Epigenetic control of metabolism and inflammation by corepressors
Transcription factors (>1500) and associated coregulators (>300) play central roles in linking epigenome alterations to transcriptional reprogramming, as they shape the chromatin/enhancer landscape (i.e. the epigenome) and determine signal responses. Although these processes are well studied for many transcription factors and coactivators, our understanding of the precise roles of corepressors is currently lacking behind.
It is further poorly understood whether and how the dysregulation of specific coregulators contributes to, or potentially even causes, disease.
Eckardt Treuter
Group leaderTranscriptional coregulators are a heterogeneous class of epigenome-modifying proteins that function in larger complexes and cooperate with transcription factors at promoters and enhancers to regulate gene expression. Our search for candidates that might be particularly relevant for metabolic and inflammatory disease pathways revealed a potential key role of a fundamental corepressor complex, consisting of the core subunits histone deacetylase HDAC3, G-protein pathway suppressor 2 (GPS2), and the related corepressors NCOR and SMRT. Although these subunits were originally identified as nuclear receptor-associated proteins, we know today that the complex controls many other transcription factors and multiple cellular pathways.
Our studies have demonstrated the involvement of the complex in the regulation of cholesterol metabolism and transport, in anti-inflammatory crosstalk mechanisms, in adipose tissue inflammation and hypertrophy, and in hepatic fatty acid oxidation and lipogenesis. Intriguingly, alterations of GPS2 expression in adipocytes and macrophages associate with systemic inflammation and diabetic risk in obese humans, suggesting the identified pathways to be conserved and clinically relevant. Thus, our current hypothesis is that inappropriate complex function, in part due to altered expression and modification of the core subunit GPS2, triggers epigenetic reprogramming and thereby alters the susceptibility to metabolic-inflammatory signals that contribute to disease.
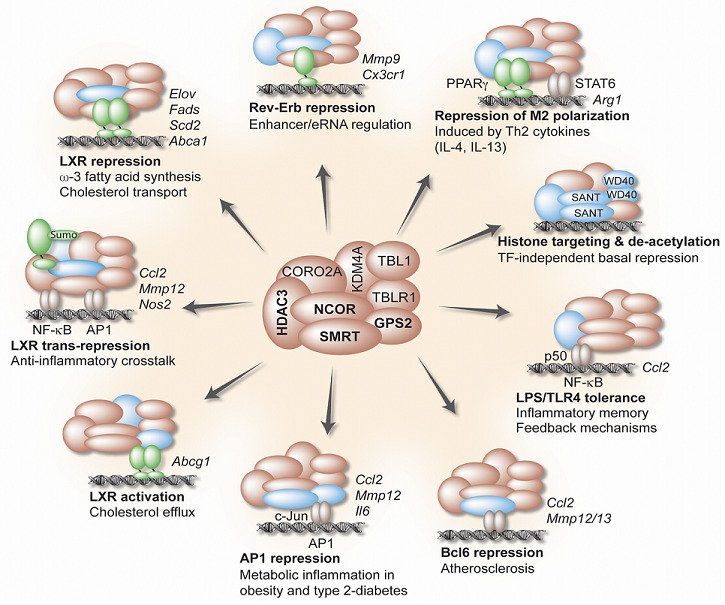
We now attempt to evaluate the hypothesis using a variety of genetically modified disease models, and in response to different dietary and pharmacological interventions. For example, we have begun to investigate the role of corepressor-mediated epigenome alterations in monocytes/macrophages in linking type 2 diabetes to the progression of atherosclerosis. In another project, we would like to understand how post-translational modifications of individual subunits modulate epigenomic and transcriptional activities of the corepressor complex (and postulated sub-complexes), and how these modifications change in response to different signals and during the development of type 2 diabetes, atherosclerosis and fatty liver disease.
Besides a better understanding of coregulator-mediated epigenome alterations during the development of metabolic-inflammatory diseases, we also expect novel insights into epigenetic memory and crosstalk mechanisms between metabolic ‘master’ cell types such as macrophages, adipocytes and hepatocytes.
Group members
Oihane Garcia-Irigoyen
Research SpecialistSviatlana Sukhanava
Phd StudentAstradeni Efthymiadou
Phd StudentZhiqiang Huang
Postdoctoral researcher, affiliated to researchLooking for a Postdoc, Master or PhD project?
We always welcome motivated Master students, PhD students and postdocs who have a relevant background and a keen interest in our research. Please contact eckardt.treuter@ki.se for potential projects and vacancies.
Research Networks
Funding
Our research is supported by grants from the Center for Innovative Medicine at Karolinska Institutet South (CIMED, Region Stockholm), the Swedish Research Council, the Novo Nordisk Foundation, the Swedish Cancer Society, the Swedish Diabetes Foundation, and by the doctoral education (KID) program at the Karolinska Institutet.
