Endocrine and age aspects of lymphoma biology - Sam Okret
Lymphomas are not generally considered as endocrine-regulated malignancies but epidemiological information and experimental data collected by us strongly suggest they are, particularly by sex hormones.
The incidence of most adult lymphoma is higher in males than in females. Furthermore, male sex generally confers a worse prognosis. The sex difference in incidence is even more pronounced for lymphomas in children and young adults. The cause of this difference is not understood and so far not well studied. The aims of our projects are to identify molecular mechanisms and biological processes involved in the sex and age differences of lymphomas with a focus on sex hormones.
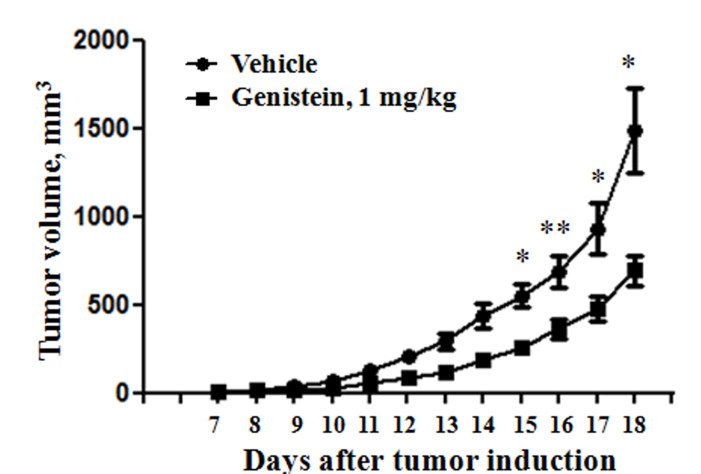
For example, we have shown that transplanted lymphoma tumours grow faster in male than female mice, a difference that was removed following ovariectomy. Furthermore, we showed that several lymphomas are highly sensitive to compounds that bind to oestrogen receptor β (ERβ) subtype, highly expressed in lymphoma cells, and cause an inhibition of tumour growth in vivo, reduce tumour vascularisation and inhibit dissemination. Contrary to this, inhibition of oestrogen synthesis promotes lymphoma progression (4). ERβ expression in lymphomas may also have a prognostic value (3). We also show that xenoestrogens (food and other compounds in the environment with oestrogen activity) administered to mice inhibit lymphoma growth in grafted mice (1). Taken together this indicates that oestrogens do indeed play a role in lymphoma development. Future research aims to identify molecular mechanisms involved in sex and age dependent differences in lymphoma development and with this knowledge to identify molecular targets for new and more personalised treatment which takes sex and age into consideration.
In addition, we have also studied mechanisms of drug resistance on lymphoma by the tumour microenvironment and how this resistance can be overcome (2).
Group members
Sam Okret
Group leader and ProfessorSelected publications
- Yakimchuk, K. Bangalore Revanna, C., Huang, D., Inzunza, J. and Okret, S. (2018) ‘Suppression of lymphoma growth by the xenoestrogens bisphenol A and genistein.’ Endocrine Connections, 7(12): 1472–1479, 2018.
- Guan, J., Huang, D., Yakimchuk, K. and Okret, S. (2018) ‘p110α inhibition overcomes stromal cell-mediated ibrutinib resistance in mantle cell lymphoma.’ Mol. Cancer Therap. 17, 1090-1100, 2018.
- Hasni, M.S., Berglund, M., Yakimchuk, K., Guan, J., Linderoth, J., Amini, R.M., Enblad, G. and Okret, S. (2017) ‘Estrogen receptor β1 in diffuse large B-cell lymphoma growth and as a prognostic biomarker.’ Leukemia & Lymphoma, 58, 418-427, 2017.
- Talabér, G., Yakimchuk, K., Guan, J., Inzunza, J. and Okret, S. (2016) ‘Inhibition of estrogen biosynthesis enhances lymphoma growth in mice.’ Oncotarget, 7, 20718-20727, 2016.
Are you looking for a MSc project or internship?
If you find what we are working with interesting and matching your profile, you are welcome to contact the group leader for more information.
