About COMBAT-MS
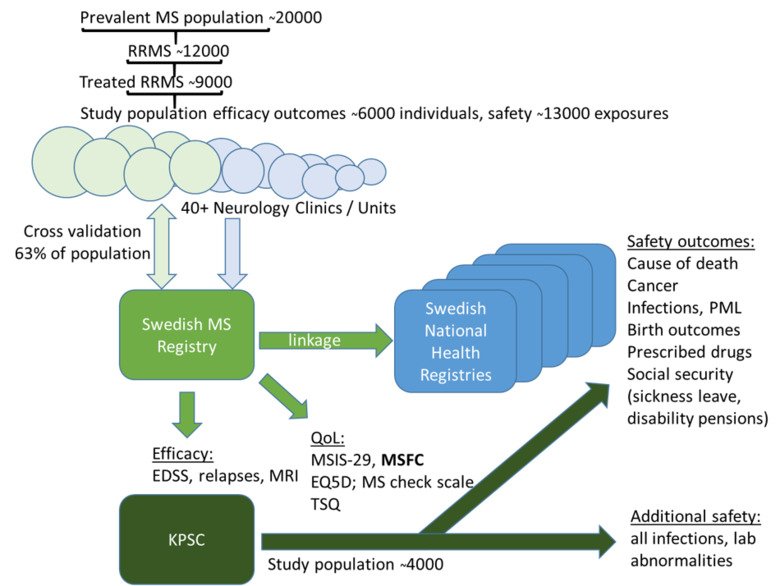
COMBAT-MS is a PCORI granted project with the objective to compare the efficacy and safety of rituximab (RTX) in comparison with other disease modifying treatments (DMTs) in multiple sclerosis (MS). It is a collaboration between Karolinska Instiutet in Stockholm, Sweden and Kaiser Permanent Southern California in Pasadena, CA, USA.
While not formally approved for MS, RTX offers several potential advantages over other MS DMTs, as clinical studies have shown that RTX is potent, relatively safe, convenient, and available worldwide, and the duration of benefit extends far beyond the clearance of the drug. Thus, it is often a good choice for patients requiring escalation therapy, or even as a first-line agent in those planning pregnancy or with healthcare access issues.
However, long-term efficacy and safety in relapsing remitting MS (RRMS) are still unknown, and these data together with quality-of-life measurement needs to be investigated. The two key decisional dilemmas we will study in COMBAT-MS are the following:
- How RTX compare with other DMTs in those patients who have already had continued disease activity on a previous DMT
- How RTX compare with other DMTs in those patients faced with choosing their first MS medication.
RTX is being increasingly used as treatment for MS in Sweden and the United States. With the help of the largest publically funded population-based registry of MS patients, we propose to utilize the Swedish Neuro Registry to address these aims in the Swedish cohort. As this registry was established in 2001 with the explicit purpose of studying long-term benefits of DMTs, we can in the beginning of this study identify a large cohort that has at least three years of treatment with RTX or other DMTs. By including new patients over a five-year period, we are confident that we can retrieve data from 6,000 patients, of whom close to 4,000 will be followed in a yearly follow-up program over three to nine years after initiation of therapy, triggering inclusion into the study. We will continue to collect yearly standardized measures of disability and quality of life to address these aims. Safety data will be combined with the multiethnic cohort of Kaiser Permanent Southern California. The contribution of the stakeholders will continue to be essential in the design, conduct, dissemination, and implementation of this study. By studying RTX, a drug that is accessible worldwide, and reporting outcomes that matter to patients and stakeholders, results from the proposed study have the potential to improve the care for RRMS patients throughout the world.
