiology, under the main supervision of Carolyn Cesta. His projects will investigate psychotropic drug use during pregnancy and how discontinuation of, or interactions between, medications used in bipolar disorder can affect mother and child. Felix’s background is in medicine and he has started residency training as a doctor in clinical pharmacology.

CPE researcher receives SRP Diabetes Postdoc Fellowship Programme
Laura Pazzagli at CPE is one of the 5 Principal Investigators who has received funding from the Strategic Research Programme (SRP) in Diabetes Postdoc Fellowship Programme. With this postdoctoral program SRP Diabetes aims to support the next generation of exceptional young scientists within the diabetes area. The funded project aims to investigate the impact of socioeconomic factors on antidiabetic medications use and discontinuation and type 2 diabetes consequences such as cardiovascular problems, kidney failure and retinopathy. Read more about the project: https://ki.se/en/srp-diabetes/srp-diabetes-postdoc-fellowships-project-4
Interested candidates for the postdoctoral position ,can find the advertisement https://ki.varbi.com/en/what:job/jobID:487660/type:job/where:4/apply:1

EMA call
CPE has together with University of Milano-Bicocca won a EMA call and is on EMA´s short list of eligible research partners for pharmacoepidemiology studies; statistical research.

CPE researcher´s article in a list among 30 must-read papers within pharmacoepidemiology
Laura Pazzagli´s paper “Methods for time-varying exposure related problems in pharmacoepidemiology: An overview” is listed among Pottegård et al.´s list over 30 must-read papers for newcomers to pharmacoepidemiology.

CPE researcher receives a large Forte junior research grant
We are happy to announce that Laura Pazzagli at CPE has received a Forte junior researcher grant of 5 985 000 SEK for the project “Understanding the role of socioeconomic factors on antidiabetic medication use patterns and long-term consequences of type 2 diabetes via novel causal inference methods.”

NorPEN 2021
The 13th Annual Nordic Pharmacoepidemiological Network (NorPEN) meeting was held as an online meeting in November.
Thank you all who participated!

Welcome Diego
Welcome Diego Hernan Giunta to CPE!
Diego is a medical doctor, specialized in internal medicine, and he has for the last 15 years been working as an epidemiologist at Hospital Italiano de Buenos Aires/ the Instituto Universitario del Hospital Italiano de Buenos Aires.
Diego says "I decided to join CPE to be an active component of a leading pharmacoepidemiologic research team with strong public health and teaching trajectory. I would like to keep on learning and enhancing our team, by been actively engaged in pharmacoepidemiologic research to improve patients effectiveness and safety”

ConcePTION
Together with our Nordic partners, CPE contributes to better drug safety for pregnant women through participation in the ConcePTION project and many other projects.

ADHD and pregnancy
Read the interview with Johan Reutfors, psychiatrist and researcher at CPE/Karolinska Institutet, were he gives an overview over the research performed related to ADHD and pregnancy.

Register-based research course
CPE and Copenhagen University organized the course “Register-based research course: Pharmacoepidemiology - drug use and safety” held at the University of Copenhagen in 2020.

COVID-19 research
CPE participates as one of 18 organizations in the CONSIGN (Covid-19 infectiON and medicineS In pregnancy) project which is initiated by EMA as an infrastructure to support the monitoring of efficacy and safety of COVID-19 treatments and vaccines when used in day-to-day clinical practice.

NorPen Webinar 2020
November 11th 2020, a short webinar was held for the NorPEN community.

Narcolepsy drug did not increase risk of fetal malformation
“This study is based on twice as many pregnancies as earlier studies, and we find no increase in the risk of malformation in infants exposed to modafinil during pregnancy,” says the study’s lead author Carolyn Cesta, researcher at CPE/KI.

Congratulations to CPE’s three masters students
From left to right:
Chaitra Srinivas, Lamya Abdi, Liwei Zhao

Joining forces to improve safety of drugs during pregnancy
CPE is part of the pan-European public private partnership ConcePTION. The overarching aim is to improve safety of drugs during pregnancy and lactation.

Treatment resistant depression increases risk for substance use disorders
Read the recently published paper in Addiction by Philip Brenner et al.

Best reviewer 2018 for Pharmacoepidemiology and Drug Safety
CPE researcher Laura Pazzagli was in August rewarded as Best reviewer 2018 for Pharmacoepidemiology and Drug Safety.

CPE postdoc Carolyn Cesta awarded MSCA Individual Fellowship!
Carolyn Cesta has been awarded the prestigious EU Horizon 2020 Marie Skłodowska-Curie Actions Individual Fellowship to investigate the efficacy and safety of prescribed drugs used for treatment of diabetes during pregnancy.

CPE researcher Laura Pazzagli awarded!
American College of Epidemiology (ACE) has September 25, 2018, awarded postdoctoral researcher Laura Pazzagli for the Best Paper Published by a Junior Investigator in the Annals of Epidemiology.

Top 20 most downloaded recent papers!
CPE researcher Laura Pazzagli’s article Methods for time-varying exposure related problems in pharmacoepidemiology: An overview, Pharmacoepidemiology and Drug Safety, was top 20 most downloaded article the last year.

Treatment-resistant depression increases the mortality
Young adults between 18-29 years with treatment-resistant depression (TRD) have more than double the risk of all-cause mortality compared with patients with other types of depression. This shows results published in the Journal of Affective Disorders which has also been reported in the Swedish newspaper Aftonbladet.
